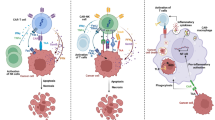
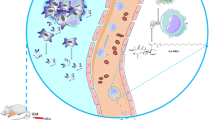
Abstract
Immunotherapy is a novel therapeutic approach for controlling and killing tumor cells by stimulating or reconstituting the immune system, among which T cells serve as immune targets. Herein, we used coenzyme Q10 (CoQ10), which has both immune activation and avoids adverse reactions, as a model drug and developed four CoQ10 submicron emulsions modified with sialic acid (SA) and/or monosialotetrahexosyl ganglioside (GM1). On the one hand, SA interacts with L-selectins on the surface of T cells after entering the circulatory system, leading to activation of T cells and enhancement of antitumor immune responses. On the other hand, owing to its immune camouflage, GM1 can prolong the circulation time of the preparation in the body, thereby increasing the accumulation of the drug at the tumor site. In vitro and in vivo experiments showed that SA-modified preparations exhibited stronger immune activation and inhibition of tumor proliferation. Pharmacokinetic experiments showed that GM1-modified preparations have longer circulation times in vivo. However, SA and GM1 co-modification did not produce a synergistic effect on the preparation. In conclusion, the SA-modified CoQ10 submicron emulsion (Q10-SE) showed optimal antitumor efficacy when administered at a medium dose (6 mg CoQ10 kg−1).
Graphical Abstract
In this study, the submicron emulsion model was used as a carrier, and the tumor-bearing mice were used as animal models. In addition, CoQ10 submicron emulsion was modified with SA-CH with active targeting function and/or GM1 with long-circulation function to explore the antitumor effects of different doses of CoQ10 submicron emulsion, and to screen the best tumor immunotherapy formulations of CoQ10.








Similar content being viewed by others
Abbreviations
- Q10-E:
-
CoQ10 submicron emulsion
- Q10-SE:
-
SA-CH modified CoQ10 submicron emulsion
- Q10-GE:
-
GM1 modified CoQ10 submicron emulsion
- Q10-SGE:
-
SA-CH and GM1 co-modified CoQ10 submicron emulsion
- DiR-E:
-
DiR submicron emulsion
- DiR-SE:
-
SA-CH modified DiR submicron emulsion
- DiR-GE:
-
GM1 modified DiR submicron emulsion
- DiR-SGE:
-
SA-CH and GM1 co-modified DiR submicron emulsion
References
Mattiuzzi C, Lippi G. Current cancer epidemiology. JJoe health G. 2019;9(4):217.
Fu L, ** W, Zhang J, Zhu L, Lu J, Zhen Y, et al. Repurposing non-oncology small-molecule drugs to improve cancer therapy: Current situation and future directions. Acta Pharm Sin B. 2022;12(2):532–57.
Bidram E, Esmaeili Y, Ranji-Burachaloo H, Al-Zaubai N, Zarrabi A, Stewart A, et al. A concise review on cancer treatment methods and delivery systems. J Drug Deliv Sci. 2019;54. https://doi.org/10.1016/j.jddst.2019.101350
Siva S, MacManus MP, Martin RF, Martin OA. Abscopal effects of radiation therapy: a clinical review for the radiobiologist. JCl 2015;356(1):82-90.
Dai WBY, Wang X, Song G, Liu TZ, He B, Zhang H, et al. Combination antitumor therapy with targeted dualnanomedicines. Adv Drug Deliver Rev. 2017;115:23–45.
Chen DS, Mellman I. Oncology meets immunology: the cancer-immunity cycle. Immunity. 2013;39(1):1–10.
Crane FL. Isolation of a quinone from beef heart mitochondria. JBBA 1957;25:220–1.
Cirilli I, Damiani E, Dludla PV, Hargreaves I, Marcheggiani F, Millichap LE, et al. Role of coenzyme Q10 in health and disease: an update on the last 10 years (2010–2020). Antioxidants (Basel). 2021;10(8):1325. https://doi.org/10.3390/antiox10081325
Fotino AD, Thompson-Paul AM, Bazzano LA. Effect of coenzyme Q10 supplementation on heart failure: a meta-analysis. JTAjocn 2013;97(2):268–75.
Mills EL, Kelly B, O'Neill LA. Mitochondria are the powerhouses of immunity. JNi 2017;18(5):488-98.
Lee SK, Lee JO, Kim JH, Kim N, You GY, Moon JW, et al. Coenzyme Q10 increases the fatty acid oxidation through AMPK-mediated PPARalpha induction in 3T3-L1 preadipocytes. Cell Signal. 2012;24(12):2329–36.
Weinberg SE, Sena LA, Chandel NS. Mitochondria in the regulation of innate and adaptive immunity. JI 2015;42(3):406-17.
