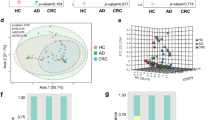
Abstract
Background
Colorectal adenomatous polyps (CAPs) are considered precancerous lesions of colorectal cancer (CRC). The gut microbiota participates in the process of digestion and, in the process, produces metabolites, mainly short-chain fatty acids (SCFAs), secondary bile acids and conjugated linoleic acid (CLA). This study aimed to investigate the gut microbiota constituents and metabolites in the faeces of CAP patients to identify microbiota or metabolites that can be used as sensitive biological predictors and to provide a theoretical basis for the clinical treatment of CAPs.
Methods
16S rRNA sequence analysis was used to detect microbial changes in the faeces of CAP patients. qPCR analysis was used to evaluate the ability of the microbiota to produce metabolites, and the contents of metabolites in faeces were detected by ion chromatography and ultra-performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS).
Results
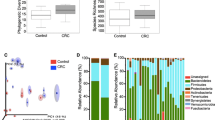
Based on the detection of the gut microbiota, patients with CAPs had increased abundances of Bacteroides and Citrobacter, and the abundances of Weissella and Lactobacillus were decreased. We also explored gene expression, and the abundance of butyrate-producing bacterial genes was significantly increased in the faeces of CAP patients, but those of secondary bile acid-producing and CLA-producing bacterial genes showed no differences in faecal samples. The acetic acid and butyric acid contents were increased in the faeces of the CAP group, and the healthy control group had higher t10,c12-CLA contents.
Conclusion
The gut microbiota analysis results, assessed in faeces, showed that Bacteroides and Citrobacter were positively correlated with CAPs, which indicated that changes in specific genera might be detrimental to intestinal health. In addition, t10,c12-CLA played an important role in protecting the intestine.
Similar content being viewed by others
Background
Colorectal cancer (CRC) is a common malignant tumour of the digestive tract, with the third highest incidence and second highest mortality among tumours worldwide [1, 2]. Colorectal adenomatous polyps (CAPs) are regarded as a critical precursor to CRC [3], and adenoma is an early neoplastic tissue that has not gained the properties of a cancer.
There are 100 trillion bacteria in the human intestine, and the collective genome of these bacteria is called the gut microbiome, which is 150 times the size of the human genome [4]. The gut microbiota is essential for the growth and physical health of the human body. The gut microbiota participates in digestion and absorption of food in the intestines and actively participates in cell-mediated immune responses, which maintain intestinal barrier function and intestinal environment stability. The imbalance of this symbiotic relationship might have an adverse effect on the host, and gut microbiota imbalance has been observed in cases of inflammatory bowel disease (IBD) [5], obesity [6], ageing [7] and cancer [8].
Moore et al. [9] applied culture methods to analyse the faeces of CRC patients and colorectal polyp patients and found that the abundances of Bacteroides and Bifidobacteria were positively correlated with the risk of colon polyps, while those of Lactobacilli and Eubacteria were related to the intestinal tract and had a protective effect. However, the types of bacteria that could be cultured in faeces were limited, and most of the bacteria could not be cultured in an in vitro environment. Therefore, the emergence of high-throughput sequencing technology and metagenomic analysis provided a better solution for analysing complex microbiome data. Fusobacterium has been identified as a risk factor for both colorectal adenomas and cancer [10], and the mechanism of the F. nucleatum association with CRC has been clarified in mice [11].
Intestinal metabolites, such as short-chain fatty acids (SCFAs) and bile acids, are strongly linked with cancerous conditions in the gut [12]. Conjugated linoleic acid (CLA) is considered a health-promoting fatty acid, and the anti-cancer properties of CAL in vivo and in vitro have been widely recognized [13]. Some Firmicutes use the butyryl-CoA:acetate CoA-transferase route to produce butyrate, and the proportion of propionate presented in faeces correlated with the relative abundance of Bacteroidetes [12]. Owning to their generation of reactive oxygen species (ROS) and reactive nitrogen species (RNS), which cause DNA damage, bile acids have been implicated in carcinogenesis.
