Abstract
Background
Muscle mass loss is an age-related process that can be exacerbated by lifestyle, environmental and other factors, but can be mitigated by good sleep. The objective of this study was to investigate the correlation between varying time lags of sleep duration and the decline in muscle mass among individuals aged 60 years or older by using real-world health monitoring data obtained from wearable devices and smart home health monitoring devices.
Methods
This study included 86,037 observations from 2,869 participants in the Mobile Support System database. Missing data were supplemented by multiple imputation. The investigation utilized generalized estimating equations and restricted cubic spline curve to examine the relationship between sleep duration and low muscle mass. Various lag structures, including 0, 1, 2, 0–1, 0–2, and 1–2 months, were fitted, and the interaction effect of observation time with sleep duration was estimated for each lag structure. Additionally, subgroup analyses were conducted. The models were adjusted for various covariates, including gender, age, body mass index, footsteps, smoking status, drinking status, marital status, number of chronic diseases, number of medications, diabetes mellitus, hyperlipidemia, coronary artery disease, respiratory disease, and musculoskeletal disease and an interaction term between time and sleep duration.
Results
The results of the generalized estimating equation showed a significant correlation (p < 0.001) between sleep duration of 8 h or more and low muscle mass in older adults, using 6–7 h of sleep as a reference. This effect was seen over time and prolonged sleep accumulated over multiple months had a greater effect on muscle mass loss than a single month. The effect of long sleep duration on muscle mass loss was significantly greater in females than in males and greater in the over-75 than in the under-75 age group. Restricted cubic spline plots showed a non-linear relationship between sleep duration and low muscle mass (p < 0.001).
Conclusions
This study found an association between sustained nighttime sleep of more than eight hours and decreased muscle mass in older adults, especially older women.
Similar content being viewed by others
Introduction
Over the course of a person’s life, age-related loss of muscle mass begins in middle age and decreases at a rate of 1% per year [1]. A longitudinal study [2] found that the rate of muscle mass loss was 0.8-0.98% for men and 0.64-0.7% for women over the age of 75. Loss of muscle mass with age leads to reduced muscle strength and limited physical function and is a significant risk factor for disability and death [3, 4]. In addition to the natural physiological process of aging [1, 5, 6], other factors [6] such as lifestyle choices [6], environment conditions, dietary habits [7, 8], and chronic diseases [9] accelerate the loss of muscle mass.
Both a prospective cohort [10] and a longitudinal study [11] had shown a U-shaped relationship between sleep duration and mortality in people over the age of sixty. Nocturnal sleep patterns in older adults change with age [12], and being sedentary or bedridden [13]can lead to disrupted circadian rhythms [12, 14], which, if maintained over time, can have a negative impact on the health of older adults [12, 15].
Sleep is recognized as an important factor in maintaining muscle mass [16].Several cross-sectional studies [17, 18] had demonstrated an association between too much or too little sleep and decreased muscle mass in older adults. The validity of research hypotheses requires evidence to further demonstrate causality, but there are fewer longitudinal studies [19,20,21]in this area. The results of related studies suggested that sleep duration affects muscle mass decline in older adults, and gender differences in the effect of sleep duration on muscle mass decline have not been consistent across these studies.
Therefore, based on real-world collected data, this study explored the longitudinal associations between sleep duration and low muscle mass in older adults, as well as differences in effects across age and gender.
Methods
Study design and participants
The dataset used in this study was extracted from a database based on a mobile support system [22]. It recorded basic user information and health-related observations of older adults aged 60 years or older recruited from communities in five Chinese cities (Taiyuan, **an, Nanchang, Guangzhou, and Hefei) between January 1, 2018, and September 31, 2022. The exclusion criteria for the data were as follows: (1) age less than 60 years; (2) the upload to the database was not in the above time period; (3) exclusion of participants with less than 1 sleep duration or low muscle mass data; (4) excluded indicators with a relatively large degree of deficiency. We preprocessed the collected data by logistic checking and outlier checking and computed the average observations per user per month. A total of 86,037 observations from 2,869 participants were included in the statistical analysis. All study procedures were approved by the Ethical Review Committee of Peking University. Informed consent was obtained from all individual participants included in the study.
