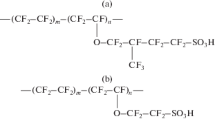Abstract
The paper presents the results of a study of water uptake, ionic conductivity, and Donnan potential in systems with perfluorosulfonic acid membranes in the H+, Li+, Na+, and K+ ionic forms and solutions of inorganic electrolytes. The properties of commercial membranes Aquivion E87-05S and Nafion 212, as well as membranes obtained from dispersions of Nafion 212 in solvents of various nature (N,N-dimethylformamide, 1-methyl-2-pyrrolidone, mixtures of isopropyl alcohol with water in a volume ratio of 80–20) have been studied. The effect of the number of functional groups, the length of the side chain of polymer macromolecules, and the morphology of the polymer in membranes on their equilibrium and transport properties depending on the nature of the counterion has been determined. The effect of relaxation and electrophoretic factors on the transfer of alkali metal ions through the system of pores and channels of perfluorosulfonic acid membranes is discussed. The slope of the concentration dependences of the Donnan potential for all highly hydrated membranes in the H+ form has been found to be close to the Nernstian one, while the selectivity to alkali metal ions increases for membranes with the highest ion exchange capacity or the lowest amount of sorbed water and diffusion permeability due to the exclusion of co-ions from the membrane phase.





Similar content being viewed by others
REFERENCES
A. Yu. Alent’ev, A. V. Volkov, I. V. Vorotyntsev, A. L. Maksimov, and A. B. Yaroslavtsev, Membr. Membr. Technol. 5, 255 (2021). https://doi.org/10.1134/S2517751621050024
A. Kusoglu and A. Z. Weber, Chem. Rev. 117, 987 (2017). https://doi.org/10.1021/acs.chemrev.6b00159
M. A. Yandrasits, M. J. Lindell, and S. J. Hamrock, Curr. Opin. Electrochem. 18, 90 (2019). https://doi.org/10.1016/j.coelec.2019.10.012
A. Makhsoos, M. Kandidayeni, B. G. Pollet, and L. Boulon, Int. J. Hydrogen Energy 48, 15341 (2023). https://doi.org/10.1016/j.ijhydene.2023.01.048
T. K. Maiti, J. Singh, P. Dixit, J. Majhi, S. Bhushan, A. Bandyopadhyay, and S. Chattopadhyay, Chem. Eng. J. Adv. 12, 100372 (2022). https://doi.org/10.1016/j.ceja.2022.100372
I. Falina, N. Loza, S. Loza, E. Titskaya, and N. Romanyuk, Membranes 11, 227 (2021). https://doi.org/10.3390/membranes11030227
D. Yu. Voropaeva, E. Yu. Safronova, S. A. Novikova, and A. B. Yaroslavtsev, Mendeleev Commun. 32, 287 (2022). https://doi.org/10.1016/J.MENCOM.2022.05.001
D. Yu. Voropaeva and A. B. Yaroslavtsev, Membr. Membr. Technol. 4, 276 (2022). https://doi.org/10.1134/S2517751622040102
A. Parasuraman, T. M. Lim, C. Menictas, and M. Skyllas-Kazacos, Electrochim. Acta 101, 27 (2013). https://doi.org/10.1016/J.ELECTACTA.2012.09.067
H. B. Noh, M. S. Won, and Y. B. Shim, Biosensors Bioelectron. 61, 554 (2014). https://doi.org/10.1016/J.BIOS.2014.06.002
L. Yu, Q. Zhang, B. Yang, Q. Xu, Q. Xu, and X. Hu, Sens. Actuators 259, 540 (2018). https://doi.org/10.1016/J.SNB.2017.12.103
E. Maciak, Sensors 19, 629 (2019). https://doi.org/10.3390/s19030629
I. A. Prikhno, E. Yu. Safronova, I. A. Stenina, P. A. Yurova, and A. B. Yaroslavtsev, Membr. Membr. Technol. 2, 265 (2020). https://doi.org/10.1134/S2517751620040095
G. Zhang, G. Yang, Q. Shen, S. Li, Z. Li, J. Liao, Z. Jiang, H. Wang, H. Zhang, and W. Ye, J. Power Sources 542, 231740 (2022). https://doi.org/10.1016/j.jpowsour.2022.231740
T. Okada, S. Moller-Holst, O. Gorseth, and S. Kjelstrup, J. Electroanal. Chem. 442, 137 (1998). https://doi.org/10.1016/S0022-0728(97)00499-3
C. C. Tai, C. L. Chen, and C. W. Liu, Int. J. Hydrog. Energy 42, 3981 (2017). https://doi.org/10.1016/j.ijhydene.2016.11.047
W. M. Yan, H. S. Chu, Y. L. Liu, F. Chen, and J. H. Jang, Int. J. Hydrog. Energy 36, 5435 (2011). https://doi.org/10.1016/j.ijhydene.2011.01.158
P. Yu. Apel’, S. Velizarov, A. V. Volkov, T. V. Eliseeva, V. V. Nikonenko, A. V. Parshina, N. D. Pis’menskaya, K. I. Popov, and A. B. Yaroslavtsev, Membr. Membr. Technol. 4, 69 (2022). https://doi.org/10.1134/S2517751622020032
M. Vijayakumar, M. S. Bhuvaneswari, P. Nachimuthu, B. Schwenzer, S. Kim, Z. Yang, Z. Liu, G. L. Graff, S. Thevuthasan, and J. Hu, J. Membr. Sci. 366, 325 (2011). https://doi.org/10.1016/j.memsci.2010.10.01
M. Vijayakumar, S. D. Burton, C. Huang, L. Li, Z. Yang, G. L. Graff, J. Liu, J. Hu, and M. Skyllas-Kazacos, J. Power Sources 95, 709 (2010). https://doi.org/10.1016/j.jpowsour.2010.05.008
A. R. Crothers, C. J. Radke, and A. Z. Weber, J. Phys. Chem. C 121, 28262 (2017). https://doi.org/10.1021/acs.jpcc.7b07360
T. Mabuchi and T. Tokumasu, J. Phys. Chem. B 122, 5922 (2018). https://doi.org/10.1021/acs.jpcb.8b02318
S. Shi, A. Z. Weber, and A. Kusoglu, Electrochim. Acta 220, 517 (2016). https://doi.org/10.1016/J.ELECTACTA.2016.10.096
L. Wu, T. Luo, X. Yang, H. Zhao, X. Wang, and Z. Zhang, Sep. Purif. Technol. 316, 123816 (2023). https://doi.org/10.1016/j.seppur.2023.123816
X. T. Le, J. Membr. Sci. 3978, 66 (2012). https://doi.org/10.1016/j.memsci.2012.01.011
D. E. Curtin, R. D. Lousenberg, T. J. Henry, P. C. Tangeman, and M. E. Tisack, J. Powed Sources 131, 41 (2004). www.sciencedirect.com/science/article/pii/ S0378775304000175.
