Abstract
Background
The BCG vaccine has been traditionally administered to prevent TB. It has been additionally used in bladder cancer patients as a therapy with success. Some observational studies found that bladder cancer patients receiving BCG may have reduced dementia risk, however, the evidence is not conclusive.
Objective
To investigate the impact of BCG vaccine on dementia risk in bladder cancer patients.
Methods
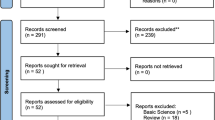
Six databases were searched from inception to January 13, 2024, for published and unpublished studies that examine the association between BCG and dementia risk in bladder cancer patients. We conducted meta-analyses using a random-effects model.
Results
Eight retrospective cohort studies were included in the systematic review and seven in the meta-analyses. Because there were studies with overlap** populations, two separate main analyses were performed reassuring the avoidance of overlap. The first analysis showed that compared to controls, BCG did not reduce dementia risk [5 studies pooled, n=88,852, HR = 0.65, 95% CI (0.40, 1.06), I2 = 85%] whereas there was a marginally significant risk reduction in the second analysis [6 studies pooled, n=70,025, HR = 0.63, 95% CI (0.40, 0.97), I2 = 83%]. Sensitivity analysis excluding the unpublished studies did not affect the outcome importantly. Additional meta-analysis showed that BCG did not reduce the risk of Alzheimer’s disease.
Conclusion
This meta-analysis of observational studies found that BCG administration in bladder cancer patients has likely a minimally positive impact on dementia risk if any. To better understand the effect of BCG on dementia, randomized controlled trials are needed.




Similar content being viewed by others
References
Nichols E, Steinmetz JD, Vollset SE, et al. Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the Global Burden of Disease Study 2019. The Lancet Public Health. 2022;7:e105–e125. DOI: https://doi.org/10.1016/S2468-2667(21)00249-8
Arvanitakis Z, Shah RC, Bennett DA. Diagnosis and Management of Dementia: Review. JAMA. 2019;322:1589. DOI: https://doi.org/10.1001/jama.2019.4782
Magrath Guimet N, Zapata-Restrepo LM, Miller BL. Advances in Treatment of Frontotemporal Dementia. J Neuropsychiatry Clin Neurosci. 2022;34:316–327. DOI: https://doi.org/10.1176/appi.neuropsych.21060166
Taylor J-P, McKeith IG, Burn DJ, et al. New evidence on the management of Lewy body dementia. Lancet Neurol. 2020;19:157–169. DOI: https://doi.org/10.1016/S1474-4422(19)30153-X
Iadecola C, Duering M, Hachinski V, et al. Vascular Cognitive Impairment and Dementia: JACC Scientific Expert Panel. J Am Coll Cardiol. 2019;73:3326–3344. DOI: https://doi.org/10.1016/j.jacc.2019.04.034
Kwon S, Iba M, Kim C, Masliah E. Immunotherapies for Aging-Related Neurodegenerative Diseases-Emerging Perspectives and New Targets. Neurotherapeutics. 2020;17:935–954. DOI: https://doi.org/10.1007/s13311-020-00853-2
Van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in Early Alzheimer’s Disease. N Engl J Med. 2023;388:9–21. DOI: https://doi.org/10.1056/NEJMoa2212948