Data collection
Measurements and definitions
Participants were required to wear an exercise bracelet daily to monitor daily steps and sleep. Height was measured with a height and weight meter (HDM-300; Zhejiang Huaju Company, China) [22], which required light clothing, no shoes, and standing. Body fat weight was measured with a multi-frequency bioelectrical impedance analyzer (InBody 720; Biospace, Seoul, Korea) with 8-point tactile electrodes [23]. Before the test, the subject should fast or empty stomach for more than two hours, avoid drinking a lot of water and strenuous exercise, remove metal objects and wear single clothes. During the measurement, the subject stood barefoot on the electrode, entered the subject’s information according to the prompts, then held the handle with both hands and lifted it upward, with both arms naturally hanging down at 30° to the body, and stood still for the measurement. The whole measurement time is about 1–2 min.
Sleep duration was categorized into 4 groups: <6 h for insufficient sleep [24], 6–7 h for reference, 7–8 h, and ≥ 8 h for excessive sleep [17]. Fat-free mass index (FFMI) was calculated as fat-free mass (kg) divided by the square of height. Low muscle mass was defined [25] as FFMI < 16.31 kg/m² for men and FFMI < 13.96 kg/m² for women.
The data from each measurement taken by the participants were automatically uploaded via Bluetooth to an application on their cell phones and stored to the cloud-system [22].
Covariates
Baseline demographic information on participants was collected through a questionnaire (Supplementary questionnaire). Smoking status was categorized into never smokers and smokers (including current and former smokers); drinking status was categorized into never drinkers and drinkers (including current and former drinkers); marital status was categorized into single (including divorced, widowed, and separated) and married; and the number of medications [19] was categorized into < 5 and ≥ 5. We calculated the number of chronic conditions and categorized them as 0 (reference value), 1, 2, and ≥ 2. Daily steps [26] was considered as an indicator to assess physical activity and categorized into 3 classes, < 4000 steps, 4000–8000 steps, > 8000 steps (reference value). BMI (kg/m²) was calculated by weight/height² and referred to the GLIM [27] criteria for the use of BMI as an indicator for the assessment of nutritional status: <18.5 kg/m² was defined as malnutrition.
Statistical analysis
Differences in demographic characteristics between participants with and without low muscle mass at baseline values were compared. Continuous variables were expressed as mean and standard deviation (SD) and analyzed by Student’s t-test. For categorical variables, correlation analyses were performed using χ² when expressed as counts (%).
We imputed missing values using a fully conditional specification with multiple imputation, using logistic regression for categorical variables and regression for continuous variables. We included daily steps, BMI, smoking status, marital status, number of chronic diseases, number of medications taken, diabetes, coronary heart disease, respiratory disease, and musculoskeletal disease as auxiliary variables in the imputed model. Drinking status, sex, age, observation time, hyperlipidemia, and an interaction term between sleep duration and observation time (but not included in the imputation model) were included in the statistical analysis model based on the imputed model. The distributions of the variables were compared between the different populated and original data sets. Regression coefficients were summarized using Rubin’s Rule. Sensitivity analyses were performed by comparing the Odds ratio and 95% confidence intervals as well as the p-values between the original data set and the imputed composite results.
Generalized estimating equations were used to assess the association between sleep duration and low muscle mass in different lag structures. We fitted different lag structures from the current month (lag0) to three lag months (lag3) as well as two months (lag0-1, 1–2) and three months (lag0-2). We examined the interaction of sleep duration with observation time and modeled three models in each lag structure. In Model A, sleep duration was not adjusted for other covariates. In Model B, age, gender, and body mass index were added to adjust for sleep duration. Model C adjusted Model B by adding smoking status, drinking status, marital status, number of daily steps, number of chronic diseases, number of medications, diabetes, hyperlipidemia, coronary heart disease, respiratory disease, and musculoskeletal disease. Stratified analyses were conducted among older adults of different genders (female and male) and age groups (60–65 years and ≥ 75 years). Restricted cubic spline curve was also used to examine nonlinear trends between sleep duration and low muscle mass in different lag structures.
All analyses were performed by SAS (version 9.4, SAS Institute Inc, Cary, CN, USA) and R, version 4.3.2 (R Project for Statistical Computing). P < 0.05 was considered statistically significant.
Results
Participants
Differences between low and non-low muscle mass participants at baseline was demonstrated in Table 1. The statistical analysis culminated in the use of 86,037 records from 2869 participants aged 60 years or older. Of these, 70.79% were female, with a mean age of (72.39 ± 6.37) years and a mean duration of observation of (29.99 ± 17.24) months. Compared with non-low-muscle-mass participants, low-muscle-mass participants were more likely to have the following characteristics: lower BMI, an average of fewer than 4,000 steps per day, and 2 or more chronic diseases.
