Abstract
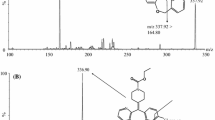
A new liquid chromatography-electrospray ionization tandem mass spectrometry (LC-ESI-MS/MS) method has been developed and validated for alfuzosin quantification in human plasma using amlodipine as an internal standard (IS). The analyte and IS were extracted using a mixture of diethyl ether-dichloroethane (70 : 30, v/v). The samples were separated on a Waters XBridge HILIC column, where a tandem mass spectrometer with a turbo ion spray interface was used for analytes detection. The chromatographic run time of the method was 4.0 min. The method was linear over the concentration range of 0.20–30 ng/mL for alfuzosin. On evaluating the selectivity of the method, no interference was observed for the two MRM transitions of alfuzosin and MRM transition for amlodipine at their respective retention times. The mean matrix effect for alfuzosin was excellent at 99.9%. The method was carefully tested for linearity, system suitability, precision and accuracy. Intra- and inter-day accuracy and precision were evaluated at three quality control levels (LQC, MQC and HQC). Stability of alfuzosin was evaluated under diverse storage conditions. The percentage accuracy for 1 : 2 diluted samples was 93.2% of their nominal concentration with the coefficient of variation of 5.6%. Hence, a highly precise, accurate, rapid, selective and sensitive LC-ESI-MS/MS method has been developed and validated for the quantification of alfuzosin in human plasma using amlodipine as the internal standard.


Similar content being viewed by others
REFERENCES
Miner, M., Rosenberg, M.T., and Perelman, M.A., Clin. Ther., 2006, vol. 28, no. 1, p. 13.
Guinebault, P., Broquaire, M., Colafranceschi, C., and Thenot, J.P., J. Chromatogr. A, 1986, vol. 353, p. 361.
Krstulovic, A.M. and Vende, J.L., Chirality, 1989, vol. 1, no. 3, p. 243.
Rouchouse, A., Manoha, M., Durand, A., and Thenot, J.P., J. Chromatogr. A, 1990, vol. 506, p. 601.
Desager, J.P., Harvengt, C., Bianchetti, G., and Rosenzweig, P., Int. J. Clin. Pharmacol. Ther. Toxicol., 1993, vol. 31, no. 11, p. 568.
Carlucci, G., Di Giuseppe, E., and Mazzeo, P., J. Liq. Chromatogr. Relat. Technol., 1994, vol. 17, no. 18, p. 3989.
Wiesner, J.L., Sutherland, F.C.W., Van Essen, G.H., Hundt, H.K.L., Swart, K.J., and Hundt, A.F., J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2003, vol. 788, no. 2, p. 361.
Mistri, H.N., Jangid, A.G., Pudage, A., Rath od, D.M., and Shrivastav, P.S., J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2008, vol. 876, no. 2, p. 236.
Hugon-Chapuis, F., Mullot, J.U., Tuffal, G., Hennion, M.C., and Pichon, V., J. Chromatogr. A, 2008, vol. 1196, p. 73.
US Department of Health and Human Services, Guidance for Industry, Bioanalytical Method Validation 2001. http://www.fda.gov/cder/guidance/index.html. Accessed June 3, 2021.
Funding
This project was supported by the Deanship of Scientific Research at Prince Sattam bin Abdulaziz University under the research project no. 2020/01/13175.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors confirm that the content of this article has no conflicts of interest.
Rights and permissions
About this article
Cite this article
Moustapha E. Moustapha, El Gamal, R.M. & Kamal, M. Development and Validation of LC-ESI-MS/MS Method for the Determination of Alfuzosin in Human Plasma. J Anal Chem 76, 1327–1335 (2021). https://doi.org/10.1134/S1061934821110095
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061934821110095