Abstract.
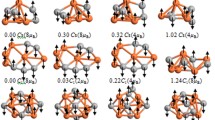
Using the density functional theory (DFT) method, the geometrical structures of \( {\rm In}_{m}X_{n}\) (\( X={\rm Se}\), Te; \( m+n=5\) clusters are optimized, and their relative stability as well as electronic, magnetic and spectral properties are calculated. The ground state structures of \( {\rm In}_{m}X_{n}\) clusters are found to be largely similar for \( X={\rm Se}\) and Te, with the exception of \( {\rm In}_{2}X_{3}\) . The energy gap exhibits the maximum for \( {\rm In}_{2}{\rm Se}_{3}\) or \( {\rm In}_{3}{\rm Te}_{2}\) . The electronic properties of \( {\rm In}_{m}X_{n}\) clusters depend on their geometrical structures and, hence, on the value of m ; and \( {\rm In}_{2}{\rm Se}_{3}\) shows a low vertical electron affinity (VEA) of about 1.60eV and a high vertical ionization potential (VIP) of about 9.33eV. The total magnetic moment is 1 or 0μ_B for the clusters with \( m={\rm odd}\) (1, 3) or even (2, 4) , respectively. The local magnetic moments of X atoms amount to about 99.9% of the total magnetic moment, while those of In atoms are merely 0.1%. The IR and Raman spectra of \( {\rm In}_{m}X_{n}\) clusters exhibit similarity for \( X={\rm Se}\) and Te with an exception of \( {\rm In}_{2}X_{3}\). The energies of the strongest peaks of \( {\rm In}_{m}{\rm Te}_{n}\) are largely smaller than the corresponding \( {\rm In}_{m}{\rm Se}_{n}\) in both IR and Raman spectra. For UV-Vis spectra, the absorption peaks at 200-400nm for all clusters and 390–780 nm for m = 1 and 3 (except \( {\rm In}_{3}{\rm Te}_{2}\) are likely to hint useful properties of UV and visible light absorption, respectively.
Similar content being viewed by others
References
A.J. Hoffman, G. Mills, H. Yee et al., J. Phys. Chem. 96, 5546 (1992)
S. Rasool, K. Saritha, K.T. Ramakrishna Reddy et al., Eur. Phys. J. Plus 132, 451 (2017)
E. Cochran, Sci. Amer. 263, 74 (1990)
D.A. Bandurin, A.V. Tyurnina, G.L. Yu et al., Nat. Nanotechnol. 12, 223 (2017)
K. Xu, L. Yin, Y. Huang et al., Nanoscale 8, 16802 (2016)
L. Debbichi, O. Eriksson, S. Lebègue, J. Phys. Chem. Lett. 6, 3098 (2015)
J.M. Maxtxain, J.E. Fowler, J.M. Ugalde, Phys. Rev. A 61, 532 (2000)
J.J. BelBruno, E. Sanville, A. Burnin et al., Chem. Phys. Lett. 478, 132 (2009)
M. Sierka, J. Dbler, J. Sauer et al., Angew. Chem. Int. Ed. 46, 3372 (2007)
J. Sun, W.C. Lu, H. Wang et al., Int. J. Quantum Chem. 107, 1915 (2007)
A.B.C. Patzer, C. Chang, E. Sedlmayr et al., Eur. Phys. J. D 32, 329 (2005)
D. van Heijnsbergen, K. Demyk, M.A. Duncan, G. Meijer et al., Phys. Chem. Chem. Phys. 5, 2515 (2003)
J.X. Guo, S.Y. Wu, L. Peng et al., Physica B 524, 1 (2017)
H. **, J.W. Li, Ying Dai et al., Phys. Chem. Chem. Phys. 19, 4855 (2017)
E.M. Sosa-Hernande, J.M. Montejano-Carrizales, P.G. Alvarado-Leyva, Eur. Phys. J. D 71, 284 (2017)
Z.C. Yu, X.R. Zhang, P.Y. Huo et al., Eur. Phys. J. Plus 132, 451 (2017)
Madhulata Shukla, Indrajit Sinha, Int. J. Quantum. Chem. 118, 25490 (2018)
K.S. Novoselov, D. Jiang, F. Schedin et al., PNAS 102, 10451 (2005)
D. Die, B.X. Zheng, L.Q. Zhao et al., Sci. Rep. 6, 31978 (2016)
C.G. Lia, J. Zhang, Y.Q. Yuan et al., Physica E 86, 303 (2017)
Y.F. Li, X.Y. Kuang, S.J. Wang et al., Phys. Lett. A 375, 1877 (2011)
S. Erkoc, S. Katirciogaelu, T. Yilmaz, J. Mol. Struct. Theochem. 542, 101 (2001)
Ralph G. Pearson, J. Am. Chem. Soc. 107, 6801 (1985)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhong, SY., Wu, SY., Zhang, F. et al. First-principles study of In mXn (X = Se, Te; m + n = 5 clusters: Structural, electronic, magnetic and spectral properties. Eur. Phys. J. Plus 133, 208 (2018). https://doi.org/10.1140/epjp/i2018-12036-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1140/epjp/i2018-12036-8