Summary
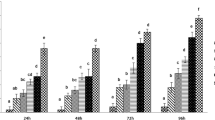
The toxicity of the mycotoxins nivalenol (NIV), deoxynivalenol (DON), and fumonisin B1 (FB1) was studied in the lepidopteran Spodoptera frugiperda (SF-9) cells, by the trypan blue dye-exclusion and 3-(4,5-dimethylthiozole-2-yl)-2,5-biphenyl tetrazolium bromide (MTT) tests, uptake analyses of cytotoxicity, and cell metabolism, respectively. Deoxytribonucleic acid analysis by flow cytometry was used to identify apoptosis and cell cycle distribution. After 48 h of exposure, the MTT and trypan blue dye-exclusion tests indicated that NIV was significantly more toxic than DON, and both were significantly more toxic than FB1. The IC50 (mycotoxin concentration resulting in 50% inhibition of proliferation) values for NIV and DON were 4.5 and 41 μM, and the CC50 (mycotoxin concentration that caused 50% cytotoxicity) values were 9.5 and 45 μM, respectively. At the highest concentration of FB1 (100 μM), there was 80% viability. With the same incubation time, cell cycles distribution showed an arrest of cells in the G0/G1 phase in the presence of NIV (up to 0.3 μM), DON (up to 3 μM), and FB1 (up to 10 μM). Morphological evidence of apoptosis was related to the toxicity of the substances in that the more toxic NIV induced late apoptosis, whereas DON and FB1 produced less-severe morphological changes characteristic of early apoptosis. This study suggests that NIV is more toxic than DON, which in turn is more toxic than FB1. These mycotoxins can modify the normal progression of the cell cycle and induce an apoptotic process.
Similar content being viewed by others
References
Abado-Bécogné, K., Toxines de Fusarium dans les cereals stockéés: evaluation de la toxicité globale sur un organisme sentinelle et mise au point d'un bioassay. Ph.D. thesis. Université de Bordeaux II, Bordeaux France; 1999.
Abramson, D. Mycotoxin formation and environmental factors. In: Bhatnagar, D.; Sinha, K. K., ed. Mycotoxins in agriculture and food safety. New York: Marcel Dekker, 1998:255–277.
Bouhet, S.; Hourcade, E.; Loiseau, N., et al. The mycotoxin fumonisin B1 alters the proliferation and the barrier function of porcine intestinal epithelial cells. Toxicol. Sci. 77:165–171; 2004.
Calò, I.; Fornelli, F.; Nenna, S.; Tursi, A.; Caiaffa, M. F.; Macchia, L. Beauvericin cytotoxicity to the invertebrate cell line SF-9. J. Appl. Genet. 44(4):515–520; 2003.
Ciacci-Zanella, J. R.; Jones, C. Fumonisin B1 a mycotoxin contaminant of cereal grains, and inducer of apoptosis via the tumour necrosis factor pathway and caspase activation. Food Chem. Toxicol. 37:703–712; 1999.
Ciacci-Zanella, J. R.; Merrill, A. H.; Wang, Jr E.; Jones, C. Characterization of cell-cycle arrest by fumonisin B1 in CV-1 cells. Food Chem. Toxicol. 36:791–804; 1998.
Darzynkiewicz, Z.; Traganos, F. Measurement of apoptosis. In: Scheper, Th. ed. Advances in biochemical engineering/biotechnology, vol. 62, BerlinL Springer-Verlag, 1998:33–73.
Dowd, P. F. Insect interaction with mycotoxin-producing fungi and their hosts. In: Bhatnagar, D.; Lillehoj, F. B.; Arora, D. K., ed. Mycotoxins in ecological system. Handbook of applied mycology, vol. 5. New York: Marcel Dekker; 1992:137–155.
Dumas, C.; Matha, V.; Quiot, J. M.; Vey, A. Effects of destruxins, cyclic depsipeptide mycotoxins, on calcium balance and phosphorylation of intracellular proteins in lepidopteran cell lines. Comp. Biochem. Physiol. C. Pharmacol. Toxicol. Endocrinol. 114:213–219; 1996.
Fertig, G.; Klop**er, M.; Miltenburger, H. G. Cell cycle kinetics of insect cell cultures compared to mammalian cell cultures. Exp. Cell Res. 189:208–212; 1990.
