Abstract
Background
Hyperlipidemia, a heterogeneous group of disorders characterized by elevated plasma lipids in the blood, causes severe health problems, leading to fatty liver disease and nonalcoholic fatty liver disease. Thymoquinone, the major active chemical component of Nigella sativa, reportedly exerts a vast array of biological effects. Various studies have reported that Thymoquinone protects against liver injury.
Aims
The aim of this study was to investigate the possible protective effects of Thymoquinone against liver injury in hyperlipidemia-induced LDL-R−/− mice.
Methods
Eight-week-old male LDL-R−/− mice were randomly divided into three groups: a control group fed a normal diet and two groups fed a high-cholesterol diet or high-cholesterol diet mixed with Thymoquinone. All groups were fed different diets for 8 weeks. Blood samples were obtained from the inferior vena cava and collected in serum tubes. The samples were then stored at − 80 °C until used. Longitudinal sections of liver tissues were fixed in 10% formalin and then embedded in paraffin for histological evaluation. The remainder of the liver tissues were snap-frozen in liquid nitrogen for reverse transcription-polymerase chain reaction or western blotting.
Results
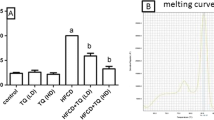
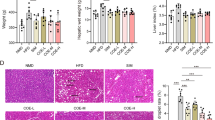
Our results demonstrated that Thymoquinone administration significantly reduced liver histological alterations by hyperlipidemia. Thymoquinone mitigated hyperlipidemia-induced liver injury as indicated by the suppression of metabolic characteristics, liver biochemical parameters, pyroptosis indicators, a macrophage marker, and the phosphatidylinositide 3-kinase signaling pathway.
Conclusions
Thymoquinone is a potential therapeutic agent for hyperlipidemia-induced liver injury.
Similar content being viewed by others
Introduction
Modern living environments and excessive energy intake have led to a tremendous increase in various metabolic diseases such as hyperlipidemia, diabetes, hypertension, and cardiovascular diseases [Limitations However, some limitations need to be noted. First, mice were randomly divided into 3 groups, we did not set a separate TQ group, therefore, the effect of TQ in ND group mice didn’t know. Second, we are not performed cell experiments to explore the protection mechanism of specific pathways of TQ on fatty liver injury. Further studies are needed to confirm this.
Data Availability
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
References
Bao CD, Sun B, Lan L, Qiao H, Zhang DF, Liu XY, et al. [Interaction between family history of diabetes and hyperlipidemia on risk of diabetes in population with normotension in Harbin: a cross-sectional study]. Zhonghua Liu **ng Bing Xue Za Zhi. 2017;38(5):611–4.
Lee HS, Lee YJ, Chung YH, Nam Y, Kim ST, Park ES, et al. Beneficial Effects of red yeast Rice on High-Fat Diet-Induced obesity, hyperlipidemia, and fatty liver in mice. J Med Food. 2015;18(10):1095–102.
Ewald N, Hardt PD, Kloer HU. Severe hypertriglyceridemia and pancreatitis: presentation and management. Curr Opin Lipidol. 2009;20(6):497–504.
Moore JB. Non-alcoholic fatty liver disease: the hepatic consequence of obesity and the metabolic syndrome. Proc Nutr Soc. 2010;69(2):211–20.
Hong Y, Deng C, Zhang J, Zhu J, Li Q. Neuroprotective effect of granulocyte colony-stimulating factor in a focal cerebral ischemic rat model with hyperlipidemia. J Huazhong Univ Sci Technolog Med Sci. 2012;32(6):872–8.
Chen YL, **ao CH, Hu ZX, Liu XS, Liu Z, Zhang WN et al. Dynamic lipid profile of hyperlipidemia mice. J Chromatogr B Analyt Technol Biomed Life Sci. 2017;1055–6:165 – 71.
Svegliati-Baroni G, Saccomanno S, Rychlicki C, Agostinelli L, De Minicis S, Candelaresi C, et al. Glucagon-like peptide-1 receptor activation stimulates hepatic lipid oxidation and restores hepatic signalling alteration induced by a high-fat diet in nonalcoholic steatohepatitis. Liver Int. 2011;31(9):1285–97.
Cairns SR, Peters TJ. Biochemical analysis of hepatic lipid in alcoholic and diabetic and control subjects. Clin Sci (Lond). 1983;65(6):645–52.