To investigate the gut microbiota profile in CAP patients, we collected faeces from CAP patients attending the First Affiliated Hospital of Kunming Medical University. High-throughput sequencing technology was used to analyse the gut microbiota in the intestinal tract, which extended the understanding of the microbial community. Analysis of the faecal metabolites of CAP patients was performed to identify the metabolites that changed and to explore the changes in the gut microbiota and its metabolites during the progression of CAP to CRC. This study will provide comprehensive information about the gut microbiota and metabolite changes in CAP patients, which will help to characterize the role of gut microbiota and metabolites in adenoma occurrence and progression, and the differences in the gut microbiota and metabolites might be considered biomarkers of CAP in the future.
Methods
Sampling
Thirty patients with colorectal adenomatous polyps were selected from the First Affiliated Hospital of Kunming Medical University from November 2017 to April 2018. Exclusion criteria included obesity, special eating habits, colorectal cancer, history of colorectal surgery, colitis (ulcerative, Crohn’s), metabolic disease (diabetes, hyperlipidaemia), and infectious disease. Thirty healthy volunteers were selected from the First Affiliated Hospital of Kunming Medical University as controls. No subjects were taking antibiotics, immunosuppressive agents, corticosteroids or probiotics within 3 months prior to sample collection. There were no significant differences in age, gender, or BMI between the two groups (P > 0.05) (Table 1). Stool samples from volunteers were preserved after admission, and samples were collected in accordance with the relevant guidelines and regulations.
DNA extraction and 16S rRNA gene amplification
Genomic DNA was extracted from stool samples and biopsy samples with the QIAamp DNA Stool Mini Kit (Qiagen, Hilden, Germany). The primers for amplification of the V3-V4 region of the bacterial 16S rRNA gene, after amplification high throughput sequencing was performed with the Illumina MiSeq platform (Illumina, CA, USA).
Bioinformatics analysis
The high-quality paired-end reads were combined to tags based on overlaps, and the consensus sequence was generated by Fast Length Adjustment of Short reads (FLASH, v1.2.11). The tags were clustered into operational taxonomic units (OTUs) by scripts in USEARCH (v7.0.1090) software, OTUs were clustered with a 97% similarity cut-off using UPARSE, and OTU representative sequences were taxonomically classified using the Ribosomal Database Project (RDP) Classifier v.2.2 trained on the Greengene_2013_5_99 database with a 0.6 confidence value as the cut-off. OTUs were filtered by removing unassigned OTUs and removing OTUs not assigned to target species.
OTUs were used for α diversity estimation. Comparison of the β diversity, which is the difference in species diversity between two groups, was performed based on the OTU abundance by QIIME (V1.80).
Specimen annotation analysis is a method that compares OTUs to a database of classified OTUs at the phylum, class, order, family and species levels, and the analysis is then presented by histograms. UniFrac analysis used phylogenetic information to compare species community differences between samples.
Metabolite determination
Ion chromatograph analysis
The faecal samples stored in the refrigerator at − 80 °C were removed, weighed to 300 mg, dissolved in 1 mL of dH2O, vortexed and mixed for 30 s. Then, the supernatant was incubated with a 0.22 μm microporous nylon membrane (water system). The liquid was filtered, placed in an EP tube and placed in a refrigerator at − 20 °C for use. Standard curves were generated by using standard solutions. A volume of 25 μL was performed into chromatographic columns (DIONEX IonPac AG11-HC 4 × 50 mm & IonPac AS11-HC 4 × 250 mm, USA) and eluted with KOH at a flow rate of 1.2 mL/min; ions were detected by a conductivity detector in an ion chromatograph (Thermo Dionex ICS-3000, USA), and the column temperature was 30 °C.
