Abstract
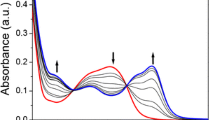
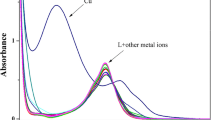
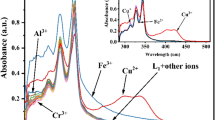
A ruthenium-bipyridine based fluorescent chemosensor was employed for the fluorometric and colorimetric Cu(II) detection in 100% aqueous medium. The successive emission quenching of the chemosensor by Cu(II) produced quenching constant (KSV) of 1.85 × 105 M–1. Binding constant (Kb) and the detection limit values for Cu2+ ion by absorption titration were calculated as 9.87 × 106 M–1 and 0.6 µM, respectively. A competitive binding method was used to investigate selectivity toward Cu ion in the presence of other metal ions. Reversibility studies were performed by adding an excess amount of sulfide ion. Reversible cycles and corresponding logic gate were also generated by absorption change. Binding of the chemosensor to Cu2+ occurs in a 1 : 1 stoichiometry using Job’s analysis. Cu(II) binding with the chemosensor was established by spectroscopic methods.








Similar content being viewed by others
REFERENCES
Festa, R.A. and Thiele, D.J., Curr. Biol., 2011, vol. 21, p. R877.
Que, E.L., Domaille, D.W., and Chang, C.J., Chem. Rev., 2008, vol. 5, p. 517.
Wu, D., Sedgwick, A.C., Gunnlaugsson, T., Akkaya, E.U., Yoon, J.T., and James, D., Chem. Soc. Rev., 2017, vol. 46, p. 7105.
Strausak, D., Mercer, J.F.B., Dieter, H.H., Stremmel, W., and Multhaup, G., Brain Res. Bull., 2001, vol. 55, p. 75.
Gupte, A. and Mumper, R.J., Cancer Treat. Rev., 2009, vol. 35, p. 32.
Waggoner, D.J., Bartnikas, T.B., and Gitlin, J.D., Neurobiol. Dis., 1999, vol. 6, p. 22.
Liu, C., Bai, R., and San, L.Q., Water Res., 2008, vol. 42, p. 511.
Acilan, C., Adiguzel, Z., Cevatemre, B., Karakas, D., Ulukaya, E., Ribeiro, N., Correia, I., and Pessoa, J.C., Biochim. Biophys. Acta, Gen. Subj., 2017, vol. 1861, p. 218.
Pourreza, N. and Hoveizavi, R., Anal. Chim. Acta, 2005, vol. 549, p. 124.
Giakisikli, G., Zachariadis, P., Kila, I., Teshima, N., and Anthemidis, A., Anal. Lett., 2016, vol. 49, p. 929.
Ji, L., Cheng, Q., Wu, K., and Yang, X., Sens. Actuators, B, 2016, vol. 231, p. 12.
Lilja, C., Betova, I., and Bo**ov, M., Electrochim. Acta, 2016, vol. 202, p. 333.
Zoriy, M.V. and Becker, J.S., Rapid Commun. Mass Spectrom., 2009, vol. 23, p. 23.
Becker, J.S., Zoriy, M.V., Pickhardt, C., Palomero-Gallagher, N., and Zilles, K., Anal. Chem., 2005, vol. 77, p. 3208.
Carter, K.P., Young, A.M., and Palmer, A.E., Chem. Rev., 2014, vol. 114, p. 4564.
Kumar, P., Kaur, S., Gupta, R., and Bowman-James, K., Pincers based on dicarboxamide and dithiocarboxamide functional groups, in: Pincer Compounds: Chemistry and Applications, Morales-Morales, D., Ed., Amsterdam: Elsevier, 2018, Ch. 14, p. 295.
Liu, Z., He, W., and Guo, Z., Chem. Soc. Rev., 2013, vol. 42, p. 1568.
Kumar, P., Kumar, V., and Gupta, R., Dalton Trans., 2020, vol. 49, p. 9544.
Cotruvo, J.A., Jr., Aron, A.T., Ramos-Torres, K.M., and Chang, C.J., Chem. Soc. Rev., 2015, vol. 44, p. 4400.