As shown in the figure above (Fig. 1), the frequency of distribution of sleep information recorded in the database for different years, as well as the average change in sleep duration and body mass index for participants with different baseline sleep duration.
The figure illustrated the changes in sleep duration(a) and BMI(b) over time in older adults with low and non-low muscle mass (Fig. 2). It showed that older adults with low muscle mass sleep longer than older adults with non-low muscle mass and that both sleep duration and BMI tend to decrease with age.
Results of multiple imputation
The subset of the mobile support system generated for this study contained 86,037 observations with a missing values rate of about 7% (Table S1). Variables with missing data were daily steps (34.78%), body mass index (30.05%), muscle mass (30.05%), sleep duration (6.92%), age (3.59%), and gender (3.59%), whereas the other variables were almost non-missing. Data were assumed missing at random and non-monotonic missing data pattern (Table S2) and seven datasets were imputed.
A comparison of the distribution of the imputed values with the observed values was showed in Table S3. The means and standard deviations of the continuous variables across the different datasets, even though there was some difference, the overall difference was small, and the distribution was relatively stable. The results of the sensitivity analysis indicated that the results of the multiple repetitions of the estimation process was stable. (Table S4).
Longitudinal association between sleep duration and low muscle mass
We used sleep duration as the independent variable and muscle mass as the dichotomous response variable, and detailed assignments of the other metrics are shown in Table S5.
The relationship between sleep duration and low muscle mass in different lag structures was showed in Table 2. The results in the different lag structures showed a statistically significant effect of slee** more than 8 h on low muscle mass. (p < 0.05) In the single-month lag model, the effect of long sleep duration on muscle mass reduction became progressively more pronounced with increasing time. In the multi-month lag model, the effect of long sleep duration was significantly greater than in the single-month lag model. While all results were statistically significant, it should be noted that the odds ratios associated with excessive sleep duration were not particularly high.
In addition, the result showed significant differences between gender and body mass index and low muscle mass, respectively. (Table S6).
There was a nonlinear correlation between sleep duration and low muscle mass onset (P < 0.001). The relationship of the sleep duration and low muscle mass exposure-dose curves for the six lag structures showed that the risk of low muscle mass incidence decreased progressively with increasing sleep duration when the sleep duration was less than 420 minutes, increased with increasing time after reaching a minimum at 370–374 minutes, and decreased slowly with increasing time after reaching a maximum at 463–467 minutes (Fig. 3).
The exposure-response curve of sleep duration and low muscle mass. The x-axis is the lag 0, 1, 2, 0–1, 0–2, 1-2-month sleep duration. Y-axis is the odds ratio, after adjusting for sex, age, smoking status, marital status, number of chronic diseases, number of medications taken, diabetes, coronary heart disease, respiratory disease, and musculoskeletal disease, drinking status, hyperlipidemia, and observation time, is shown by the red solid line, and Light red areas represent 95% confidence intervals
Subgroup analyses
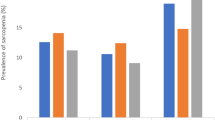
In subgroup analyses (Fig. 4), significant correlations between long sleep duration and low muscle mass were found only in males at lags of 2 months, 0–1 month, and 1–2 months, whereas in the remaining subgroups significant correlations were found for each sleep lag construct. The effect of long sleep duration on muscle mass loss was significantly greater in females than in males and greater in the multi-month model than in the single-month model. In the age subgroups, the effect of multi-month cumulative long sleep duration on low muscle mass was lower in the over-75 than in the under-75 age group, whereas in the one-month lag structure this effect was lower only in the lag 0-month than in the under-75 age group. As the subgroup analyses showed, while some results were statistically significant, the odds ratios were not high.
Discussion
Our study was a longitudinal study based on real data from community-dwelling older adults managed on a cloud-based platform. We examined the linear and nonlinear relationships between sleep duration and low muscle mass in older adults over the age of 60. This finding indicated that there was a potential association between sustained sleep duration of more than 8 h and loss of muscle mass, and that this effect became apparent gradually over time. The effect of long sleep duration was also clearly observed in women and older adults of different ages. In all age groups, the effect of a cumulative multi-month lag in sleep duration did not show a greater effect with advanced age, rather it was more pronounced with a single-month lag. However, this potential correlation was not strong, reflecting the fact that the reduction in muscle mass is a multifactorial process, and that longer sleep duration may be a contributing factor, but not a major influence. Therefore, while longer sleep duration may be associated with reduced muscle mass, it is important to interpret these findings in the context of wider health factors and to see it as one of the ways to understand and respond to changes in muscle mass in older people.
