Abstract
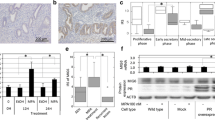
Adenomyosis is a fairly common gynecologic disease with unknown pathogenesis. We sought to investigate as to whether the promoter of progesterone receptor isoform B (PR-B) is hypermethylated in adenomyosis and to investigate the treatment of ectopic endometrial stromal cells with trichostatin A (TSA), a histone deacetylase inhibitor (HDI), and 5-aza-2-deoxycytidine (ADC), a demethylation agent, on PR-B gene and protein expression, and on cell viability. Ectopic endometrial tissue specimens were obtained from 9 women with adenomyosis whereas control endometrial tissue samples were obtained from 8 women with surgically diagnosed benign ovarian cysts but without any clinical history of endometriosis/adenomyosis/myoma. Endometrial stromal cells were isolated, purified, cultured, and analyzed by methylation-specific polymerase chain reaction (PCR), real-time reverse transcriptase PCR (RT-PCR), and Western blot analysis, cell viability assays, and fluorescence-activated cell sorting. We found that none of the normal endometrial stromal cells had PR-B promoter methylation. In contrast, 2 out of 3 ectopic endometrial stromall cells had PR-B hypermethylation (P <.05). The treatment with both TSA and ADC elevated PR-B gene and protein expression in ectopic, but not in normal, endometrial stromal cells. Both TSA and ADC treatment dose-dependently reduced cell viability of ectopic endometrial stromal cells. Trichostatin A and ADC treatment also suppressed the cell cycle progression in ectopic endometrial stromal cells. Thus, this study provides the first piece of evidence that adenomyosis has epigenetic aberration and may also be an epigenetic disease amenable to rectification by pharmacological means. This perspective may shed new light onto the pathogenesis of adenomyosis and lead to novel ways to treat the disease.
Similar content being viewed by others
References
Bergeron C, Amant F, Ferenczy A. Pathology and physiopathology of adenomyosis. Best Pract Res Clin Obstet Gynaecol. 2006;20(4): 511–521.
Farquhar C, Brosens I. Medical and surgical management of adenomyosis. Best Pract Res Clin Obstet Gynaecol. 2006;20(4): 603–616.
Kitawaki J. Adenomyosis: the pathophysiology of an oestrogen-dependent disease. Best Pract Res Clin Obstet Gynaecol. 2006;20(4):493–502.
Grow DR, Filer RB. Treatment of adenomyosis with long-term GnRH analogues: a case report. Obstet Gynecol. 1991;78(3 pt 2):538–539.
Cho S, Nam A, Kim H, et al. Clinical effects of the levonorgestrelreleasing intrauterine device in patients with adenomyosis. Am J Obstet Gynecol. 2008;198(4):373.e1–373.e7.
Ota H, Igarashi S, Hatazawa J, Tanaka T. Is adenomyosis an immune disease? Hum Reprod Update. 1998;4(4):360–367.
Maia H. Jr., Maltez A, Studart E, Athayde C, Coutinho EM. Effect of menstrual cycle and hormonal treatment on ki-67 and bcl-2 expression and adenomyosis. Gynecol Endocrinol. 2005;20(3): 127–131.
Kunz G, Beil D, Huppert P, Noe M, Kissler S, Leyendecker G. Adenomyosis in endometriosis-prevalence and impact on fertility. Evidence from magnetic resonance imaging. Hum Reprod. 2005; 20(8):2309–2316.
Kunz G, Herbertz M, Beil D, Huppert P, Leyendecker G. Adenomyosis as a disorder of the early and late human reproductive period. Reprod Biomed Online. 2007;15(6):681–685.
Wu MY, Ho HN. The role of cytokines in endometriosis. Am J Reprod Immunol. 2003;49(5):285–296.
Ulukus M, Ulukus EC, Tavmergen Goker EN, Tavmergen E, Zheng W, Arici A. Expression of interleukin-8 and monocyte chemotactic protein 1 in women with endometriosis. Fertil Steril. 2009;91(3):687–693.
