Abstract
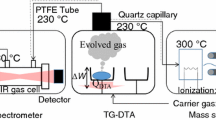
Thermal decomposition of UO2(NO3)2·6H2O to U3O8 was studied by thermogravimetric (TG), differential thermogravimetric (DTG), and differential thermal (DTA) analyses in combination with gaschromatographic and chemical methods. The process is described by kinetic data recorded under the non-isothermal conditions. UO2(NO3)2·6H2O transforms into UO3 in eight main stages in the 40–300°C range, and UO3 into U3O8, in three stages with the peaks of weight loss at 580, 620, and 645°C.
Similar content being viewed by others
References
Dawson, J.K., Wait, E., Alcock, K., and Chilton, D.R., J. Chem. Soc., 1956, no. 9, pp. 3531–3540.
Ondreijcik, R.R., and Garrett, T.P., J. Phys. Chem., 1961, vol. 65, no. 3, pp. 470–473.
Lodding, W. and Ojamao, L., J. Inorg. Nucl. Chem., 1965, vol. 27, no. 6, pp. 1261–1268.
Hartland, S. and Nesbitt, R.J., J. Appl. Chem., 1964, vol. 14, p. 406.
Smith, W.H., J. Inorg. Nucl. Chem., 1968, vol. 30, no. 7, pp. 1761–1768.
Vdovenko, V.M. and Sokolov, A.P., Radiokhimiya, 1959, vol. 1, no. 2, p. 178.
Author information
Authors and Affiliations
Additional information
Original Russian Text © R.D. Kozlova, V.A. Matyukha, N.V. Dedov, 2007, published in Radiokhimiya, 2007, Vol. 49, No. 2, pp. 116–119.
Rights and permissions
About this article
Cite this article
Kozlova, R.D., Matyukha, V.A. & Dedov, N.V. Mechanism and kinetics of thermal decomposition of uranyl nitrate hexahydrate under the nonisothermal conditions. Radiochemistry 49, 130–134 (2007). https://doi.org/10.1134/S1066362207020063
Received:
Issue Date:
DOI: https://doi.org/10.1134/S1066362207020063