Our assessment performed with generalized estimating equations indicated a potential association between sustained sleep of 8 h or more and the progression of low muscle mass in older adults. There are not many studies on the dose-response relationship between sleep duration and low muscle mass in older adults. However, some studies [10, 11, 28]have shown a nonlinear relationship between sleep duration and low muscle mass or sarcopenia in older adults. Hence, we also further verified the relationship between sleep duration and low muscle mass using a restricted cubic spline curve, which showed that when sleep duration was less than 7 h, the risk of develo** sarcopenia decreased with longer sleep duration. Nonetheless, when sleep duration exceeded 7 h, sleep duration was associated with the development of hypomuscular mass disorder. This suggested that habitual long sleep duration in older adults is detrimental to muscle mass loss. This result was consistent with other longitudinal studies [19, 20].A longitudinal study [21] of Chinese older adults found that both longer and shorter sleep duration were associated with muscle loss. The difference was that the study was conducted with older adults in rural and suburban China, whereas our study focused on community-based older adults in a large city. Rural and urban older adults have different lifestyles and environments, and these differences make thresholds for low muscle mass inconsistent. In addition, we examined the effect of lagged sleep duration at different periods in the past three months on low muscle mass, whereas most of the other studies focused on short-term sleep duration, such as the past month and seven days, and were mostly based on self-reporting [17, 19] with some recall bias.
Surprisingly, our study found that men were more likely to experience a loss of muscle mass, and older women were instead more likely to experience this after accounting for sleep duration. The results vary from study to study due to different hormone levels, sample sizes, and definitions of muscle mass in different genders [29].However, current research generally recognizes men as a risk factor for muscle mass loss [30,31,32].When considering sleep duration as a factor, a systematic review concluded that longer sleep duration is more likely to be associated with sarcopenia in older adults, especially women [33]. In contrast, another longitudinal study [20] showed that long sleep duration was associated with low muscle mass only in community-based male older adults. The reason for the inconsistency with previous findings may be that most of the participants in our study were women, and although the onset and rate of muscle mass loss in men is earlier than that in women, changes in hormone levels [5, 34] due to menopause in women accelerate bone loss and muscle decay, and female older adults instead lose more muscle mass in later life [35].
It is important to note that aging plays an important role in muscle mass loss [16], which is mainly due to muscle fiber atrophy and lack of exercise [2, 4, 6]. Aging, which is responsible for chronic diseases, also leads to a faster loss of muscle mass in older adults than in younger adults. Our study found that the cumulative effect of multiple months of sleep on low muscle mass was stronger than that of a single month of sleep, so we must also be wary of older adults who are chronically drowsy for more than a month, especially in the age group of 60–75 years.
Unfortunately sleep structure and duration change with age [12]: either too much or too little persistent nocturnal sleep leads to circadian rhythm disturbances [12, 36], hormone secretion [5, 12, 16, 37] and altered body metabolism [38, 39]. Sleep duration contributes to the loss of muscle mass mainly by affecting the balance of muscle protein synthesis, catabolism and metabolism [6, 12, 37].For example: decreased secretion of growth hormone, testosterone and Insulin-like Growth Factor 1(IGF-1), and insulin resistance inhibit the activity of the Insulin-like Growth Factor 1 (IGF-1)/Phosphoinositide 3-Kinase (PI3K)/Protein Kinase B (Akt) and the mechanistic Target of Rapamycin (mTOR), which reduces muscle protein synthesis and promotes the secretion of Muscle Growth Inhibitory Factor (MGIF) and the expression of Regulated in development and DNA damage responses 1 (REDD1);Increased secretion of cortisol inhibits mTOR activity, promotes REDD1 expression to reduce muscle protein synthesis, and enhances the effects of Atrogin-1 and Muscle RING-finger protein-1 (MuRF-1) through activation of Forkhead box O (FOXO), leading to muscle protein degradation; Chronic inflammation [5, 16, 40], as indicated by elevated levels of pro-inflammatory markers such as c-reactive protein and Interleukin- 6(IL-6), affects muscle anabolism and catabolism. In addition, excessive sleep, and prolonged bed rest [13] lead to reduced daytime physical activity and the effects of chronic diseases.