Ron-Harel N, Santos D, Ghergurovich JM, Sage PT, Reddy A, Lovitch SB, et al. Mitochondrial biogenesis and proteome remodeling promote one-carbon metabolism for T cell activation. Cell Metab. 2016;24(1):104–17.
Fouad AA, Al-Mulhim AS, Jresat I. Therapeutic effect of coenzyme Q10 against experimentally-induced hepatocellular carcinoma in rats. Environ Toxicol Pharmacol. 2013;35(1):100–8.
Liu H-T, Huang Y-C, Cheng S-B, Huang Y-T, Lin P-T. Effects of coenzyme Q10 supplementation on antioxidant capacity and inflammation in hepatocellular carcinoma patients after surgery: a randomized, placebo-controlled trial. JNj 2015;15(1):1–9.
Garg S, Dhavala S, Krumova K, Kiebish M, Vishnudas V, Gesta S, et al. Membrane fluidity in cancer cell membranes as a therapeutic target: validation using bpm 31510. Biophys J. 2015;108(2):246a-a.
Chawla SP, Hendifar A, Chua VS, Quon D, Narasimhan V, Lavinski Y, et al. Phase 1 study of bpm 31510 (ubidecaranone) in advanced solid tumors: updated analysis of a novel treatment with promising activity. Ann Oncol. 2012;23:166.
Sun J, Patel CB, Jang T, Merchant M, Chen C, Kazerounian S, et al. High levels of ubidecarenone (oxidized CoQ10) delivered using a drug-lipid conjugate nanodispersion (BPM31510) differentially affect redox status and growth in malignant glioma versus non-tumor cells. Sci Rep. 2020;10(1):13899.
Varki A. Sialic acids in human health and disease. Trends Mol Med. 2008;14(8):351–60.
Kelm S, Schauer RJIroc. Sialic acids in molecular and cellular interactions. 1997;175:137-240.
Lehmann F, Tiralongo E, Tiralongo JJC, CMLS MLS. Sialic acid-specific lectins: occurrence, specificity and function. 2006;63(12):1331-54.
Bi S, Baum LGJBeBA-GS. Sialic acids in T cell development and function. 2009;1790(12):1599-610.
Ehrhardt C, Kneuer C, Bakowsky UJAddr. Selectins—an emerging target for drug delivery. 2004;56(4):527–49.
Meijer SL, Dols A, Hu HM, Chu Y, Romero P, Urba WJ, et al. Reduced L-selectin (CD62LLow) expression identifies tumor-specific type 1 T cells from lymph nodes draining an autologous tumor cell vaccine. Cell Immunol. 2004;227(2):93–102.
Zhou S, Zhang T, Peng B, Luo X, Liu X, Hu L, et al. Targeted delivery of epirubicin to tumor-associated macrophages by sialic acid-cholesterol conjugate modified liposomes with improved antitumor activity. 2017;523(1):203-16.
DeNinno MP. The synthesis and glycosidation of N-acetylneuraminic acid. JS 1991;1991(08):583-93.
** A-p, Xu Z-X, Liu F-L, Xu Y-L. Neuroprotective effects of monosialotetrahexosylganglioside. JNRR 2015;10(8):1343.
Gabizon A, Papahadjopoulos D. Liposome formulations with prolonged circulation time in blood and enhanced uptake by tumors. JPotnaos 1988;85(18):6949-53.
Park KJJoCR. Attenuating the immunogenicity of PEGylated liposomes by gangliosides. 2017;100(250):116.
Zhang T, Zhou SL, Kang L, Luo X, Liu Y, Song YZ, et al. The effect of monosialylganglioside mix modifying the PEGylated liposomal epirubicin on the accelerated blood clearance phenomenon. Asian J Pharm Sci. 2017;12(2):134–42.
Wang Y, Wang C, Deng Y, Song YJEJoPS. A new application of monosialotetrahexosylganglioside in pharmaceutics: preparation of freeze-thaw-resistant coenzyme Q10 emulsions. 2021;159:105701.
Zheng JS, Zheng SY, Zhang YB, Yu B, Zheng WJ, Yang F, et al. Sialic acid surface decoration enhances cellular uptake and apoptosis-inducing activity of selenium nanoparticles. Colloid Surface B. 2011;83(1):183–7.
Müller R, Schmidt S, Buttle I, Akkar A, Schmitt J, Brömer SJIjop. SolEmuls®—novel technology for the formulation of iv emulsions with poorly soluble drugs. 2004;269(2):293–302.
Wang Y, Wang C, Deng Y, Song Y. A new application of monosialotetrahexosylganglioside in pharmaceutics: preparation of freeze-thaw-resistant coenzyme Q10 emulsions. Eur J Pharm Sci. 2021;159: 105701.
