Abstract
Background
Previous studies have indicated that the deposition of abdominal adipose tissue was associated with the abnormalities of cardiometabolic components. The aim of this study was to examine the relationship of visceral adipose tissue (VAT), subcutaneous adipose tissue (SAT) and metabolic status and the different effects between males and females.
Methods
The 1388 eligible subjects were recruited in a baseline survey of metabolic syndrome in China, from two communities in Hangzhou and Chengdu. Areas of abdominal VAT and SAT were measured by magnetic resonance imaging (MRI). Serum total triglycerides (TG), high-density lipoprotein cholesterol (HDL-C) were measured by an automated biochemical analyzer. Metabolic abnormality (MA) was defined more than one abnormal metabolic components, which was based on the definition of metabolic syndrome (IDF 2005). Multiple logistic regression was used to calculate the odds ratios (ORs) and 95% confidence intervals (95%CI). Predictive value was assessed by area under the curve (AUC), net reclassification improvement (NRI), and integrated discrimination improvement (IDI), respectively.
Results
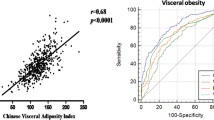
Their mean age was 53.8 years (SD: 7.1 years), the mean body mass index (BMI) was 23.7 kg/m2, and 44.8% of the subjects were male. Both male and female with MA had higher VAT levels compared to subjects with normal metabolism (MN), and male had higher SAT levels than female (P < 0.05). Higher VAT was significantly associated with MA with ORs in the fourth quartile (Q4) of 6.537 (95% CI = 3.394–12.591) for male and 3.364 (95% CI = 1.898–5.962) for female (P for trend < 0.05). In female, VAT could increase the risk of metabolic abnormalities, but SAT could increase the risk of MA in the second and fourth quartiles (Q2 and Q4) only at BMI > 24 kg/m2. In male, VAT improved the predictive value of MA compared to BMI and waist circumference (WC), the AUC was 0.727 (95% CI = 0.687–0.767), the NRI was 0.139 (95% CI = 0.070–0.208) and 0.106 (95% CI = 0.038–0.173), and the IDI was 0.074 (95% CI = 0.053–0.095) and 0.046 (95% CI = 0.026–0.066). Similar results were found in female.
Conclusions
In male, VAT and SAT could increase the risk of metabolic abnormalities both at BMI < 24 kg/m2 and at BMI ≥ 24 kg/m2. In female, VAT could increase the risk of metabolic abnormalities but SAT could increase the risk of MA in the second and fourth quartiles (Q2 and Q4) only at BMI > 24 kg/m2. Deposition of abdominal adipose tissue was associated with metabolic abnormalities. VAT improved the predictive power of MA.
Similar content being viewed by others
Introduction
Obesity, especially central obesity, is a well-established risk factor for a several diseases, such as dyslipidemia, type 2 diabetes (T2DM), cardiovascular diseases (CVD), and all-cause mortality [1, 10, 11, 25,26,27]. Several Caucasian studies have shown that VAT is more strongly associated with type 2 diabetes, hypertension and hyperlipidemia in female [10, 25, 28]. In our Additional file 1: Table S3, we observed that the effect of VAT on high TG and low-HDL was higher in male, indicating that VAT may have more striking effect on lipid metabolism in male than female. The possible reason maybe that only a limited number of confounders were adjusted, which may have affected the results. An expanded study of the Chinese population is necessary to determine the gender differences in the contribution of VAT. In general, the relationship between VAT and metabolic outcomes is relatively stable, which may be related to multiple biological effects of VAT.
SAT is known to have adverse effects on a variety of metabolic risk factors and may have unique pathogenic properties independent of BMI [1, 6, 25, 29], and the effects of different levels of SAT on cardiometabolic factors are inconsistent [1, 6, 13, 19, 25, 30]. Consistent with previous studies [30,31,32], our study (See Additional file 1: Tables S1, S2) showed that higher SAT was not associated with hypertension, higher TG, and lower HDL-C risk after adjustment for age, smoke, drink, and menstrual history (for women), and SAT may be a protective factor for blood sugar. Several studies with European or African populations have found independent associations of SAT with high blood pressure (H-BP) and HDL-C [1, 14, 26], suggesting that SAT has different effects in different ethnic groups. A possible explanation for this sex difference in SAT is the different sex steroid hormone profiles, as these sex hormones are important in regulating adipose tissue distribution and energy metabolism [33, 34]. There are also several hypotheses for the protective effect of SAT to explain this observation. One is that smaller adipocytes, SAT are more sensitive to insulin and have a greater capacity to absorb fatty acids and triglycerides and therefore can act as a powerful buffer to prevent excess fat from entering non-adipose tissue [35]. On the other hand, SAT can secrete more favorable adipokines such as adiponectin, with antidiabetics and antiatherogenic properties [18, 23]. Therefore, the different effects of SAT on metabolic outcomes may be related to its biological functions. Since SAT has different effects on metabolic components in different sexes, it may result in a less stable correlation between SAT and metabolic abnormality.