Gelderblom, W. C. A.; Jaskiewicz, K.; Marasas, W. F. O.; Thiel, P. G.; Horak, R. M.; Vleggaar, R.; Kriek, N. P. Fumonisins—novel mycotoxins with cancer promoting activity produced by Fusarium moniliforme. Appl. Environ. Microbiol. 54:1806–1811; 1998.
Gelderblom, W. C. A.; Cawood, S.; Snyman, S. D.; Vleggaar, R.; Marasas, W. F. O. Structure-activity relationship of fumonsins in short-term carcinogenesis and cytotoxicity assay. Food Chem. Toxicol. 31:407–414; 1993.
Holme, J. A.; Morrison, E.; Samuelsen, J. T.; Wiger, R.; Låg, M.; Schwarz, P. E.; Bernhoft, A.; Refnes, M. Mechanism involved in the induction of apoptosis by T-2 and HT-2 toxins in HL-60 human promyelocytic leukemia cells. Cell Biol. Toxicol. 19:53–68; 2003.
International Programe on Chemical Safety (IPCS), Fumonisin B1. Environmental Health Criteria 219. Geneva: World Health Organization; 2001.
Li, F. Q.; Luo, X. Y.; Yoshizawa, T. Mycotoxins (trichothecenes, zearalenone and functionsins) in cereals cereals associated with human red-mold intoxications stored since 1989 and 1991 in China. Nat. Toxins 7:93–97; 1999.
Liu, J. C.; Boucias, D. G.; Pendland, J. C.; Liu, W. Z.; Maruniak, J. The mode of action of hirsutellin A on eukaryotic cells. J. Invertebr. Pathol. 67:224–228; 1996.
Logrieco, A.; Moretti, A.; Fornelli, E., et al. Fusaproliferin production by Fusarium subglutinans and its toxicity to Artenia salina, SF-9 insect cells, and IARC/LCL 171 human B lymphocytes. Appl. Environ. Microbiol. 62:3378–3384; 1996.
Lòpez-Garcia, R.; Park, D. L. Effectiveness of postharvest procedures in management of mycotoxin hazards. In: Bhatagar, D.; Sinha, K. K., ed. Mycotoxins in agriculture and food safety. New York: Marcel Dekker; 1998:407–433.
Marasas, W. F. O.; Kellerman, T. S.; Gelderblom, W. C. A.; Coetzer, J. A. W.; Thiel, P. G.; Van Der Lugt, J. J. Leucoencephalomalacia in a horse induced by fumonisin B1 isolated from Fusarium moniliforme. Ondersterpoort J. Vet. Res. 55:197–203; 1988.
Minervini, F.; Fornelli, F.; Flynn, K. M. Toxicity and apoptosis induced by the mycotoxins nivalenol, deoxynivalenol and fumonisin B1 in a human erythroleukemia cell line. Toxicol. In Vitro 18:21–28; 2004.
Miura, K.; Aminova, L.; Murayama, Y. Fusarenon-X induced apoptosis in HL-60 cells depends on caspase activation and cytochrome c release. Toxicology 172:103–112; 2002.
Mobio, T. A.; Anane, R.; Baudrimont, I.; Carratù, M. R.; Shier, T. W.; Dano, S. D.; Ueno, Y.; Creppy, E. E. Epigenetic properties of fumonisin B1: cell cycle arrest and DNA base modification in C6 glioma cells. Toxicol. Appl. Pharmacol. 164:91–96; 2000a.
Mobio, T. A.; Baudrimont, I.; Sanni, A., et al. Prevention by vitamin E of DNa fragmentation and apoptosis induced by fumonisin B1 in C6 glioma cells. Arch. Toxicol. 74:112–119; 2000b.
Oh, G. S.; Hong, K. H.; Oh, H., et al. 4-Acetyl-12,13-epoxyl-9-trichothecene-3,15-diol isolated from the fruiting bodies of Isaria japonica Yasuda induces apoptosis of human leukemia cells (HL-60). Biol. Pharm. Bull. 24:785–789; 2001.
Panigrahi, S. Bioassay of mycotoxins using terrestrial and aquatic, animal and plant species. Food Chem. Toxicol. 31:767–790; 1993.