Dorfmüller Th. Dynamic light scattering — applications of photon correlation spectroscopy. Berichte der Bunsengesellschaft für physikalische Chemie. 1985;91:498–9.
Sui DZ, Meng XM, Li CZ, Tang XY, Qin Y, Zhang N, et al. The fate of sialic acid and peg modified epirubicin liposomes in aged versus young cells and tumor mice models. Pharmaceutics. 2022;14(3):545. https://doi.org/10.3390/pharmaceutics14030545
Cheon IS, Park SM, Lee HJ, Hong JE, Ji SY, Shim BS, et al. Functional characteristics of porcine peripheral T cells stimulated with IL-2 or IL-2 and PMA. Res Vet Sci. 2014;96(1):54–61.
Ye YX, Zhang YX, Lu XY, Huang XY, Zeng XF, Lai XQ, et al. The anti-inflammatory effect of the SOCC blocker SK&F 96365 on mouse lymphocytes after stimulation by Con A or PMA/ionomycin. Immunobiology. 2011;216(9):1044–53.
Ding JQ, Zhao D, Hu YW, Liu MQ, Liao XR, Zhao BW, et al. Terminating the renewal of tumor-associated macrophages: a sialic acid-based targeted delivery strategy for cancer immunotherapy. Int J Pharmaceut. 2019;571:118706. https://doi.org/10.1016/j.ijpharm.2019.118706
Shi J, Zhou S, Kang L, Ling H, Chen J, Duan L, et al. Evaluation of the antitumor effects of vitamin K2 (menaquinone-7) nanoemulsions modified with sialic acid-cholesterol conjugate. Drug Deliv Transl Res. 2018;8(1):1–11.
Qiu Q, Lu M, Li C, Luo X, Liu X, Hu L, et al. Novel self-assembled ibrutinib-phospholipid complex for potently peroral delivery of poorly soluble drugs with pH-dependent solubility. AAPS PharmSciTech. 2018;19(8):3571–83.
Luo X, Hu L, Zheng H, et al. Neutrophil-mediated delivery of pixantrone-loaded liposomes decorated with poly(sialic acid)-octadecylamine conjugate for lung cancer treatment. Drug Deliv. 2018;25:1200–12.
Chen M, Wu WJ, Wang S, Lai XX, Liu MY, Sun YM, et al. Neutrophils as emerging immunotherapeutic targets: indirect treatment of tumors by regulating the tumor immune environment based on a sialic acid derivative-modified nanocomplex platform. Int J Pharmaceut. 2022;620.
Ding J, Sui D, Liu M, et al. Sialic acid conjugate-modified liposomes enable tumor homing of epirubicin via neutrophil/monocyte infiltration for tumor therapy. Acta Biomaterialia. 2021;134:702–15.
Drummond DC, Meyer O, Hong K, et al. Optimizing liposomes for delivery of chemotherapeutic agents to solid tumors. Pharmacol Rev. 1999;51:691–743.
Ziegler SF, Ramsdell F, Hjerrild KA, et al. Molecular characterization of the early activation antigen CD69: a type II membrane glycoprotein related to a family of natural killer cell activation antigens. Eur J Immunol. 1993;23:1643–8.
Kimura MY, Hayashizaki K, Tokoyoda K, Takamura S, Motohashi S, Nakayama T. Crucial role for CD 69 in allergic inflammatory responses: CD 69‐Myl9 system in the pathogenesis of airway inflammation. JIr 2017;278(1):87–100.
Hayashizaki K, Kimura MY, Tokoyoda K, Hosokawa H, Shinoda K, Hirahara K, et al. Myosin light chains 9 and 12 are functional ligands for CD69 that regulate airway inflammation. 2016;1(3):eaaf9154-eaaf.
Witherden DA, Abernethy NJ, Kimpton WG, Cahill RNJEjoi. Changes in thymic export of L‐selectin+ γδ and αβ T cells during fetal and postnatal development. 1994;24(5):1234–9.
Tedder TF, Steeber DA, Chen A, Engel PJTFJ. The selecting: vascular adhesion molecules. 1995;9(10):866–73.
Gajbhiye V, Ganesh N, Barve J, Jain NKJEJoPS. Synthesis, characterization and targeting potential of zidovudine loaded sialic acid conjugated-mannosylated poly (propyleneimine) dendrimers. 2013;48(4–5):668–79.
Chen M, Wu W, Wang S, Lai X, Liu M, Sun Y, et al. Neutrophils as emerging immunotherapeutic targets: indirect treatment of tumors by regulating the tumor immune environment based on a sialic acid derivative-modified nanocomplex platform. 2022;620:121684.
Qiu Q, Li C, Song Y, Shi T, Luo X, Zhang H, et al. Targeted delivery of ibrutinib to tumor-associated macrophages by sialic acid-stearic acid conjugate modified nanocomplexes for cancer immunotherapy. 2019;92:184-95.