Jorfi M, Maaser-Hecker A, Tanzi RE. The neuroimmune axis of Alzheimer’s disease. Genome Med. 2023;15:6. DOI: https://doi.org/10.1186/s13073-023-01155-w
De Strooper B, Karran E. The Cellular Phase of Alzheimer’s Disease. Cell. 2016;164:603–615. DOI: https://doi.org/10.1016/j.cell.2015.12.056
Bettcher BM, Tansey MG, Dorothée G, Heneka MT. Peripheral and central immune system crosstalk in Alzheimer disease - a research prospectus. Nat Rev Neurol. 2021;17:689–701. DOI: https://doi.org/10.1038/s41582-021-00549-x
Lange C, Aaby P, Behr MA, et al. 100 years of Mycobacterium bovis bacille Calmette-Guérin. Lancet Infect Dis. 2022;22:e2–e12. DOI: https://doi.org/10.1016/S1473-3099(21)00403-5
Lobo N, Brooks NA, Zlotta AR, et al. 100 years of Bacillus Calmette-Guérin immunotherapy: from cattle to COVID-19. Nat Rev Urol. 2021;18:611–622. DOI: https://doi.org/10.1038/s41585-021-00481-1
Ristori G, Romano S, Cannoni S, et al. Effects of Bacille Calmette-Guerin after the first demyelinating event in the CNS. Neurology. 2014;82:41–48. DOI: https://doi.org/10.1212/01.wnl.0000438216.93319.ab
Weinberg MS, Zafar A, Magdamo C, et al. Association of BCG Vaccine Treatment With Death and Dementia in Patients With Non-Muscle-Invasive Bladder Cancer. JAMA Netw Open. 2023;6:e2314336. DOI: https://doi.org/10.1001/jamanetworkopen.2023.14336
Wang E, Hagberg O, Malmström P-U. The association between BCG treatment in patients with bladder cancer and subsequent risk of develo** Alzheimer and other dementia.-A Swedish nationwide cohort study from 1997 to 2019. PLoS One. 2023;18:e0292174. DOI: https://doi.org/10.1371/journal.pone.0292174
Gofrit ON, Klein BY, Cohen IR, Ben-Hur T, Greenblatt CL, Bercovier H. Bacillus Calmette-Guérin (BCG) therapy lowers the incidence of Alzheimer’s disease in bladder cancer patients. Ginsberg SD, editor. PLoS ONE. 2019;14:e0224433. DOI: https://doi.org/10.1371/journal.pone.0224433
ABSTRACTS of ICPE 2022, the 38th International Conference on Pharmacoepidemiology and Therapeutic Risk Management (ICPE), Copenhagen, Denmark, 26–28 August, 2022: ORAL PRESENTATIONS. Pharmacoepidemiology and Drug. 2022;31:3–628. DOI: https://doi.org/10.1002/pds.5518
Han C, Wang J, Chen Y-L, Guan C-P, Zhang Y-A, Wang M-S. The role of Bacillus Calmette-Guérin administration on the risk of dementia in bladder cancer patients: a systematic review and meta-analysis. Front Aging Neurosci. 2023;15:1243588. DOI: https://doi.org/10.3389/fnagi.2023.1243588
Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. DOI: https://doi.org/10.1136/bmj.n71
ROBINS-E Development Group (Higgins J, Morgan R, Rooney A, Taylor K, Thayer K, Silva R, Lemeris C, Akl A, Arroyave W, Bateson T, Berkman N, Demers P, Forastiere F, Glenn B, Hróbjartsson A, Kirrane E, LaKind J, Luben T, Lunn R, McAleenan A, McGuinness L, Meerpohl J, Mehta S, Nachman R, Obbagy J, O’Connor A, Radke E, Savović J, Schubauer-Berigan M, Schwingl P, Schunemann H, Shea B, Steenland K, Stewart T, Straif K, Tilling K, Verbeek V, Vermeulen R, Viswanathan M, Zahm S, Sterne J). Risk Of Bias In Non-randomized Studies - of Exposure (ROBINS-E). Launch version, 20 June 2023. Available from: https://www.riskofbias.info/welcome/robins-e-tool.