Zheng, Z.-B., Kang, S.-Y., Zhao, Y., Zhang, N., Yi, X., and Wang, K.-Z., Sens. Actuators, B, 2015, vol. 221, p. 614.
Lee, Y.H., Park, N., Park, Y.B., Hwang, Y.J., Kang, C., and Kim, J.S., Chem. Commun., 2014, vol. 50, p. 3197.
Kumar, V., Kumar, P., and Gupta, R., RSC Adv., 2017, vol. 7, p. 23127.
You, Y., Han, Y., Lee, Y.-M., Park, S.Y., Nam, W., and Lippard, S.J., J. Am. Chem. Soc., 2011, vol. 133, p. 11488.
Li, M., Liang, Q., Zheng, M., Fang, C., Peng, S., and Zhao, M., Dalton Trans., 2013, vol. 42, p. 13509.
Zhao, Q., Li, F.Y., and Huang, C.H., Chem. Soc. Rev., 2010, vol. 39, p. 3007.
Zhang, Y., Liu Z., Zhang, Y., Xu, Y., Li, H., Wang, C., Lu, A., and Sun, S., Sens. Actuators, B, 2015, vol. 211, p. 449.
Kumar, P. and Kumar, S., Detection of bio-relevant metal ions by luminescent Ru(II)-polypyridyl based sensors, in Ruthenium: An Element Loved by Researchers, Ishida, H., Ed., London: Intechopen, 2021, p. 1.
Zhang, Y., Liu, Z., Yang, K., Zhang, Y., Xu, Y., Li, H., Wang, C., Lu, A., and Sun, S.A., Sci. Rep., 2015, vol. 5, p. 8172.
Kumar, S., Singh, S., Kumar, A., and Kumar, P., Dalton Trans., 2021, vol. 50, p. 2705.
Lin, Q.T., Pei, L.M., Xu, W.C., Chao, H., and Ji, L.N., Inorg. Chem. Commun., 2012, vol. 16, p. 104.
Kumar, P. and Kumar, S., J. Mol. Struct., 2020, vol. 1202, p. 127242.
Kumar, S., Arora, A., Kaushal, J., Oswal, P., Kumar, A., and Kumar, P., Inorg. Chem. Commun., 2019, vol. 107, p. 107500.
Arora, A., Kaushal, J., Kumar, A., Kumar, P., and Kumar, S., ChemistrySelect, 2019, vol. 4, p. 6140.
Jiang, Y., Shen, R., Wei, G., Cheng, Y., and Wang, B., Tetrahedron, 2016, vol. 72, p. 2354.
Liu, X.-W., Shen, Y.-M., Lu, J.-L., Chen, Y.-D., Li, L., and Zhang, D.-S., Spectrochim. Acta, Part A, 2010, vol. 77, p. 522.
Benesi, H.A. and Hildebrand, J.H., J. Am. Chem. Soc., 1949, vol. 71, p. 2703.
Prabha, D., Singh, D., Kumar, P., and Gupta, R., Inorg. Chem., 2021, vol. 60, p. 17889.
Lal, S., Prakash, K., Hooda, S., and Kumar, P., J. Mol. Struct., 2020, vol. 1199, p. 127003.
Lal, S., Kumar, S., Hooda, S., and Kumar, P., J. Photochem. Photobiol., A, 2018, vol. 364, p. 811.
Erbas-Cakmak, S., Kolemen, S., Sedgwick, A.C., Gunnlaugsson, T., James, T.D., Yoon, J., and Akkaya, E.U., Chem. Soc. Rev., 2018, vol. 47, p. 2228.
ACKNOWLEDGMENTS
VK and AS are thankful to Meerut College Meerut for providing infrastructure. Amit thanks the Department of Electronics and Communication Engineering, SRM Institute of Science and Technology, NCR Campus for providing the infrastructure.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
No potential conflicts of interest was reported by the authors.
Rights and permissions
About this article
Cite this article
Singh, V., Sindhu, A. & Amit Fluorescence-Based Recognition of Cu(II) Ion in Aqueous Solutions by Ruthenium(II) Complex Linked with Pyridine. J Anal Chem 78, 628–634 (2023). https://doi.org/10.1134/S1061934823050143
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061934823050143