Chiu GN, Bally MB, Mayer LDJBeBA-B. Selective protein interactions with phosphatidylserine containing liposomes alter the steric stabilization properties of poly (ethylene glycol). 2001;1510(1-2):56-69.
Fischer HC, Hauck TS, Gómez-Aristizábal A, Chan WCJAM. Exploring primary liver macrophages for studying quantum dot interactions with biological systems. 2010;22(23):2520-4.
Khlebtsov N, Dykman L. Biodistribution and toxicity of engineered gold nanoparticles: a review of in vitro and in vivo studies. JCSR 2011;40(3):1647-71.
**ao J-H, Zhang Y, Liang G-Y, Liu R-M, Li X-G, Zhang L-T, et al. Synergistic antitumor efficacy of antibacterial helvolic acid from Cordyceps taii and cyclophosphamide in a tumor mouse model. 2017;242(2):214-22.
Damia G, D’Incalci MJEJoC. Contemporary pre-clinical development of anticancer agents–what are the optimal preclinical models? 2009;45(16):2768–81.
Liu M, Luo X, Qiu Q, Kang L, Li T, Ding J, et al. Redox-and pH-sensitive glycan (polysialic acid) derivatives and F127 mixed micelles for tumor-targeted drug delivery. 2018;15(12):5534–45.
Byrne JD, Betancourt T, Brannon-Peppas LJAddr. Active targeting schemes for nanoparticle systems in cancer therapeutics. 2008;60(15):1615-26.
Raker VK, Becker C, Landfester K, Steinbrink KJC. Targeted activation of T cells with IL-2-coupled nanoparticles. 2020;9(9):2063.
Donia M, Hansen M, Sendrup SL, Iversen TZ, Ellebæk E, Andersen MH, et al. Methods to improve adoptive T-cell therapy for melanoma: IFN-γ enhances anticancer responses of cell products for infusion. 2013;133(2):545-52.
Levy M, Schutze W, Fuhrer C, Benita SJJom. Characterization of diazepam submicron emulsion interface: role of oleic acid. 1994;11(1):79-92.
Chen JX, Chen SQ, Yang XB, Wang SM, Wu WY. Efficacy and safety of Brucea javanica oil emulsion injection as adjuvant therapy for cancer: an overview of systematic reviews and meta-analyses. Phytomedicine. 2022;102.
Ting YW, Chiou YS, Pan MH, Ho CT, Huang QR. In vitro and in vivo anti-cancer activity of tangeretin against colorectal cancer was enhanced by emulsion-based delivery system. J Funct Foods. 2015;15:264–73.
Mohammadzadeh V, Rahiman N, Hosseinikhah SM, Barani M, Rahdar A, Jaafari MR, et al. Novel EPR-enhanced strategies for targeted drug delivery in pancreatic cancer: an update. Journal of Drug Delivery Science and Technology. 2022;73.
Kalyane D, Raval N, Maheshwari R, Tambe V, Kalia K, Tekade RK. Employment of enhanced permeability and retention effect (EPR): nanoparticle-based precision tools for targeting of therapeutic and diagnostic agent in cancer. Mater Sci Eng C Mater Biol Appl. 2019;98:1252–76.
Varki A, Gagneux PJAotNYAoS. Multifarious roles of sialic acids in immunity. 2012;1253(1).
Ajit, Glycobiology VJ. Since there are PAMPs and DAMPs, there must be SAMPs? Glycan “self-associated molecular patterns” dampen innate immunity, but pathogens can mimic them. 2011.
Zhang KM, Zhao HR. Perspectives in the stability of emulsion explosive. Adv Colloid Interface Sci. 2022;307: 102745.
Ouyang J, Meng Y. Quantitative effect of droplet size and emulsion viscosity on the storage stability of asphalt emulsion. Constr Build Mater. 2022;342.
Funding
This study was supported by the Career Development Support Plan for Young and Middle-aged Teachers at the Shenyang Pharmaceutical University (ZDN2021009).
Author information
Authors and Affiliations
Contributions
Conceptualization: **anmin Meng, Na Yan, Yihui Deng, and Yanzhi Song; data curation: **anmin Meng, Na Yan, and Yanzhi Song; acquisition: Na Yan, Tiantian Guo, Mingqi Wang, and Kaituo Zhang; writing: **anmin Meng; analysis: Dezhi Sui and Meng Chen; supervision: **nrong Liu. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Meng, X., Yan, N., Guo, T. et al. Antitumor Immunotherapy of Sialic Acid and/or GM1 Modified Coenzyme Q10 Submicron Emulsion. AAPS PharmSciTech 23, 283 (2022). https://doi.org/10.1208/s12249-022-02426-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-022-02426-2