Ota H, Igarashi S, Sasaki M, Tanaka T. Distribution of cyclooxygenase-2 in eutopic and ectopic endometrium in endometriosis and adenomyosis. Hum Reprod. 2001;16(3):561–566.
Ota H, Igarashi S, Hatazawa J, Tanaka T. Immunohistochemical assessment of superoxide dismutase expression in the endometrium in endometriosis and adenomyosis. Fertil Steril. 1999;72(1):129–134.
Ota H, Igarashi S, Hatazawa J, Tanaka T. Endothelial nitric oxide synthase in the endometrium during the menstrual cycle in patients with endometriosis and adenomyosis. Fertil Steril. 1998;69(2):303–308.
Ota H, Igarashi S, Kato N, Tanaka T. Aberrant expression of glutathione peroxidase in eutopic and ectopic endometrium in endometriosis and adenomyosis. Fertil Steril. 2000;74(2):313–318.
Ota H, Igarashi S, Tanaka T. Xanthine oxidase in eutopic and ectopic endometrium in endometriosis and adenomyosis. Fertil Steril. 2001;75(4):785–790.
Bulun SE, Cheng YH, Yin P, et al. Progesterone resistance in endometriosis: link to failure to metabolize estradiol. Mol Cell Endocrinol. 2006;248(1–2):94–103.
Guo SW. Nuclear factor-kappab (NF-kappaB): an unsuspected major culprit in the pathogenesis of endometriosis that is still at large? Gynecol Obstet Invest. 2007;63(2):71–97.
Hardy DB, Janowski BA, Corey DR, Mendelson CR. Progesterone receptor plays a major antiinflammatory role in human myometrial cells by antagonism of nuclear factor-kappaB activation of cyclooxygenase 2 expression. Mol Endocrinol. 2006;20(11): 2724–2733.
Kalkhoven E, Wissink S, van der Saag PT, van der Burg B. Negative interaction between the RelA(p65) subunit of NF-kappaB and the progesterone receptor. J Biol Chem. 1996;271(11): 6217–6224.
Yamamoto K, Arakawa T, Ueda N, Yamamoto S. Transcriptional roles of nuclear factor kappa B and nuclear factor-interleukin-6 in the tumor necrosis factor alpha-dependent induction of cyclooxygenase-2 in MC3T3-E1 cells. J Biol Chem. 1995; 270(52):31315–31320.
Nie J, Lu Y, Liu X., Guo SW. Immunoreactivity of progesterone receptor isoform B, nuclear factor kappaB, and IkappaBalpha in adenomyosis. Fertil Steril.2009;92(3):886–889.
Wu Y, Strawn E, Basir Z, Halverson G, Guo SW. Promoter hypermethylation of progesterone receptor isoform B (PR-B) in endometriosis. Epigenetics. 2006;1(2):106–111.
Ryan IP, Schriock ED, Taylor RN. Isolation, characterization, and comparison of human endometrial and endometriosis cells in vitro. J Clin Endocrinol Metab. 1994; 78(3):642–649.
Rossi MJ, Chegini N, Masterson BJ. Presence of epidermal growth factor, platelet-derived growth factor, and their receptors in human myometrial tissue and smooth muscle cells: their action in smooth muscle cells in vitro. Endocrinology. 1992;130(3): 1716–1727.
Hirota Y, Osuga Y, Hirata T, et al. Possible involvement of thrombin/protease-activated receptor 1 system in the pathogenesis of endometriosis. J Clin Endocrinol Metab. 2005;90(6): 3673–3679.
Liu Y, Peterson DA, Kimura H, Schubert D. Mechanism of cellular 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) reduction. J Neurochem. 1997; 69(2):581–593.
Serafim TL, Oliveira PJ, Sardao VA, Perkins E, Parke D, Holy J. Different concentrations of berberine result in distinct cellular localization patterns and cell cycle effects in a melanoma cell line. Cancer Chemother Pharmacol. 2008;61(6):1007–1018.