Previous studies have shown that baseline and changes in VAT were independent predictors of future dyslipidemia, but BMI and SAT were not associated with future development of atherosclerotic dyslipidemia [36]. This result is consistent to our study that VAT is a better predictor for MA compared with BMI and WC.
There are some advantages in our study. Areas of SAT and VAT were measured using MRI, which is the gold standard method of determining abdominal adipose tissue [37]. The data, including anthropometric and questionnaire-based information, were collected by trained health professionals, and the biochemical measurements followed the standard protocols. Our study also has some limitations. First, we cannot infer a causal relationship between the adipose indices and the metabolic abnormality because of the cross-sectional design. Second, this study included limited confounding factors, such as not including regional fat distribution, such as deep SAT and superficial SAT, and medication use, which may have biased the results. Thirdly, the sample size of this study was relatively small. Finally, our data were based on only one single ethnic group, thus the results may not be applied to other ethnicities.
Conclusions
In male, VAT and SAT could increase the risk of metabolic abnormalities both at BMI < 24 kg/m2 and at BMI ≥ 24 kg/m2. In female, VAT could increase the risk of metabolic abnormalities but SAT could increase the risk of MA in the second and fourth quartiles (Q2 and Q4) only at BMI > 24 kg/m2. Deposition of abdominal adipose tissue was associated with metabolic abnormalities. VAT improved the predictive power of MA.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- SAT:
-
Subcutaneous adipose tissue
- VAT:
-
Visceral adipose tissue
- MRI:
-
Nuclear magnetic resonance imaging
- BMI:
-
Body mass index
- WC:
-
Waist circumference
- WHR:
-
Waist-to-hip ratio
- SBP:
-
Systolic blood pressure
- DBP:
-
Diastolic blood pressure
- FPG:
-
Fasting plasma glucose
- OGTT-2h:
-
2-Hour post oral glucose tolerance test
- TC:
-
Total cholesterol
- TG:
-
Triglyceride
- HDL-C:
-
High density lipoprotein cholesterol
- LDL-C:
-
Low density lipoprotein cholesterol
- MA:
-
Metabolic abnormality
- MN:
-
Metabolic normality
References
Abraham TM, Pedley A, Massaro JM, Hoffmann U, Fox CS. Association between visceral and subcutaneous adipose depots and incident cardiovascular disease risk factors. Circulation. 2015;132(17):1639–47.
Rothney MP, Catapano AL, **a J, Wacker WK, Tidone C, Grigore L, et al. Abdominal visceral fat measurement using dual-energy X-ray: association with cardiometabolic risk factors. Obesity (Silver Spring, Md). 2013;21(9):1798–802.
Jackson AS, Stanforth PR, Gagnon J, Rankinen T, Leon AS, Rao DC, et al. The effect of sex, age and race on estimating percentage body fat from body mass index: The Heritage Family Study. Int J Obes Relat Metab Disord J Int Assoc Study Obes. 2002;26(6):789–96.
Keum N, Lee DH, Kim R, Greenwood DC, Giovannucci EL. Visceral adiposity and colorectal adenomas: dose-response meta-analysis of observational studies. Ann Oncol. 2015;26(6):1101–9.
Chen P, Hou X, Hu G, Wei L, Jiao L, Wang H, et al. Abdominal subcutaneous adipose tissue: a favorable adipose depot for diabetes? Cardiovasc Diabetol. 2018;17(1):93.
Liu J, Fox CS, Hickson DA, May WD, Hairston KG, Carr JJ, et al. Impact of abdominal visceral and subcutaneous adipose tissue on cardiometabolic risk factors: the Jackson Heart Study. J Clin Endocrinol Metab. 2010;95(12):5419–26.