UPLC-MS/MS analysis
The faecal samples stored in the refrigerator at − 80 °C were removed, weighed to 200 mg, dissolved in 1 mL of methanol, vortexed and mixed for 30 s. The supernatant was then incubated with a 0.22 μm microporous nylon membrane (organic system). The liquid was filtered, placed in an EP tube and placed in a refrigerator at − 20 °C for use. Standard curves were generated by using standard solutions.
UPLC conditions
The mobile phase consisted of 0.05% ammonia (5 mM aqueous solution) in water as solution A and acetonitrile as solution B. The flow rate of the mobile phase through the column was 0.4 mL/min (Waters BEH C18 1.7 μm, 50*2.1 mm, USA) at a temperature of 40 °C. The injection volume was 1 μL. Mass spectrometry conditions included electrospray ionization, negative ion mode, multiple reaction detection, air as the desolvation gas, nitrogen as the cone gas, and argon as the collision gas.
Real-time PCR analysis
To explore bacteria that produce specific metabolites, real-time PCR was used (TIANLONG Gentier 96, ** the gut microbiome of diet-induced obese mice. BMC Biol. 2017;15(1):120." href="/article/10.1186/s13099-020-00395-0#ref-CR40" id="ref-link-section-d91300678e1641">40, 41], and the Bacteroides abundance showed a significant increase in the CAP group. The differences in secondary bile acid-producing bacteria were not statistically significant in this study. Mullish et al. [42] found that the BaiCD operon was not present in all bacteria with 7α-dehydroxylating ability, which has been considered an important process for secondary bile acid formation in faeces [13].
Metabolomics provides a qualitative and quantitative method of metabolite in analysis that can complete analysis along with microbiology. Metabolites (small molecules < 1500 Da) are cellular metabolism intermediates or end products, that can be produced directly by the host organism or can be derived from various other external sources, such as the diet, microbes, or xenobiotic sources [43]. Biological systems display complex and analytical limitations, and it is not possible to identify all the metabolites present in a specimen. Studies on metabolites and diseases indicate changes in diabetes [44], cardiovascular disease and heart failure [45, 46], autism [47] and anxiety [48]. As research progresses, metagenomic markers can be utilized for early disease diagnosis or cancer screening, and gut microbiota biology can indicate the effectiveness of cancer therapies and has predictive potential [49].
Conclusion
In this study, gut microbiota analysis in the faeces showed that the abundances of Weissella and Lactobacillus were decreased, and those of Bacteroides and Citrobacter were increased in the CAP group. The increased abundances of Bacteroides and Citrobacter were positively correlated with CAPs, which indicated that changes in specific genera might be detrimental to intestinal health. In addition, metabolite detection showed changes in butyrate content, indicating that additional experiments are needed to investigate the function of butyrate in the intestinal environment. The increased concentration of t10,c12-CLA plays an important role in protecting the intestine. Analysis of the gut microbiota demonstrated carriage of operons producing metabolites, which indicated that additional functional operons might exist in the gut microbiota or intracellularly. Therefore, further studies focusing on lifestyle, diet and other factors in different populations are required to confirm the effect on intestinal health.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424.
Cresci GA, Emmy B. Gut microbiome: what we do and don’t know. Nutr Clin Pract. 2015;30(6):734–46.
Feng Q, Liang S, Jia H, Stadlmayr A, Tang L, Lan Z, et al. Gut microbiome development along the colorectal adenoma-carcinoma sequence. Nat Commun. 2015;6:6528.
Shreiner AB, Kao JY, Young VB. The gut microbiome in health and in disease. Curr Opon Gastroenterol. 2015;31(1):69–75.
Katsuyoshi M, Takanori K. The gut microbiota and inflammatory bowel disease. Semin Immunopathol. 2015;37(1):47–55.