Yuan C, Liu C, Wang T, He Y, Zhou Z, Dun Y, et al. Chikusetsu saponin IVa ameliorates high fat diet-induced inflammation in adipose tissue of mice through inhibition of NLRP3 inflammasome activation and NF-κB signaling. Oncotarget. 2017;8(19):31023–40.
Zhong H, Liu M, Ji Y, Ma M, Chen K, Liang T, et al. Genipin reverses HFD-Induced Liver damage and inhibits UCP2-Mediated pyroptosis in mice. Cell Physiol Biochem. 2018;49(5):1885–97.
Gao YL, Zhai JH, Chai YF. Recent advances in the Molecular Mechanisms underlying pyroptosis in Sepsis. Mediators Inflamm. 2018;2018:5823823.
Song F, Hou J, Chen Z, Cheng B, Lei R, Cui P, et al. Sphingosine-1-phosphate receptor 2 signaling promotes caspase-11-dependent macrophage pyroptosis and worsens Escherichia coli Sepsis Outcome. Anesthesiology. 2018;129(2):311–20.
Zhu P, Duan L, Chen J, **ong A, Xu Q, Zhang H, et al. Gene silencing of NALP3 protects against liver ischemia-reperfusion injury in mice. Hum Gene Ther. 2011;22(7):853–64.
Henao-Mejia J, Elinav E, ** C, Hao L, Mehal WZ, Strowig T, et al. Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature. 2012;482(7384):179–85.
Artlett CM. The role of the NLRP3 inflammasome in fibrosis. Open Rheumatol J. 2012;6:80–6.
Miao CM, Jiang XW, He K, Li PZ, Liu ZJ, Cao D, et al. Bone marrow stromal cells attenuate LPS-induced mouse acute liver injury via the prostaglandin E 2-dependent repression of the NLRP3 inflammasome in Kupffer cells. Immunol Lett. 2016;179:102–13.
Krauss RM. Dietary and genetic probes of atherogenic dyslipidemia. Arterioscler Thromb Vasc Biol. 2005;25(11):2265–72.
Neuvonen PJ, Niemi M, Backman JT. Drug interactions with lipid-lowering drugs: mechanisms and clinical relevance. Clin Pharmacol Ther. 2006;80(6):565–81.
Armitage J. The safety of statins in clinical practice. Lancet. 2007;370(9601):1781–90.
Conforti A, Magro L, Moretti U, Scotto S, Motola D, Salvo F, et al. Fluvastatin and hepatic reactions: a signal from spontaneous reporting in Italy. Drug Saf. 2006;29(12):1163–72.
Darakhshan S, Bidmeshki Pour A, Hosseinzadeh Colagar A, Sisakhtnezhad S. Thymoquinone and its therapeutic potentials. Pharmacol Res. 2015;95–6:138 – 58.
Jaswal A, Sinha N, Bhadauria M, Shrivastava S, Shukla S. Therapeutic potential of thymoquinone against anti-tuberculosis drugs induced liver damage. Environ Toxicol Pharmacol. 2013;36(3):779–86.
Tobar Leitão SA, Soares DDS, Carvas Junior N, Zimmer R, Ludwig NF, Andrades M. Study of anesthetics for euthanasia in rats and mice: a systematic review and meta-analysis on the impact upon biological outcomes (SAFE-RM). Life Sci. 2021;284:119916.
Ragheb A, Elbarbry F, Prasad K, Mohamed A, Ahmed MS, Shoker A. Attenuation of the development of hypercholesterolemic atherosclerosis by thymoquinone. Int J Angiol. 2008;17(4):186–92.
Yuan L, Han X, Li W, Ren D, Yang X. Isoorientin prevents Hyperlipidemia and Liver Injury by regulating lipid metabolism, antioxidant capability, and Inflammatory Cytokine Release in High-Fructose-Fed mice. J Agric Food Chem. 2016;64(13):2682–9.
Nili-Ahmadabadi A, Alibolandi P, Ranjbar A, Mousavi L, Nili-Ahmadabadi H, Larki-Harchegani A, et al. Thymoquinone attenuates hepatotoxicity and oxidative damage caused by diazinon: an in vivo study. Res Pharm Sci. 2018;13(6):500–8.
de Winther MP, Hofker MH. Scavenging new insights into atherogenesis. J Clin Invest. 2000;105(8):1039–41.