This study revealed a causal relationship between sleep duration at different lags and low muscle mass in older adults, and the data from these studies are from real-world, long-term follow-up records, allowing for better dynamic analysis of observational variables to demonstrate the reliability of our findings. However, there were some limitations of this study: firstly, we were unable to assess other potential confounders such as information reflecting social status and dietary information; and secondly, medical and medication histories were collected through questionnaires rather than obtained by reviewing medical records.
Sleep duration is an important characteristic of sleep, and measuring the association between sleep and muscle in older adults should consider not only the effect of total sleep duration, but also the effect of other characteristics such as sleep quality and sleep efficiency [41]. Therefore, further future research would be to explore the association between different sleep characteristics and muscle mass after fully considering other covariates.
Conclusion
Sleep duration in older adults may reflect their physical health to some extent. Our study confirmed a longitudinal association between long sleep duration and low muscle mass in the last three months, especially in older women. We should be alert to older adults who habitually maintain a nightly sleep duration of 8 h or more, which is important to promote their healthy aging.
Data availability
The data that support the findings of this study are available on request from the corresponding author, [**aotong Dai], upon reasonable request.
References
Wilkinson DJ, Piasecki M, Atherton PJ. The age-related loss of skeletal muscle mass and function: measurement and physiology of muscle fibre atrophy and muscle fibre loss in humans. Ageing Res Rev. 2018;47:123–32.
Mitchell WK, Williams J, Atherton P, Larvin M, Lund J, Narici M. Sarcopenia, Dynapenia, and the impact of advancing age on human skeletal muscle size and strength; a quantitative review. Front Physiol. 2012;3:260.
Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, Martin FC, Michel JP, Rolland Y, Schneider SM, et al. Sarcopenia: European consensus on definition and diagnosis: report of the European Working Group on Sarcopenia in Older people. Age Ageing. 2010;39(4):412–23.
Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T, Cooper C, Landi F, Rolland Y, Sayer AA, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019;48(1):16–31.
Keller K, Engelhardt M. Strength and muscle mass loss with aging process. Age and strength loss. Muscles Ligaments Tendons J. 2013;3(4):346–50.
Shur NF, Creedon L, Skirrow S, Atherton PJ, MacDonald IA, Lund J, Greenhaff PL. Age-related changes in muscle architecture and metabolism in humans: the likely contribution of physical inactivity to age-related functional decline. Ageing Res Rev. 2021;68:101344.
Ouyang Y, Huang F, Zhang X, Li L, Zhang B, Wang Z, Wang H. Association of Dietary Protein Intake with muscle Mass in Elderly Chinese: a cross-sectional study. Nutrients 2022, 14(23).
Mithal A, Bonjour JP, Boonen S, Burckhardt P, Degens H, El Hajj Fuleihan G, Josse R, Lips P, Morales Torres J, Rizzoli R, et al. Impact of nutrition on muscle mass, strength, and performance in older adults. Osteoporos International: J Established as Result Cooperation between Eur Foundation Osteoporos Natl Osteoporos Foundation USA. 2013;24(5):1555–66.
Kalyani RR, Corriere M, Ferrucci L. Age-related and disease-related muscle loss: the effect of diabetes, obesity, and other diseases. Lancet Diabetes Endocrinol. 2014;2(10):819–29.
Yeo Y, Ma SH, Park SK, Chang SH, Shin HR, Kang D, Yoo KY. A prospective cohort study on the relationship of sleep duration with all-cause and disease-specific mortality in the Korean Multi-center Cancer Cohort study. J Prev Med Public Health = Yebang Uihakhoe Chi. 2013;46(5):271–81.
Gangwisch JE, Heymsfield SB, Boden-Albala B, Buijs RM, Kreier F, Opler MG, Pickering TG, Rundle AG, Zammit GK, Malaspina D. Sleep duration associated with mortality in elderly, but not middle-aged, adults in a large US sample. Sleep. 2008;31(8):1087–96.
Piovezan RD, Abucham J, Dos Santos RV, Mello MT, Tufik S, Poyares D. The impact of sleep on age-related Sarcopenia: possible connections and clinical implications. Ageing Res Rev. 2015;23(Pt B):210–20.