Rosenbaum M, Knight R, Leibel RL. The gut microbiota in human energy homeostasis and obesity. Trends Endocrinol Metab. 2015;26(9):493.
Shen X, Miao J, Wan Q, Wang S, Li M, Pu F, et al. Possible correlation between gut microbiota and immunity among healthy middle-aged and elderly people in southwest China. Gut Pathog. 2018;10:4.
Tilg H, Adolph TE, Gerner RR, Moschen AR. The intestinal microbiota in colorectal cancer. Cancer Cell. 2018;33(6):954.
Moore WE, Moore LH. Intestinal floras of populations that have a high risk of colon cancer. Appl Environ Microbiol. 1995;61(9):3202–7.
Rajagopala SV, Vashee S, Oldfield LM, Suzuki Y, Venter JC, Telenti A, et al. The human microbiome and cancer. Cancer Prev Res. 2017;10(4):226–34.
Yachida S, Mizutani S, Shiroma H, Shiba S, Nakajima T, Sakamoto T, et al. Metagenomic and metabolomic analyses reveal distinct stage-specific phenotypes of the gut microbiota in colorectal cancer. Nat Med. 2019;25(6):968–76.
Louis P, Hold GL, Flint HJ. The gut microbiota, bacterial metabolites and colorectal cancer. Nat Rev Microbiol. 2014;12(10):661–72.
Degen C, Habermann N, Piegholdt S, Glei M, Jahreis G. Human colon cell culture models of different transformation stages to assess conjugated linoleic acid and conjugated linolenic acid metabolism: challenges and chances. Toxicol In Vitro. 2012;26(6):985–92.
Graf D, Di Cagno R, Fak F, Flint HJ, Nyman M, Saarela M, et al. Contribution of diet to the composition of the human gut microbiota. Microb Ecol Health Dis. 2015;26:26164.
Sun HD, Jiang ZY. Recent progress in gut microbiota and bile acid metabolism. Chin Bull Life Sci. 2016;28:1405–9.
Yadav M, Verma MK, Chauhan NS. A review of metabolic potential of human gut microbiome in human nutrition. Arch Microbiol. 2018;200(2):203–17.
Davenport JR, Su T, Zhao Z, Coleman HG, Smalley WE, Ness RM, et al. Modifiable lifestyle factors associated with risk of sessile serrated polyps, conventional adenomas and hyperplastic polyps. Gut. 2016;67(3):456.
Chen CY, Duan Y, Mao J, Niu M, Du Y. Analysis of gut microbiota change in colorectal adenomatous polyps patients by 16S rRNA sequencing. Chin J Lab Med. 2020. https://doi.org/10.3760/cma.j.issn.1009-9158.2020.02.014.
Thomas AM, Jesus EC, Lopes A, Aguiar A Jr, Begnami MD, Rocha RM, et al. Tissue-associated bacterial alterations in rectal carcinoma patients revealed by 16S rRNA community profiling. Front Cell Infect Microbiol. 2016;6:179.
Flemer B, Lynch DB, Brown JM, Jeffery IB, Ryan FJ, Claesson MJ, et al. Tumour-associated and non-tumour-associated microbiota in colorectal cancer. Gut. 2017;66(4):633–43.
Gao Z, Guo B, Gao R, Zhu Q, Qin H. Microbiota disbiosis is associated with colorectal cancer. Front Microbiol. 2015;6:20.
Wu N, Yang X, Zhang R, Li J, **ao X, Hu Y, et al. Dysbiosis signature of fecal microbiota in colorectal cancer patients. Microb Ecol. 2013;66(2):462–70.
Yu LC, Wei SC, Ni YH. Impact of microbiota in colorectal carcinogenesis: lessons from experimental models. Intest Res. 2018;16(3):346–57.
Flanagan L, Schmid J, Ebert M, Soucek P, Kunicka T, Liska V, et al. Fusobacterium nucleatumassociates with stages of colorectal neoplasia development, colorectal cancer and disease outcome. Eur J Clin Microbiol Infect Dis. 2014;33(8):1381–90.