Veroniki AA, Jackson D, Viechtbauer W, et al. Methods to estimate the between-study variance and its uncertainty in meta-analysis. Res Synth Methods. 2016;7:55–79. DOI: https://doi.org/10.1002/jrsm.1164
Makrakis D, Holt SK, Bernick C, Grivas P, Gore JL, Wright JL. Intravesical BCG and Incidence of Alzheimer Disease in Patients With Bladder Cancer: Results From an Administrative Dataset. Alzheimer Dis Assoc Disord. 2022;36:307–311. DOI: https://doi.org/10.1097/wad.0000000000000530
Kim JI, Zhu D, Barry E, et al. Intravesical Bacillus Calmette-Guérin Treatment Is Inversely Associated With the Risk of Develo** Alzheimer Disease or Other Dementia Among Patients With Non-muscle-invasive Bladder Cancer. Clin Genitourin Cancer. 2021;19:e409–e416. DOI: https://doi.org/10.1016/j.clgc.2021.05.001
Klinger D, Hill BL, Barda N, et al. Bladder Cancer Immunotherapy by BCG Is Associated with a Significantly Reduced Risk of Alzheimer’s Disease and Parkinson’s Disease. Vaccines (Basel). 2021;9:491. DOI: https://doi.org/10.3390/vaccines9050491
Tijms BM, Vromen EM, Mjaavatten O, et al. Cerebrospinal fluid proteomics in patients with Alzheimer’s disease reveals five molecular subtypes with distinct genetic risk profiles. Nat Aging. Epub 2024 Jan 9. DOI: https://doi.org/10.1038/s43587-023-00550-7
Bettcher BM, Tansey MG, Dorothée G, Heneka MT. Peripheral and central immune system crosstalk in Alzheimer disease - a research prospectus. Nat Rev Neurol. 2021;17:689–701. DOI: https://doi.org/10.1038/s41582-021-00549-x
Taniguchi K, Koga S, Nishikido M, et al. Systemic immune response after intravesical instillation of bacille Calmette-Guérin (BCG) for superficial bladder cancer. Clin Exp Immunol. 1999;115:131–135. DOI: https://doi.org/10.1046/j.1365-2249.1999.00756.x
Zuo Z, Qi F, Yang J, et al. Immunization with Bacillus Calmette-Guérin (BCG) alleviates neuroinflammation and cognitive deficits in APP/PS1 mice via the recruitment of inflammation-resolving monocytes to the brain. Neurobiol Dis. 2017;101:27–39. DOI: https://doi.org/10.1016/j.nbd.2017.02.001
Li J, Zhan L, Qin C. The double-sided effects of Mycobacterium Bovis bacillus Calmette-Guérin vaccine. NPJ Vaccines. 2021;6:14. DOI: https://doi.org/10.1038/s41541-020-00278-0
Li Y, Liu X, Wang W, et al. Low-dose IL-2 expands CD4+ regulatory T cells with a suppressive function in vitro via the STAT5-dependent pathway in patients with chronic kidney diseases. Ren Fail. 2018;40:280–288. DOI:10.1080/0886022X.2018.1456462
Baek H, Ye M, Kang G-H, et al. Neuroprotective effects of CD4+CD25+Foxp3+ regulatory T cells in a 3xTg-AD Alzheimer’s disease model. Oncotarget. 2016;7:69347–69357. DOI: https://doi.org/10.18632/oncotarget.12469
Keefe RC, Takahashi H, Tran L, et al. BCG therapy is associated with long-term, durable induction of Treg signature genes by epigenetic modulation. Sci Rep. 2021;11:14933. DOI: https://doi.org/10.1038/s41598-021-94529-2
Dow CT, Greenblatt CL, Chan ED, Dow JF. Evaluation of BCG Vaccination and Plasma Amyloid: A Prospective, Pilot Study with Implications for Alzheimer’s Disease. Microorganisms. 2022;10:424. DOI: https://doi.org/10.3390/microorganisms10020424
Zuo Z, Qi F, **ng Z, et al. Bacille Calmette-Guérin attenuates vascular amyloid pathology and maximizes synaptic preservation in APP/PS1 mice following active amyloid-β immunotherapy. Neurobiol Aging. 2021;101:94–108. DOI: https://doi.org/10.1016/j.neurobiolaging.2021.01.001
Li Q, Wang X, Wang ZH, et al. Changes in dendritic complexity and spine morphology following BCG immunization in APP/PS1 mice. Hum Vaccin Immunother. 2022;18:2121568. DOI: https://doi.org/10.1080/21645515.2022.2121568
Harrer, M., Cuijpers, P., Furukawa, T.A., & Ebert, D.D. (2021). Doing Meta-Analysis with R: A Hands-On Guide. Boca Raton, FL and London: Chapmann & Hall/CRC Press. ISBN 978-0-367-61007-4.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest: On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
How to cite this article: M. Ibrahim, P. Kim, R. Marawar et al. Bacillus Calmette-Guerin (BCG) Vaccine Impact on Dementia Risk in Bladder Cancer Patients: A Systematic Review and Meta-analysis. J Prev Alz Dis 2024; https://doi.org/10.14283/jpad.2024.94
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Ibrahim, M., Kim, P., Marawar, R. et al. Bacillus Calmette-Guerin (BCG) Vaccine Impact on Dementia Risk in Bladder Cancer Patients: A Systematic Review and Meta-Analysis. J Prev Alzheimers Dis (2024). https://doi.org/10.14283/jpad.2024.94
Received:
Accepted:
Published:
DOI: https://doi.org/10.14283/jpad.2024.94