Dirks ML, Wall BT, van de Valk B, Holloway TM, Holloway GP, Chabowski A, Goossens GH, van Loon LJ. One week of Bed Rest leads to substantial muscle atrophy and induces whole-body insulin resistance in the absence of skeletal muscle lipid Accumulation. Diabetes. 2016;65(10):2862–75.
Choi YI, Park DK, Chung JW, Kim KO, Kwon KA, Kim YJ. Circadian rhythm disruption is associated with an increased risk of Sarcopenia: a nationwide population-based study in Korea. Sci Rep. 2019;9(1):12015.
Wang S, Wu Y, Ungvari GS, Ng CH, Forester BP, Gatchel JR, Chiu HFK, Kou C, Fu Y, Qi Y, et al. Sleep duration and its association with demographics, lifestyle factors, poor mental health and chronic diseases in older Chinese adults. Psychiatry Res. 2017;257:212–8.
Buchmann N, Spira D, Norman K, Demuth I, Eckardt R, Steinhagen-Thiessen E. Sleep, muscle Mass and muscle function in older people. Deutsches Arzteblatt Int. 2016;113(15):253–60.
Tan X, Titova OE, Lindberg E, Elmståhl S, Lind L, Schiöth HB, Benedict C. Association between Self-reported sleep duration and body composition in Middle-aged and older adults. J Clin Sleep Medicine: JCSM: Official Publication Am Acad Sleep Med. 2019;15(3):431–5.
Kim K, Shin D, Jung GU, Lee D, Park SM. Association between sleep duration, fat mass, lean mass and obesity in Korean adults: the fourth and fifth Korea National Health and Nutrition Examination Surveys. J Sleep Res. 2017;26(4):453–60.
Nakakubo S, Doi T, Tsutsumimoto K, Kurita S, Ishii H, Shimada H. Sleep duration and progression to Sarcopenia in Japanese community-dwelling older adults: a 4 year longitudinal study. J cachexia Sarcopenia Muscle. 2021;12(4):1034–41.
Lee H, Kim S, Kim BS, Kim M, Yang J, Bae H, Won CW. Sexual difference in Effect of Long Sleep Duration on Incident Sarcopenia after two years in Community-Dwelling older adults. Annals Geriatric Med Res. 2022;26(3):264–74.
Han P, Hou L, Liang Z, Chen W, Li J, Cheng Y, Zhou W, Zeng S, Pan J, Xu L, et al. Both short and long sleep durations are risk factors for Sarcopenia in Suburban-Dwelling older Chinese individuals: a 3-Year longitudinal study. Nat Sci Sleep. 2022;14:1089–96.
Zhou M, Zhang N, Zhang Y, Yan X, Li M, Guo W, Guo X, He H, Guo K, Ma G. Effect of Mobile-based lifestyle intervention on weight loss among the overweight and obese Elderly Population in China: a Randomized Controlled Trial. Int J Environ Res Public Health 2021, 18(16).
Guo X, Xu Y, He H, Cai H, Zhang J, Li Y, Yan X, Zhang M, Zhang N, Maddela RL, et al. Visceral fat reduction is positively associated with blood pressure reduction in overweight or obese males but not females: an observational study. Nutr Metabolism. 2019;16:44.
Svensson T, Saito E, Svensson AK, Melander O, Orho-Melander M, Mimura M, Rahman S, Sawada N, Koh WP, Shu XO, et al. Association of Sleep Duration with All- and major-cause mortality among adults in Japan, China, Singapore, and Korea. JAMA Netw open. 2021;4(9):e2122837.
Zeng P, Wu S, Han Y, Liu J, Zhang Y, Zhang E, Zhang Y, Gong H, Pang J, Tang Z, et al. Differences in body composition and physical functions associated with Sarcopenia in Chinese elderly: reference values and prevalence. Arch Gerontol Geriatr. 2015;60(1):118–23.
Saint-Maurice PF, Troiano RP, Bassett DR Jr., Graubard BI, Carlson SA, Shiroma EJ, Fulton JE, Matthews CE. Association of Daily Step Count and Step Intensity with Mortality among US adults. JAMA. 2020;323(12):1151–60.
Cederholm T, Jensen GL, Correia M, Gonzalez MC, Fukushima R, Higashiguchi T, Baptista G, Barazzoni R, Blaauw R, Coats AJS, et al. GLIM criteria for the diagnosis of malnutrition - A consensus report from the global clinical nutrition community. J cachexia Sarcopenia Muscle. 2019;10(1):207–17.
