Abstract
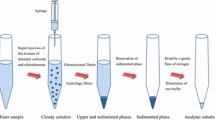
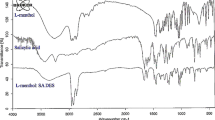
This research presents the use of deep eutectic solvents as a green and cost-effective extraction solvent in dispersive liquid–liquid microextraction of cadmium in food and environmental water specimen, where sodium diethyldithiocarbamate was employed as a complexing agent. A flame atomic absorption spectrometer was then used to quantify cadmium. The impacts of some effective variables such as deep eutectic solvent type and volume, pH, disperser solvent type and volume, diethyldithiocarbamate concentration, and extraction time were examined and optimized. Under optimal conditions, limit of detection was measured as 0.9 μg/L, while limit of quantification, enrichment factor, and relative standard deviation were 2.9 μg/L, 50, and 1.4%, respectively. The developed extraction method was successfully employed for Cd quantification in food and water. Recoveries of spiked samples varied in the range of 96.0–102.5%.








Similar content being viewed by others
REFERENCES
Nordberg, G.F., BioMetals, 2004, vol. 17, no. 5, p. 485.
Baldantoni, D., Morra, L., Zaccardelli, M., and Al-fani, A., Ecotoxicol. Environ. Saf., 2016, vol. 123, p. 89.
Khan, S.I., Ahmed, A.K.M., and Yunus, M., J. Health Pop. Nutr., 2010, vol. 28, no. 6, p. 578.
Moore, J.W., Inorganic Contaminants of Surface Water: Research and Monitoring Priorities, New York: Springer, 2009.
Drinking Water Contaminants—Standards and Regulations, United States Environmental Protection Agency. http://water.-epa.gov/drink/contaminants/basicinformation/historical/ upload/Archived-Consumer-Factsheet-on-Cadmium.pdf.
Anthemidis, A.N., Zachariadis, G.A., Farastelis, C.G., and Stratis, J.A., Talanta, 2004, vol. 62, no. 3, p. 437.
Vanderpool, R.A. and Buckley, W.T., Anal. Chem., 1999, vol. 71, no. 3, p. 652.
Atsumi, K., Minami, T., and Ueda, J., Anal. Sci., 2005, vol. 21, no. 6, p. 647.
Jalbani, N. and Soylak, M., Ecotoxicol. Environ. Saf., 2014, vol. 102, p. 174.
Krawczyk, M. and Jeszka-Skowron, M., Microchem. J., 2016, vol. 126, p. 296.
Li, Z., Chen, J., Liu, M., and Yang, Y., Anal. Methods, 2014, vol. 6, no. 10, p. 3241.
Ning, J., Jiao, Y., Zhao, J., Meng, L., and Yang, Y., Water Sci. Technol., 2014, vol. 70, no. 4, p. 605.
Zeini Jahromi, E., Bidari, A., Assadi, Y., Milani Hosseini, M.R., and Jamali, M.R., Anal. Chim. Acta, 2007, vol. 585, no. 2, p. 305.
Rajabi, M., Kamalabadi, M., Jamali, M.R., Zolgharnein, J., and Asanjarani N., Hum. Exp. Toxicol., 2013, vol. 32, no. 6, p. 620.
Shirkhanloo, H., Ghazaghi, M., and Zavvar Mousavi, H., J. Mol. Liq., 2016, vol. 218, p. 478.
Pena-Pereira, F. and Namieśnik, J., ChemSusChem, 2014, vol. 7, no. 7, p. 1784.
Zhang, Q., Vigier, K.D.O., Royer, S., and Jerome, F., Chem. Soc. Rev., 2012, vol. 41, no. 21, p. 7108.
Tang, B. and Row, K.H., Monatsh. Chem., 2013, vol. 144, no. 10, p. 1427.
Smith, E.L., Abbott, A.P., and Ryder, K.S., Chem. Rev., 2014, vol. 114, no. 21, p. 11060.
Tang, B., Zhang, H., and Row, K.H., J. Sep. Sci., 2015, vol. 38, no. 6, p. 1053.
Arain, M.B., Yilmaz, E., and Soylak, M., J. Mol. Liq., 2016, vol. 224, p. 538.
Yilmaz, E. and Soylak, M., Talanta, 2016, vol. 160, p. 680.
Es’haghi, Z., Khalili, M., Khazaeifar, A., and Rounaghi, G.H., Electrochim. Acta, 2011, vol. 56, no. 9, p. 3139.
Florindo, C., Branco, L.C., and Marrucho, I.M., Fluid Phase Equilibr., 2017, vol. 448, p. 135.
Farajzadeh, M.A., Afshar Mogaddam, M.R., and Aghanassa, M., Anal. Methods, 2016, vol. 8, no. 12, p. 2576.
Chamsaz, M., Atarodi, A., Efekhari, M., Asadpour, S., and Adibi, M., J. Adv. Res., 2013, vol. 4, no. 1, p. 35.
Ma, J.J., Du, X., Zhang, J.W., Li, J.C., and Wang, L.Z., Talanta, 2009, vol. 80, no. 2, p. 980.
Sitko, R., Gliwinska, B., Zawisza, B., and Feist, B., J. Anal. Atom. Spectrom., 2013, vol. 28, no. 3, p. 405.
Meng, L., Ning, J., and Yang, Y., Water Sci. Technol., 2014, vol. 70, no. 12, p. 1913.
Bosch Ojeda, C., Sánchez Rojas, F., and Cano Pavón, J.M., Am. J. Anal. Chem., 2010, vol. 1, no. 3, p. 127.
ACKNOWLEDGMENTS
The author gratefully appreciates the support provided by Payame Noor University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Jamali, M., Nakhaei, J.M., Sohrabnezhad, S. et al. A Green-Solvent Based Dispersive Liquid–Liquid Microextraction for Preconcentration and Cadmium Determination in Water and Food Samples by Flame Atomic Absorption Spectrometry. J Anal Chem 78, 980–987 (2023). https://doi.org/10.1134/S1061934823080075
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061934823080075