Marks P, Richon VM, Rifkind RA. Histone deacetylase inhibitors: inducers of differentiation or apoptosis of transformed cells. J Natl Cancer Inst. 2000;92(15):1210–6.
Bultman SJ. Interplay between diet, gut microbiota, epigenetic events, and colorectal cancer. Mol Nutr Food Res. 2017. https://doi.org/10.1002/mnfr.201500902.
Belcheva A, Irrazabal T, Robertson Susan J, Streutker C, Maughan H, Rubino S, et al. Gut microbial metabolism drives transformation of MSH2-deficient colon epithelial cells. Cell. 2014;158(2):288–99.
Ferrer-Picon E, Dotti I, Corraliza AM, Mayorgas A, Esteller M, Perales JC, et al. Intestinal inflammation modulates the epithelial response to butyrate in patients with inflammatory bowel disease. Inflamm Bowel Dis. 2020;26(1):43–55.
Chen H, Yu Y, Wang J, Lin Y, Kong X, Yang C, et al. Decreased dietary fiber intake and structural alteration of gut microbiota in patients with advanced colorectal adenoma. Am J Clin Nutr. 2013;97(5):1044–52.
Keku TO, Dulal S, Deveaux A, Jovov B, Han X. The gastrointestinal microbiota and colorectal cancer. Am J Physiol Gastrointest Liver Physiol. 2015;308(5):G351.
Reiser R. The biochemical conversions of conjugated dienoic and trienoic fatty acids. Arch Biochem Biophys. 1951;32(1):113–20.
Li K, Zhou N, Li HY, Wang L. Physiological function for conjugated linoleic acid. Food Res Dev. 2012;33:226–8.
Torre AD, Debiton E, Durand D, Chardigny JM, Berdeaux O, Loreau O, et al. Conjugated linoleic acid isomers and their conjugated derivatives inhibit growth of human cancer cell lines. Anticancer Res. 2005;25(6B):3943–9.
Tanmahasamut P, Liu J, Hendry LB, Sidell N. Conjugated linoleic acid blocks estrogen signaling in human breast cancer cells. J Nutr. 2004;134(3):674.
Cho HJ, Kim EJ, Lim SS, Kim MK, Sung M-K, Kim J-S, et al. Trans-10, cis-12, not cis-9, trans-11, conjugated linoleic acid inhibits G1-S progression in HT-29 human colon cancer cells. J Nutr. 2006;136(4):893–8.
Raimondi S, Amaretti A, Leonardi A, Quartieri A, Gozzoli C, Rossi M. Conjugated linoleic acid production by bifidobacteria: screening, kinetic, and composition. BioMed Res Int. 2016;2016(6):1–8.
Cao H, Xu M, Dong W, Deng B, Wang S, Zhang Y, et al. Secondary bile acid-induced dysbiosis promotes intestinal carcinogenesis. Int J Cancer. 2017;140(11):2545–56.
Tong JL, Ran ZH, Shen J, Fan GQ, **ao SD. Association between fecal bile acids and colorectal cancer: a meta-analysis of observational studies. Yonsei Med J. 2008;49(5):792.
Feng H-Y, Chen Y-C. Role of bile acids in carcinogenesis of pancreatic cancer: an old topic with new perspective. World J Gastroenterol. 2016;22(33):7463–77.
Zheng X, Huang F, Zhao A, Lei S, Zhang Y, ** the gut microbiome of diet-induced obese mice. BMC Biol. 2017;15(1):120.
Ruengsomwong S, La-Ongkham O, Jiang J, Wannissorn B, Nakayama J, Nitisinprasert S. Microbial community of healthy Thai vegetarians and non-vegetarians, their core gut microbiota, and pathogen risk. J Microbiol Biotechnol. 2016;26(10):1723–35.