Kwon H, Kim D, Kim JS. Body fat distribution and the risk of incident metabolic syndrome: a longitudinal cohort study. Sci Rep. 2017;7(1):10955.
Porter SA, Massaro JM, Hoffmann U, Vasan RS, O’Donnel CJ, Fox CS. Abdominal subcutaneous adipose tissue: a protective fat depot? Diabetes Care. 2009;32(6):1068–75.
Hoyer D, Boyko EJ, McNeely MJ, Leonetti DL, Kahn SE, Fujimoto WY. Subcutaneous thigh fat area is unrelated to risk of type 2 diabetes in a prospective study of Japanese Americans. Diabetologia. 2011;54(11):2795–800.
Karlsson T, Rask-Andersen M, Pan G, Höglund J, Wadelius C, Ek WE, et al. Contribution of genetics to visceral adiposity and its relation to cardiovascular and metabolic disease. Nat Med. 2019;25(9):1390–5.
Chen Y, Zhang Z, Wang J, Sun H, Zhao X, Cheng X, et al. Sex differences in the association of abdominal adipose tissue and anthropometric data with untreated hypertension in a Chinese population. Biol Sex Differ. 2020;11(1):38.
Lesser IA, Gasevic D, Lear SA. The effect of body fat distribution on ethnic differences in cardiometabolic risk factors of Chinese and Europeans. Appl Physiol Nutr Metab. 2013;38(7):701–6.
Rønn PF, Andersen GS, Lauritzen T, Christensen DL, Aadahl M, Carstensen B, et al. Ethnic differences in anthropometric measures and abdominal fat distribution: a cross-sectional pooled study in Inuit, Africans and Europeans. J Epidemiol Community Health. 2017;71(6):536–43.
Bertoli S, Leone A, Vignati L, Spadafranca A, Bedogni G, Vanzulli A, et al. Metabolic correlates of subcutaneous and visceral abdominal fat measured by ultrasonography: a comparison with waist circumference. Nutr J. 2016;15:2.
Zheng R, Yang M, Bao Y, Li H, Shan Z, Zhang B, et al. Prevalence and determinants of metabolic health in subjects with obesity in Chinese population. Int J Environ Res Public Health. 2015;12(11):13662–77.
Alberti KGMM, Zimmet P, Shaw J. The metabolic syndrome—a new worldwide definition. The Lancet. 2005;366(9491):1059–62.
Liu Y, Liu J, Gao Y, Zheng D, Pan W, Nie M, et al. The body composition in early pregnancy is associated with the risk of development of gestational diabetes mellitus late during the second trimester. Diabetes Metab Syndr Obes Targets Ther. 2020;13:2367–74.
Ibrahim MM. Subcutaneous and visceral adipose tissue: structural and functional differences. Obes Rev. 2010;11(1):11–8.
Tang L, Zhang F, Tong N. The association of visceral adipose tissue and subcutaneous adipose tissue with metabolic risk factors in a large population of Chinese adults. Clin Endocrinol (Oxf). 2016;85(1):46–53.
Kahn CR, Wang G, Lee KY. Altered adipose tissue and adipocyte function in the pathogenesis of metabolic syndrome. J Clin Investig. 2019;129(10):3990–4000.
Despres JP, Lemieux I, Bergeron J, Pibarot P, Mathieu P, Larose E, et al. Abdominal obesity and the metabolic syndrome: contribution to global cardiometabolic risk. Arterioscler Thromb Vasc Biol. 2008;28(6):1039–49.
Misra A, Vikram NK. Clinical and pathophysiological consequences of abdominal adiposity and abdominal adipose tissue depots. Nutrition (Burbank, Los Angeles County, Calif). 2003;19(5):457–66.
Chait A, den Hartigh LJ. Adipose tissue distribution, inflammation and its metabolic consequences, including diabetes and cardiovascular disease. Front Cardiovasc Med. 2020;7:22.
Després JP, Lemieux I. Abdominal obesity and metabolic syndrome. Nature. 2006;444(7121):881–7.
Fox CS, Massaro JM, Hoffmann U, Pou KM, Maurovich-Horvat P, Liu CY, et al. Abdominal visceral and subcutaneous adipose tissue compartments: association with metabolic risk factors in the Framingham Heart Study. Circulation. 2007;116(1):39–48.