Pestka, J. J.; Yan, D.; King, L. E. Flow cytometric analysis of the effects of in vitro exposure to vomitoxin (deoxynivalenol) on apoptosis in murine T, B and IgA+ cells. Food Chem. Toxicol. 32:1125–1136; 1994.
Rheeder, J. P.; Marasas, W. F. O.; Thiel, P. G.; Sydenham, E. W.; Shephard, G. S.; van Schalkwyk, D. J. Fusarium moniliforme and fumonisins in corn in relation to human esophageal cancer in Transkei. Phytopathology 82:353–357; 1992.
Richard, J. L.; Thurston, J. R. Diagnosis of mycotoxicoses. Dordrecht: Kluwer; 1986.
Rotter, B. A.; Prelusky, D. B.; Pestka, J. J. Toxicology of deoxynivalenol (vomitoxin). J. Toxicol. Environ. Health 48:1–34; 1996.
Schmelz, E. M.; Dombrink-Kurtzman, M. A.; Roberts, P. C.; Kozutsumi, Y.; Kawasaki, T.; Merrill, A. H. Induction of apoptosis by fumonisin B1 in HT29 cells is mediated by the accumulation of endogenous free sphingoid bases. Toxicol. Appl. Pharmacol. 148:252–260; 1998.
Shifrin, V. I.; Anderson, P. Trichothecene mycotoxins trigger a ribotoxic stress response that activates c-Jun N-terminal kinase and p38 mitogen-activated protein kinase and induces apoptosis. J. Biol. Chem. 274:13985–13992; 1999.
Tiemann, U.; Viergutz, T.; Jonas, L.; Schneider, F. Influence of the mycotoxins α- and β-zearalenol and deoxynivalenol on the cell cycle of cultured porcine endometrial cells. Reprod. Toxicol. 17:209–218; 2003.
Tolenson, W. H.; Couch, L. H.; Melchior, W. B., et al. Fumonisin B1 induced apoptosis in cultured keratinocytes through sphingamine accumulation and ceramide depletion. Int. J. Oncol. 14:833–843; 1999.
Tollenson, W. H.; Melchior, W. B.; Morris, S. M.; McGarrity, L. J.; Domon, O. E.; Muskhelishvili, L.; James, S. J.; Howard, P. C. Apoptotic and anti-proliferative effects of fumonisin B1 in human keratinocytes, fibroblasts, esophageal epithelial cells and hepatoma cells. Carcinogenesis 17:239–249; 1996.
Vey, A.; Hoagland, R. E.; But, T. M. Toxic metabolites of fungal biocontrol agents. In: Butt, T. M.; Jackson, C. W.; Magan, N., ed. Fungi as biocontrol agents: progress, problems and potential. Wallingford: CABI Publishing; 2001:311–346.
Visconti, A.; Minervini, F.; Lucifero, G.; Gambatesa, V. Cytotoxic and immunotoxic effects of Fusarium mycotoxins using a rapid colorimetric bioassay. Mycopathologia 113:181–186; 1991.
WHO/FAO. Safety evaluation of certain mycotoxins in food. WHO Food Additives Series 47/FAO Food and Nutrition Paper 74. Geneva: World Health Organization; 2001:170–555.
Yang, G. H.; Jarvis, B. B.; Chung, Y. J.; Pestka, J. J. Apoptosis induction by the satratoxin and other trichothecene mycotoxins: relationship to ERK, p38 MARK and SAPK/JNK activation. Toxicol. Appl. Pharmacol. 164:149–160; 2000.
Zhang, H.; Nagashima, H.; Goto, T. Natural occurrence of mycotoxins in corn samples from high and low risk areas for human esophageal cancer in China. Mycotoxins 44:29–35; 1997.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fornelli, F., Minervini, F. & Mulè, G. Cytotoxicity induced by nivalenol, deoxynivalenol, and fumonisin B1 in the SF-9 insect cell line. In Vitro Cell.Dev.Biol.-Animal 40, 166–171 (2004). https://doi.org/10.1290/1543-706X(2004)40<166:CIBNDA>2.0.CO;2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1543-706X(2004)40<166:CIBNDA>2.0.CO;2