Rudel LL, Lee RG, Parini P. ACAT2 is a target for treatment of coronary heart disease associated with hypercholesterolemia. Arterioscler Thromb Vasc Biol. 2005;25(6):1112–8.
Chen Y, He X, Yuan X, Hong J, Bhat O, Li G, et al. NLRP3 inflammasome formation and activation in nonalcoholic steatohepatitis: therapeutic target for Antimetabolic Syndrome Remedy FTZ. Oxid Med Cell Longev. 2018;2018:2901871.
Hendrikx T, Bieghs V, Walenbergh SM, van Gorp PJ, Verheyen F, Jeurissen ML, et al. Macrophage specific caspase-1/11 deficiency protects against cholesterol crystallization and hepatic inflammation in hyperlipidemic mice. PLoS ONE. 2013;8(12):e78792.
Liu H, Sun Y, Zhang Y, Yang G, Guo L, Zhao Y, et al. Role of Thymoquinone in Cardiac damage caused by Sepsis from BALB/c mice. Inflammation. 2019;42(2):516–25.
Periyanayagam S, Arumugam G, Ravikumar A, Ganesan VS. Thymoquinone ameliorates NLRP3-mediated inflammation in the pancreas of albino Wistar rats fed ethanol and high-fat diet. J Basic Clin Physiol Pharmacol. 2015;26(6):623–32.
Miao B, Degterev A. Targeting phospshatidylinositol 3-kinase signaling with novel phosphatidylinositol 3,4,5-triphosphate antagonists. Autophagy. 2011;7(6):650–1.
Reif S, Lang A, Lindquist JN, Yata Y, Gabele E, Scanga A, et al. The role of focal adhesion kinase-phosphatidylinositol 3-kinase-akt signaling in hepatic stellate cell proliferation and type I collagen expression. J Biol Chem. 2003;278(10):8083–90.
Hu C, Yang J, He Q, Luo Y, Chen Z, Yang L, et al. CysLTR1 blockage ameliorates Liver Injury caused by aluminum-overload via PI3K/AKT/mTOR-Mediated autophagy activation in vivo and in Vitro. Mol Pharm. 2018;15(5):1996–2006.
Ma J, Hu X, Li J, Wu D, Lan Q, Wang Q, et al. Enhancing conventional chemotherapy drug cisplatin-induced anti-tumor effects on human gastric cancer cells both in vitro and in vivo by Thymoquinone targeting PTEN gene. Oncotarget. 2017;8(49):85926–39.
Bai T, Yang Y, Wu YL, Jiang S, Lee JJ, Lian LH, et al. Thymoquinone alleviates thioacetamide-induced hepatic fibrosis and inflammation by activating LKB1-AMPK signaling pathway in mice. Int Immunopharmacol. 2014;19(2):351–7.
Bai T, Lian LH, Wu YL, Wan Y, Nan JX. Thymoquinone attenuates liver fibrosis via PI3K and TLR4 signaling pathways in activated hepatic stellate cells. Int Immunopharmacol. 2013;15(2):275–81.
Wang Y, Gao H, Zhang W, Zhang W, Fang L. Thymoquinone inhibits lipopolysaccharide-induced inflammatory mediators in BV2 microglial cells. Int Immunopharmacol. 2015;26(1):169–73.
Acknowledgements
Not applicable.
Funding
The study was supported by grants from China’s Dalian Medical Science Research Program (grant no. 1911107).
Author information
Authors and Affiliations
Contributions
**aoqing Zhang and Fei Wang designed this study; Wei Yao helped perform experiments; Yuhua Hao analyzed data and interpreted the results of experiments; Wei Yao prepared figures and drafted the manuscript; Yuhua Hao, Yuling Wu and **aoqing Zhang helped revise the manuscript; and Fei Wang provided the research funds. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All procedures were performed in accordance with the Declaration of Helsinki and complied with relevant guidelines and regulations. Approval was granted by the Ethical Committee of the Affiliated Zhongshan Hospital of the Dalian University of China (201902010).
Consent for publication
Not applicable.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Wang, F., Yao, W., Yu, D. et al. Protective role of thymoquinone in hyperlipidemia-induced liver injury in LDL-R−/−mice. BMC Gastroenterol 23, 276 (2023). https://doi.org/10.1186/s12876-023-02895-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12876-023-02895-0