A. V. Parshina, E. A. Ryzhkova, E. Yu. Safronova, D. V. Safronov, A. A. Lysova, O. V. Bobreshova, and A. B. Yaroslavtsev, Pet. Chem. 55, 816 (2015). https://doi.org/10.1134/S0965544115100175
S. Subianto, M. Pica, M. Casciola, P. Cojocaru, L. Merlo, G. Hards, and D. J. Jones, J. Power Sources 233, 216 (2013). https://doi.org/10.1016/j.jpowsour.2012.12.121
M. Yue, S. Jemei, N. Zerhouni, and R. Gouriveau, Renew. Energy 179, 2277 (2021). https://doi.org/10.1016/j.renene.2021.08.045
R. B. Moore and C. R. Martin, Anal. Chem. 58, 2569 (1986). https://doi.org/10.1021/ac00125a046
C. Welch, A. Labouriau, R. Hjelm, B. Orler, C. Johnston, and Y. S. Kim, ACS Macro Lett. 1, 1403 (2012). https://doi.org/10.1021/mz3005204
B. Loppinet, G. Gebel, and C. E. Williams, J. Phys. Chem. B 101, 1884 (1997). https://doi.org/10.1021/jp9623047
S. A. Berlinger, P. J. Dudenas, A. Bird, X. Chen, G. Freychet, B. D. McCloskey, A. Kusoglu, and A. Z. Weber, ACS Appl. Polym. Mater. 2, 5824 (2020). https://doi.org/10.1021/acsapm.0c01076
F. M. Collette, F. Thominette, H. Mendil-Jakani, and G. Gebel, J. Membr. Sci. 435, 242 (2013). https://doi.org/10.1016/j.memsci.2013.02.002
A. Tarokh, K. Karan, S. Ponnurangam, and M. D. Ato-mistic, Macromolecules 53, 288 (2020). https://doi.org/10.1021/acs.macromol.9b01663
H.-L. Lin, T. L. Yu, C.-H. Huang, and T.-L. Lin, J. Polym. Sci. B 43, 3044 (2005). https://doi.org/10.1002/polb.20599
Y. S. Kim, C. F. Welch, R. P. Hjelm, N. H. Mack, A. Labouriau, and E. B. Orler, Macromolecules 48, 2161 (2015). https://doi.org/10.1021/ma502538k
E. Yu. Safronova, D. Yu. Voropaeva, D. V. Safronov, N. Stretton, A. V. Parshina, and A. B. Yaroslavtsev, Membranes 13, 13 (2023). https://doi.org/10.3390/MEMBRANES13010013
A. Skulimowska, M. Dupont, M. Zaton, S. Sunde, L. Merlo, D. J. Jones, and J. Rozière, Int. J. Hydrogen Energy 39, 6307 (2014). https://doi.org/10.1016/j.ijhydene.2014.02.082
A. Parshina, E. Safronova, A. Osipov, E. Lapshina, A. Yelnikova, O. Bobreshova, and A. Yaroslavtsev, Membranes 9, 142 (2019). https://doi.org/10.3390/membranes9110142
I. A. Stenina and A. B. Yaroslavtsev, Membranes 11, 198 (2021). https://doi.org/10.3390/membranes11030198
I. A. Stenina and A. B. Yaroslavtsev, Inorg. Mater. 53, 253 (2017). https://doi.org/10.1134/S0020168517030104
G. Suresh, Y. Scindia, A. Pandey, and A. Goswami, J. Membr. Sci. 250, 39 (2005). https://doi.org/10.1016/j.memsci.2004.10.013
N. Agmon, Chem. Phys. Lett. 244, 456 (1995). https://doi.org/10.1016/0009-2614(95)00905-J
M. Legras, Y. Hirata, Q. T. Nguyen, D. Langevin, and M. Métayer, Desalination 147, 351 (2002). https://doi.org/10.1016/S0011-9164(02)00608-2
E. Yu. Safronova, A. K. Osipov, and A. B. Yaroslavtsev, Pet. Chem. 58, 130 (2018). https://doi.org/10.1134/S0965544118020044
Funding
The study was supported the Russian Science Foundation, grant no. 21-73-10149, https://rscf.ru/project/21-73-10149.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by V. Avdeeva
Rights and permissions
About this article
Cite this article
Parshina, A.V., Safronova, E.Y., Yelnikova, A.S. et al. Effect of the Nature of Counterion on Properties of Perfluorosulfonic Acid Membranes with Long and Short Side Chains. Membr. Membr. Technol. 5, 323–332 (2023). https://doi.org/10.1134/S2517751623050062
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2517751623050062