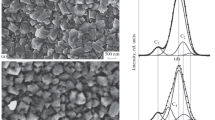
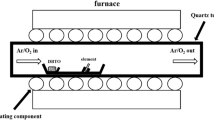
Abstract
A boron-doped diamond electrode with a network structure of PdNi alloy nanowires, which is deposited onto its surface by laser ablation in superfluid helium, is considered as a possible sensor for formaldehyde. It is shown that the electrode is highly sensitive to trace amounts of formaldehyde.





Similar content being viewed by others
REFERENCES
Zhou, Z.L., Kang, T.F., Zhang, Y., and Cheng, S.Y., Electrochemical sensor for formaldehyde based on Pt–Pd nanoparticles and a Nafion-modified glassy carbon electrode, Microchim. Acta, 2009, vol. 164, p. 133. https://doi.org/10.1007/s00604-008-0046-x
Qiao, J., Guo, Y., Song, J., Zhang, Y., Sun, T., Shuang, S., and Dong, C., Synthesis of a palladium-graphene material and its application for formaldehyde determination, Anal. Lett., 2013, vol. 46, p. 1454. https://doi.org/10.1080/00032719.2012.751543
Zhang, J., Shangguan, L., and Dong, C., Electrocatalytic oxidation of formaldehyde and formic acid at Pd nanoparticles modified glassy carbon electrode, Micro Nano Lett., 2013, vol. 8, p. 704. https://doi.org/10.1049/mnl.2013.0186
Ejaz, A., Ahmed, M.S., and Jeon, S., Synergistic effect of 1,4-benzenedimethaneamine assembled graphene supported palladium for formaldehyde oxidation reaction in alkaline media, J. Electrochem. Soc., 2016, vol. 163, p. B163. https://doi.org/10.1149/2.0821605jes
Kongkaew, S., Kanatharana, P., Thavarungkul, P., and Limbut, W., A preparation of homogeneous distribution of palladium nanoparticle on poly (acrylic acid)-functionalized graphene oxide modified electrode for formalin oxidation, Electrochim. Acta, 2017, vol. 247, p. 229. https://doi.org/10.1016/j.electacta.2017.06.131
Bennett, J.A., Wang, J., Show, Y., and Swain, G.M., Effect of sp 2-bonded nondiamond carbon impurity on the response of boron-doped polycrystalline diamond thin-film electrodes, J. Electrochem. Soc., 2004, vol. 151, p. E306. https://doi.org/10.1149/1.1780111
Gordon, E.B., Karabulin, A.V., Matyushenko, V.I., and Khodos, I.I., Experimental study of thermal stability of thin nanowires, J. Phys. Chem. A, 2015, vol. 119, p. 2490. https://doi.org/10.1021/jp5087834
Liu, Z., Yin, Y., Yang, D., Zhang, C., Ming, P., Li, B., and Yang, S., Efficient synthesis of Pt–Co nanowires as cathode catalysts for proton exchange membrane fuel cells, RSC Adv., 2020, vol. 10, p. 6287. https://doi.org/10.1039/D0RA00264J
Khudhayer, W.J., Shaikh, A.U., and Karabacak, T., Platinum nanorod arrays with preferred morphological and crystal properties for oxygen reduction reaction, Adv. Sci. Lett., 2011, vol. 4, p. 3551. https://doi.org/10.1166/asl.2011.1867
Nash, A. and Nash, P., The Ni–Pd (Nickel–Palladium) system, Bull. Alloy Phase Diagr., 1984, vol. 5, p. 446. https://doi.org/10.1007/BF02872890
Yi, Y., Weinberg, G., Prenzel, M., Greiner, M., Heumann, S., Becker, S., and Schlögl, R., Electrochemical corrosion of a glassy carbon electrode, Catal. Today, 2017, vol. 295, p. 32. https://doi.org/10.1016/j.cattod.2017.07.013
Krivenko, A.G., Manzhos, R.A., and Kochergin, V.K., Effect of plasma-assisted electrochemical treatment of glassy carbon electrode on the reversible and irreversible electrode reactions, Russ. J. Electrochem., 2019, vol. 55, p. 663. https://doi.org/10.1134/S102319351907005X
Podlovchenko, B.I., Maksimov, Yu.M., Gladysheva, T.D., and Volkov, D.S., Role of oxides in the electrochemical dissolution of Pd and its alloys, Mendeleev Commun., 2021, vol. 31, p. 561. https://doi.org/10.1016/j.mencom.2021.07.042
Wang, K.-W., Chung, S.-R., and Liu, C.-W., Surface segregation of PdxNi100 – x alloy nanoparticles, J. Phys. Chem. C, 2008, vol. 112, p. 10242. https://doi.org/10.1021/jp800908k
Yan, R.-W. and **, B.-K., Study of the electrochemical oxidation mechanism of formaldehyde on gold electrode in alkaline solution, Chin. Chem. Lett., 2013, vol. 24, p. 159. https://doi.org/10.1016/j.cclet.2013.01.023
Gor’kov, K.V., Talagaeva, N.V., Kleinikova, S.A., Dremova, N.N., Vorotyntsev, M.A., and Zolotukhina, E.V., Palladium–polypyrrole composites as prospective catalysts for formaldehyde electrooxidation in alkaline solutions, Electrochim. Acta, 2020, vol. 345, p. 136164. https://doi.org/10.1016/j.electacta.2020.136164
Doronin, S.V., Manzhos, R.A., and Krivenko, A.G., EDL structure and peculiarities of ferricyanide cyclic voltammetry for silver deposits on gold, Electrochem. Commun., 2015, vol. 57, p. 35. https://doi.org/10.1016/j.elecom.2015.05.003
ACKNOWLEDGMENTS
The work was performed using the equipment of the Scientific Center in Chernogolovka, Osipyan Institute of Solid State Physics, Russian Academy of Sciences.
Funding
The work was performed on the State Assignment, State Registration numbers AAAA-A19-119061890019-5, АААА-А19-119071190040-5, 122040500073-4, and 075-00355-2100.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by T. Kabanova
Delivered at the 20th All-Russian Meeting “Electrochemistry of Organic Compounds” (EKhOS-2022), Novocherkassk, October 18–22, 2022.
Rights and permissions
About this article
Cite this article
Manzhos, R.A., Kochergin, V.K., Krivenko, A.G. et al. Oxidation of Formaldehyde on PdNi Nanowires Synthetized in Superfluid Helium. Russ J Electrochem 59, 714–718 (2023). https://doi.org/10.1134/S1023193523100105
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193523100105