Abstract
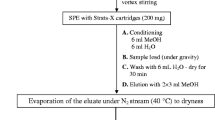
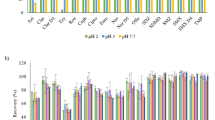
An approach to the highly sensitive and selective determination of the bromine-containing antiviral drug umifenovir (Arbidol) and its metabolites in natural and waste water, activated sludge, and bottom sediments by chromatography-mass spectrometry based on a combination of two techniques– inductively coupled plasma mass spectrometry (ICP MS) and electrospray ionization high-resolution mass spectrometry (ESI HRMS) is developed. Reversed-phase chromatographic separation and detection based on ESI HRMS provide the reliable detection and identification of analytes in complex matrices, while the use of ICP-MS with 79Br signal detection makes it possible to exclude matrix effects and use a single analytical standard for quantitative analysis. The use of solid-phase extraction and pressurized liquid extraction as sample preparation methods made it possible to achieve limits of detection at a level of 0.2 ng/L and 2 µg/kg for liquid and solid samples, respectively. The developed approach was successfully tested in the analysis of real samples. It is shown that the concentrations of analytes in urban wastewaters are in the range 4.4–260 ng/L. The highest concentration (up to 3.7 mg/kg) is characteristic of activated sludge, which acts as an effective sorbent for umifenovir and its transformation products.



Similar content being viewed by others
REFERENCES
Suda, K.J., Hunkler, R.J., Matusiak, L.M., and Schumock, G.T., Pharmacotherapy, 2015, vol. 35, no. 11, p. 991. https://doi.org/10.1002/phar.1656
Trofimov, F.A., Tsyshkova, N.G., Zotova, S.A., and Grinev, A.N., Pharm. Chem. J., 1993, vol. 27, no. 1, p. 75. https://doi.org/10.1007/BF00772858
Pecheur, E.I., Borisevich, V., Halfmann, P., Morrey, J.D., Smee, D.F., Prichard, M., Mire, C.E., Kawaoka, Y., Geisbert, T.W., and Polyak, S.J., J. Virol., 2016, vol. 90, no. 6, p. 3086. https://doi.org/10.1128/JVI.02077-15
Instructions for the medical use of the Arbidol preparation. https://arbidol.ru/files/arbidol_maksimum_kapsuly_200_mg.pdf?v=20210224. Accessed April 8, 2023.
Kuroda, K., Li, C., Dhangar, K., and Kumar, M., Sci. Total Environ., 2021, vol. 776, p. 145740. https://doi.org/10.1016/j.scitotenv.2021.145740
Ul’yanovskii, N.V., Kosyakov, D.S., Sypalov, S.A., Varsegov, I.S., Shavrina, I.S., and Lebedev, A.T., Sci. Total Environ., 2022, vol. 805, p. 150380. https://doi.org/10.1016/j.scitotenv.2021.150380
Proskurnina, E.V., Izmailov, D.Y., Sozarukova, M.M., Zhuravleva, T.A., Leneva, I.A., and Poromov, A.A., Molecules, 2020, vol. 25, no. 7, p. 1577. https://doi.org/10.3390/molecules25071577
Muller, A.L., Mello, P.A., Mesko, M.F., Duarte, F.A., Dressler, V.L., Muller, E.I., and Flores, E.M., J. Anal. At. Spectrom., 2012, vol. 27, no. 11, p. 1889. https://doi.org/10.1039/C2JA30212H
**e, R., Johnson, W., Spayd, S., Hall, G.S., and Buckley, B., Anal. Chim. Acta, 2006, vol. 578, no. 2, p. 186. https://doi.org/10.1016/j.aca.2006.06.076
de Souza, S.S., Campiglia, A.D., and Barbosa, F., Jr., Anal. Chim. Acta, 2013, vol. 761, p. 11. https://doi.org/10.1016/j.aca.2012.11.038
McSheehy, S., Pohl, P., Lobinski, R., and Szpunar, J., Analyst, 2001, vol. 126, no. 7, p. 1055. https://doi.org/10.1039/B102225N
Bierla, K., Dernovics, M., Vacchina, V., Szpunar, J., Bertin, G., and Lobinski, R., Anal. Bioanal. Chem., 2008, vol. 390, p. 1789. https://doi.org/10.1007/s00216-008-1883-5
Klencsar, B., Li, S., Balcaen, L., and Vanhaecke, F., TrAC, Trends Anal. Chem., 2018, vol. 104, p. 118. https://doi.org/10.1016/j.trac.2017.09.020
Liu, D., Chen, G., Huo, Z., Liu, H., Ji, W., and Liu, H., Chromatographia, 2019, vol. 82, p. 927. https://doi.org/10.1007/s10337-019-03730-z
Brauer, G., Handbook of Preparative Inorganic Chemistry, New York: Academic, 1993.
Lyon, T.D.B., Robin, P.A., Watson, W.S., and Littlejohn, D., J. Anal. At. Spectrom., 2005, vol. 20, no. 8, p. 757. https://doi.org/10.1039/B417529H
Kosyakov, D.S., Ul’yanovskii, N.V., Popov, M.S., Latkin, T.B., and Lebedev, A.T., Water Res., 2017, vol. 127, p. 183. https://doi.org/10.1016/j.watres.2017.10.008
Funding
The work was performed using the equipment of the Core Facility Center “Arktika” of the M.V. Lomonosov Northern (Arctic) Federal University with the financial support of the Russian Science Foundation, project no. 21-13-00377.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by V. Kudrinskaya
Rights and permissions
About this article
Cite this article
Sypalov, S.A., Ul’yanovskii, N.V., Kosyakov, D.S. et al. Determination of Umifenovir and Its Metabolites by High-Performance Liquid Chromatography with Combined Mass Spectrometric Detection. J Anal Chem 78, 1191–1198 (2023). https://doi.org/10.1134/S1061934823090150
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061934823090150