Abstract
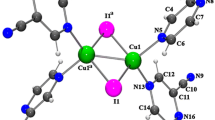
Synthesis and single crystal X-ray diffraction study were carried for compound {[Cu(C11H16N4)2(N3)](ClO4)} · H2O (I). The structure is molecular, and the Cu2+ ion is in a five-coordinated compressed trigonal bipyramid environment. Copper ion is bound to five N atoms, in which four N atoms are from two chlelating ligands 2-(piperidin-1-ylmethyl)pyridine and the fifth N donor is from a monodentate azido ligand. The complex cations [Cu(C11H16N4)2(N3)]+, the perchlorate anions, and solvent water molecules are further joined into three dimensional supramolecular networks by rich hydrogen bonds including strong O-H…N between solvent water and azide ion and O-H…O between solvent water and perchlorate ion, and weak hydrogen bonds C-H…O, and weak bifurcated hydrogen bonds C-H/C-H…N in which N atom of azide ion serving as bifurcated acceptor and two C-H groups as donors.
Similar content being viewed by others
References
Schubert, U.S. and Eschbaumer, C., Angew. Chem. Int. Ed., 2002, vol. 41, p. 2892.
Zhou, M. and Roovers, J., Macromolecules, 2001, vol. 34, p. 244.
Oh, M. and Mirkin, C.A., Nature, 2005, vol. 438, p. 651.
Oh, M. and Mirkin, C.A., Angew. Chem. Int. Ed., 2006, vol. 45, p. 5492.
Rakitin, Y.V. and Kalinnikov, V.T., Russ. Chem. Bull. Int. Ed., 2004, vol. 53, p. 766.
Halcrow, M.A., Huffman, J.C., and Christou, G., Angew. Chem. Int. Ed., 1995, vol. 8, p. 889.
Ribas, J., Escuer, A., Monfort, M., et al., Coord. Chem. Rev., 1999, vol. 193–195, p. 1027.
Bai, S.Q., Fang, C.J., and Yan, C.H., Chin. J. Inorg. Chem., 2006, vol. 12, p. 2123.
Sheldrick G.M., SHELX-97, Release 97-1, Göttingen (Germany): Univ. of Göttingen, 1997.
Aronica, C., Jeanneau, E., El Moll, H., et al., Chem. Eur. J., 2007, vol. 13, p. 3666.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Wang, C.F., Zhang, Z.L., Wang, Y.L. et al. A novel azide copper complex: [[Azido{bis[2-(piperidin-1-ylmethyl)pyridine]}copper(II)]perchlorate] hydrate. Russ J Coord Chem 35, 789–792 (2009). https://doi.org/10.1134/S1070328409100145
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328409100145