Mullish BH, Pechlivanis A, Barker GF, Thursz MR, Marchesi JR, McDonald JAK. Functional microbiomics: evaluation of gut microbiota-bile acid metabolism interactions in health and disease. Methods. 2018;149:49–58.
Johnson CH, Ivanisevic J, Siuzdak G. Metabolomics: beyond biomarkers and towards mechanisms. Nat Rev Mol Cell Biol. 2016;17(7):451–9.
Brunkwall L, Orho-Melander M. The gut microbiome as a target for prevention and treatment of hyperglycaemia in type 2 diabetes: from current human evidence to future possibilities. Diabetologia. 2017;60(6):943–51.
Li DY, Tang WHW. Gut microbiota and atherosclerosis. Curr Atheroscler Rep. 2017;19(10):39.
Kitai T, Tang WHW. Gut microbiota in cardiovascular disease and heart failure. Clin Sci. 2018;132(1):85–91.
Hughes HK, Rose D, Ashwood P. The gut microbiota and dysbiosis in autism spectrum disorders. Curr Neurol Neurosci Rep. 2018;18(11):81.
Malan-Muller S, Valles-Colomer M, Raes J, Lowry CA, Seedat S, Hemmings SMJ. The gut microbiome and mental health: implications for anxiety- and trauma-related disorders. OMICS. 2018;22(2):90–107.
Wong SH, Kwong TNY, Wu CY, Yu J. Clinical applications of gut microbiota in cancer biology. Semin Cancer Biol. 2019;55:28–36.
Acknowledgements
We thank Dr. Yinglei Miao (the First Affiliated Hospital of Kunming Medical University) for technical advice, Dr. Yang Sun (the First Affiliated Hospital of Kunming Medical University) for assistance with sample collection and high-throughput sequencing analysis, and experiment guidance. The authors would like to thank Springer Nature Author Services (https://authorservices.springernature.com/) for the English language review. The authors express their gratitude to the anonymous reviewers for their valuable comments and suggestions.
Clarification
We have confirmed the text “Bacteroides, butyric acid and t10,c12-CLA Changes in Colorectal Adenomatous Polyps” (https://doi.org/10.21203/rs.3.rs-40596/v1) is our deposition as pre-print of this manuscript at Research Square (https://www.researchsquare.com/article/rs-40596/v1).
Funding
This work was supported by funding from the National Natural Science Foundation of China (NO. 81960382); Natural Science Foundation of Yunnan Province (NO. 2018FA043); Joint special fund for applied basic research in Yunnan Province (NO. 2017FE467 (-173)) and (NO. 2019FE001 (-060)); Training plan for medical decipline leaders of Yunnan health and family planning Commission (NO. D-2017023).
Author information
Authors and Affiliations
Contributions
CC, MN, JP and SL performed the data analysis and interpretation; JP, YD and YD participated in the discussion and interpretations of the results; CC, HL, QH, and JM drafted the manuscript; ND and YD designed the study, and YD revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This research was approved by the Ethics Committee of the First Affiliated Hospital of Kunming Medical University ((2017) L-15). All volunteers signed informed consent.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1 Figure S1.
The alpha-diversity comparison of faeces between two groups, A Chao, B Ace, C Shannon and D Simpson index were present microbial community abundance and diversity. NS: None significantly differences. Figure S2. PCoA analysis: PC1 coordinates represent the main coordinate component that caused the largest difference in the sample, and PC2 represents the second coordinate component. Figure S3. Heatmap of Weighted UniFrac analysis. On top, the cluster tree presented sample phylogenetic relationships, the change of diversity ratio along with colour from blue to red.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Chen, C., Niu, M., Pan, J. et al. Bacteroides, butyric acid and t10,c12-CLA changes in colorectal adenomatous polyp patients. Gut Pathog 13, 1 (2021). https://doi.org/10.1186/s13099-020-00395-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13099-020-00395-0