Chien MY, Wang LY, Chen HC. The relationship of Sleep duration with obesity and Sarcopenia in Community-Dwelling older adults. Gerontology. 2015;61(5):399–406.
Petermann-Rocha F, Balntzi V, Gray SR, Lara J, Ho FK, Pell JP, Celis-Morales C. Global prevalence of Sarcopenia and severe Sarcopenia: a systematic review and meta-analysis. J cachexia Sarcopenia Muscle. 2022;13(1):86–99.
Trevisan C, Vetrano DL, Calvani R, Picca A, Welmer AK. Twelve-year Sarcopenia trajectories in older adults: results from a population-based study. J cachexia Sarcopenia Muscle. 2022;13(1):254–63.
Lau EM, Lynn HS, Woo JW, Kwok TC, Melton LJ 3. Prevalence of and risk factors for Sarcopenia in elderly Chinese men and women. Journals Gerontol Ser Biol Sci Med Sci. 2005;60(2):213–6.
Chen X, Hou L, Zhang Y, Dong B. Analysis of the prevalence of Sarcopenia and its risk factors in the Elderly in the Chengdu Community. J Nutr Health Aging. 2021;25(5):600–5.
Li X, He J, Sun Q. Sleep duration and Sarcopenia: an updated systematic review and Meta-analysis. J Am Med Dir Assoc. 2023;24(8):1193–e12061195.
Sipilä S, Törmäkangas T, Sillanpää E, Aukee P, Kujala UM, Kovanen V, Laakkonen EK. Muscle and bone mass in middle-aged women: role of menopausal status and physical activity. J cachexia Sarcopenia Muscle. 2020;11(3):698–709.
Kim KM, Lim S, Oh TJ, Moon JH, Choi SH, Lim JY, Kim KW, Park KS, Jang HC. Longitudinal changes in muscle Mass and Strength, and Bone Mass in older adults: gender-specific associations between muscle and bone losses. Journals Gerontol Ser Biol Sci Med Sci. 2018;73(8):1062–9.
Morrison M, Halson SL, Weakley J, Hawley JA. Sleep, circadian biology and skeletal muscle interactions: implications for metabolic health. Sleep Med Rev. 2022;66:101700.
Dattilo M, Antunes HK, Medeiros A, Mônico Neto M, Souza HS, Tufik S, de Mello MT. Sleep and muscle recovery: endocrinological and molecular basis for a new and promising hypothesis. Med Hypotheses. 2011;77(2):220–2.
Baranwal N, Yu PK, Siegel NS. Sleep physiology, pathophysiology, and sleep hygiene. Prog Cardiovasc Dis. 2023;77:59–69.
Stich FM, Huwiler S, D’Hulst G, Lustenberger C. The potential role of Sleep in promoting a healthy body composition: underlying mechanisms determining muscle, Fat, and bone Mass and their Association with Sleep. Neuroendocrinology. 2022;112(7):673–701.
Dowd JB, Goldman N, Weinstein M. Sleep duration, sleep quality, and biomarkers of inflammation in a Taiwanese population. Ann Epidemiol. 2011;21(11):799–806.
Kim M, Sasai H, Kojima N, Kim H. Objectively measured night-to-night sleep variations are associated with body composition in very elderly women. J Sleep Res. 2015;24(6):639–47.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
Qiongyu Huang, Hongsheng Lin, and **aotong Dai were responsible for the conceptual and methodological parts of this project; Qiongyu Huang, Hongsheng Lin, and **ao Han were responsible for analyzing the data, interpreting the results, and writing the original manuscript; and Liuwei Zhang, Dafang Chen, and **aotong Dai were responsible for the reviews and revisions as well as the funding applications. The main coordinators and implementers of this project were **aotong Dai and Dafang Chen. All authors have read and approved this version.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The study was approved by the Ethics Committee of Peking University and the study procedures were in accordance with the Peking University Guidelines for the Research Process. Informed consent was obtained from all individual participants included in the study.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Huang, Q., Lin, H., ** more than 8 h: a silent factor contributing to decreased muscle mass in Chinese community-dwelling older adults. BMC Public Health 24, 1246 (2024). https://doi.org/10.1186/s12889-024-18520-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12889-024-18520-y