Rønn PF, Andersen GS, Lauritzen T, Christensen DL, Aadahl M, Carstensen B, et al. Abdominal visceral and subcutaneous adipose tissue and associations with cardiometabolic risk in Inuit, Africans and Europeans: a cross-sectional study. BMJ Open. 2020;10(9):e038071.
Bidulescu A, Liu J, Hickson DA, Hairston KG, Fox ER, Arnett DK, et al. Gender differences in the association of visceral and subcutaneous adiposity with adiponectin in African Americans: the Jackson Heart Study. BMC Cardiovasc Disord. 2013;13:9.
Borel AL, Nazare JA, Smith J, Aschner P, Barter P, Van Gaal L, et al. Visceral, subcutaneous abdominal adiposity and liver fat content distribution in normal glucose tolerance, impaired fasting glucose and/or impaired glucose tolerance. Int J Obes (Lond). 2015;39(3):495–501.
Oka R, Miura K, Sakurai M, Nakamura K, Yagi K, Miyamoto S, et al. Impacts of visceral adipose tissue and subcutaneous adipose tissue on metabolic risk factors in middle-aged Japanese. Obesity (Silver Spring). 2010;18(1):153–60.
Matsha TE, Ismail S, Speelman A, Hon GM, Davids S, Erasmus RT, et al. Visceral and subcutaneous adipose tissue association with metabolic syndrome and its components in a South African population. Clin Nutr ESPEN. 2019;32:76–81.
Koh H, Hayashi T, Sato KK, Harita N, Maeda I, Nishizawa Y, et al. Visceral adiposity, not abdominal subcutaneous fat area, is associated with high blood pressure in Japanese men: the Ohtori study. Hypertens Res. 2011;34(5):565–72.
Zhao X, Gang X, Liu Y, Sun C, Han Q, Wang G. Using metabolomic profiles as biomarkers for insulin resistance in childhood obesity: a systematic review. J Diabetes Res. 2016;2016:8160545.
Palmer BF, Clegg DJ. The sexual dimorphism of obesity. Mol Cell Endocrinol. 2015;402:113–9.
Karastergiou K. The interplay between sex, ethnicity, and adipose tissue characteristics. Curr Obes Rep. 2015;4(2):269–78.
Kim S, Cho B, Lee H, Choi K, Hwang SS, Kim D, et al. Distribution of abdominal visceral and subcutaneous adipose tissue and metabolic syndrome in a Korean population. Diabetes Care. 2011;34(2):504–6.
Hwang YC, Fujimoto WY, Hayashi T, Kahn SE, Leonetti DL, Boyko EJ. Increased visceral adipose tissue is an independent predictor for future development of atherogenic dyslipidemia. J Clin Endocrinol Metab. 2016;101(2):678–85.
Maislin G, Ahmed MM, Gooneratne N, Thorne-Fitzgerald M, Kim C, Teff K, et al. Single slice vs. volumetric MR assessment of visceral adipose tissue: reliability and validity among the overweight and obese. Obesity (Silver Spring). 2012;20(10):2124–32.
Acknowledgements
We also would like to thank all the participants and investigators that took part in this study.
Funding
This work was supported by the grants from National Key Research and Development Program of China (2017YFC0907004), Hangzhou Science and Technology Project (20171226Y27), and Zhejiang Health Science and technology Project (2021KY268), Key Medical Discipline of Hangzhou (Disinfection and Vector Biological Control). The funder has no role in the design of the study, collection, analysis, and interpretation of data.
Author information
Authors and Affiliations
Contributions
Conceptualization, YZ; Data curation, QC; Formal analysis, QC and XS; Funding acquisition, XZ and YZ; Investigation, QL and JZ; Project administration, XZ and YZ; Supervision, YZ; Writing—original draft, XZ and QC; Writing—review and editing, YZ, QW, and ZC. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The study was approved by the institutional review board at Zhejiang University, Zhejiang, China. All participants were given their written informed consents.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1.
Supplementary Tables.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zhang, X., Chen, Q., Sun, X. et al. Association between MRI-based visceral adipose tissues and metabolic abnormality in a Chinese population: a cross-sectional study. Nutr Metab (Lond) 19, 16 (2022). https://doi.org/10.1186/s12986-022-00651-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12986-022-00651-x