Sakaguchi H, Fujimoto J, Hong BL, Nakagawa Y, Tamaya T. Drastic decrease of progesterone receptor form B but not A mRNA reflects poor patient prognosis in endometrial cancers. Gynecol Oncol. 2004;93(2):394–399.
Sasaki M, Dharia A, Oh BR, Tanaka Y, Fujimoto S, Dahiya R. Progesterone receptor B gene inactivation and CpG hypermethylation in human uterine endometrial cancer. Cancer Res. 2001; 61(1): 97–102.
Tukey JW. Exploratory Data Analysis. Reading, MA: Addison-Wesley; 1977.
Inhaka R, Gentleman RR. R: a language for data analysis and graphics. J Comput Graph Stat. 1996;5(3):1923–1927.
Matalliotakis IM, Kourtis AI, Panidis DK. Adenomyosis. Obstet Gynecol Clin North Am. 2003;30(1):63–82, viii.
Devlieger R, D’Hooghe T, Timmerman D. Uterine adenomyosis in the infertility clinic. Hum Reprod Update. 2003;9(2):139–147.
Peric H, Fraser IS. The symptomatology of adenomyosis. Best Pract Res Clin Obstet Gynaecol. 2006;20(4):547–555.
Guo SW. Epigenetics of endometriosis. Mol Hum Reprod. 2009;15(10):587–607.
Wen DX, Xu YF, Mais DE, Goldman ME, McDonnell DP. The A and B isoforms of the human progesterone receptor operate through distinct signaling pathways within target cells. Mol Cell Biol. 1994;14(12):8356–8364.
Robertson KD, Wolffe AP. DNA methylation in health and disease. Nat Rev Genet. 2000;1(1):11–19.
Liu X, Guo SW. A pilot study on the off-label use of valproic acid to treat adenomyosis. Fertil Steril. 2008;89(1):246–250.
Matsuoka H, Fujimura T, Mori H, Aramori I, Mutoh S. Mechanism of HDAC inhibitor FR235222-mediated IL-2 transcriptional repression in Jurkat cells. Int Immunopharmacol. 2007;7(11): 1422–1432.
Yang AS, Estecio MR, Garcia-Manero G, Kantarjian HM, Issa JP. Comment on “Chromosomal instability and tumors promoted by DNA hypomethylation” and “Induction of tumors in nice by genomic hypomethylation.”. Science. 2003;302(5648): 1153;author reply 1153.
Lubbert M, Wijermans P, Kunzmann R, et al. Cytogenetic responses in high-risk myelodysplastic syndrome following low-dose treatment with the DNA methylation inhibitor 5-aza-2’-deoxycytidine. Br J Haematol. 2001;114(2):349–357.
Garcia-Manero M, Santana GT, Alcazar JL. Relationship between microvascular density and expression of vascular endothelial growth factor in patients with ovarian endometriosis. J Womens Health (Larchmt). 2008;17(5):777–782.
Wu Y, Starzinsky-Powitz A, Guo SW. Constitutive and TNFα-stimulated activation of nuclear factor κB (NF-κB) in immortalized endometriotic cells and their suppression by trichostatin A (TSA). Gynecol Obstet Invest. 2010;70(1):23–33.
Gonzalez-Ramos R, Donnez J, Defrere S, et al. Nuclear factor-kappa B is constitutively activated in peritoneal endometriosis. Mol Hum Reprod. 2007;13(7):503–509.
Liu XS, Yuan L, Guo SW. Vaproic acid as a therapy for adenomyosis: a comparative case series. Reprod Sci. IN PRESS.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nie, J., Liu, X. & Guo, SW. Promoter Hypermethylation of Progesterone Receptor Isoform B (PR-B) in Adenomyosis and Its Rectification by a Histone Deacetylase Inhibitor and a Demethylation Agent. Reprod. Sci. 17, 995–1005 (2010). https://doi.org/10.1177/1933719110377118
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719110377118