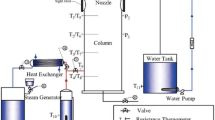
Abstract
Transient processes are experimentally investigated at the initial stage of heat and mass transfer with steam absorption on the surface of 57.8 % water solution of lithium bromide in a bath with a diameter of 70 mm. The experiments were carried out both with a pure solution and with a solution where n-octanol surfactant was added. It is shown that with a sharp steam pressure increase in the volume above the surface of an absorbing solution, local temperature inhomogeneities are generated on the solution surface. The growth rate of these inhomogeneities is proportional to the growth rate of the steam pressure. It was found that, in contrast to the solution without surfactant, where convective flow is observed only during a short time interval after steam supply to the absorber, in a fixed layer of water-LiBr solution with surfactant addition, there is stable thermocapillary convection in the near-surface layer of solution during absorption. It is shown that for variable bottom topography, there can be space-localized zones, where the temperature on the solution surface is determined by the shape of bottom topography.
Similar content being viewed by others
Abbreviations
- a :
-
liquid temperature conductivity, m2/s
- c :
-
surfactant concentration, parts per million (ppm)
- f(t):
-
empirical function of time
- G :
-
dimension parameter for function f(t), °C
- h :
-
liquid layer height, m
- P :
-
pressure, Pa
- r :
-
distance from the cuvette center, m
- q :
-
specific heat of water dissolution in solution, J/kg
- t :
-
time from the moment of steam supply, s
- t* :
-
reference time for empirical function f(t), s
- T :
-
temperature, °C
- x, y :
-
coordinates on the surface, m.
- ΔP :
-
pressure drop, Pa, η — dynamic viscosity, Pa·s
- Θ:
-
temperature field inhomogeneity parameter, °C, λ — length of heat radiation recorded by thermal imager, µm
- ξ :
-
weight concentration of LiBr in solution, kg/kg or %
- ρ :
-
density, kg/m3
- σ :
-
surface tension coefficient, N/m
- τ :
-
characteristic time, s
- Ω:
-
local region.
- \(\bar q\) :
-
specific heat of water dissolution in solution
- s :
-
exponent
- \(\bar η\) :
-
dynamic viscosity
- \(\bar σ\) :
-
surface tension
- \(\bar ρ\) :
-
density.
- 0:
-
initial value
- 1:
-
stage of establishing pressure in the absorber
- i, j :
-
indices for parameter G
- max and min:
-
maximum and minimum values in the local area Ω.
References
N.I. Grigoryeva and V.E. Nakoryakov, Exact solution of combined heat- and mass-transfer problem during film absorption, J. Engng Phys. Thermophys., 1977, Vol. 33, No. 5, P. 1349–1353.
G. Grossman, Simultaneous heat and mass transfer in film absorption under laminar flow, Inter. J. Heat Mass Transfer, 1983, Vol. 26, No. 3, P. 357–371.
V.E. Nakoryakov and N.I. Grigoryeva, Heat and mass transfer at film absorption with a change in the liquid phase volume, Theor. Found. Chem. Engng, 1995, Vol. 29, No. 3, P. 242–248.
H. Daiguji, E. Hihara, and T. Saito, Mechanism of absorption enhancement by surfactant, Inter. J. Heat Mass Transfer, 1997, Vol. 40, No. 8, P. 1743–1752.
V.E. Nakoryakov, N.S. Bufetov, and R.A. Dekhtyar’, Effect of surfactant added in small amounts on nonisothermal absorption: an experimental study, J. Appl. Mech. Tech. Phys., 2004, Vol. 45, No. 2, P. 276–280.
V.E. Nakoryakov, N.I. Grigoryeva, N.S. Bufetov, and R.A. Dekhtyar, Heat and mass transfer intensification at stream absorption by surfactant additives, Inter. J. Heat and Mass Transfer, 2008, Vol. 51, Nos. 21–22, P. 5175–5181.
V.E. Nakoryakov, N.S. Bufetov, N.I. Grigor’eva, and R.A. Dekhtyar’, Heat and mass transfer during absorption by a stagnant solution layer, J. Appl. Mech. Tech. Phys., 2003, Vol. 44, No. 2, P. 236–241.
V.E. Nakoryakov, N.I. Grigoryeva, N.S. Bufetov, R.A. Dekhtyar, and I.V. Marchuk, Vapor absorption by immobile solution layer, Inter. J. Heat and Mass Transfer, 2004, Vol. 47, Nos. 6–7, P. 1525–1533.
S.Ya. Misyura, Evaporation and heat transfer of a sessile drop of aqueous salt solution on heated wall, Inter. J. Heat and Mass Transfer, 2018, Vol. 116, P. 667–674.
S.Ya. Misyura, Evaporation of a sessile water drop and a drop of aqueous salt solution, Scientific Reports, 2017, Vol. 7, No. 1, Art. 14759.
K.I. Stepanov, D.G. Mukhin, S.V. Alekseenko, and O.V. Volkova, Experimental study of negative temperatures in lithium-bromide absorption refrigerating machines, Thermophysics and Aeromechanics, 2015, Vol. 22, No. 4, P. 481–489.
V.G. Levich, Physicochemical Hydrodynamics, Englewood Cliffs, N.J., Prentice-Hall, 1962.
V.E. Nakoryakov, N.I. Grigoryeva, N.S. Bufetov, and R.A. Dekhtyar, Heat and mass transfer at vapor absorption on a row of horizontal tubes, Modern science: research, ideas, results, technologies. Collection of scientific papers, “NPVK Trakon”, Kiev, 2011, Iss. 2, P. 86–90.
M. Mortazavi, R.N. Isfahani, S. Bigham, and S. Moghaddam, Absorption characteristics of falling film LiBr (lithium bromide) solution over a finned structure, Energy, 2015, Vol. 87, P. 270–278.
D. Yu, J. Chung, and S. Moghaddam, Parametric study of water vapor absorption into a constrained thin film of lithium bromide solution, Inter. J. Heat and Mass Transfer, 2012, Vol. 55, P. 5687–5695.
N.S. Bufetov, R.A. Dekhtyar, and V.V. Ovchinnikov, An experimental investigation of thermocapillary convection in solution of lithium bromide with non-isothermal absorption, Vestnik of MSTU, 2016, Vol. 19, No. 3, P. 673–679.
H. Löwer, Thermodynamische und physikalische Eigenschaften der wässrigen Lithiumbromid-Lösung, PhD Thesis, Technische Hochschule Karlsruhe, 1960.
W. Yao, H. Bjurstroem, and F. Setterwall, Surface tension of lithium bromide solutions with heat-transfer additives, J. Chem. Engng Data, 1991, Vol. 36, P. 96–98.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bufetov, N.S., Dekhtyar, R.A. & Ovchinnikov, V.V. Investigation of transient regimes with steam absorption by water solution of lithium bromide. Thermophys. Aeromech. 28, 125–136 (2021). https://doi.org/10.1134/S0869864321010121
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0869864321010121