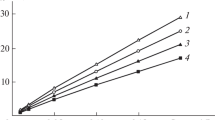
Abstract—The activation energy of the electrical conductivity of ammonium hydroxide–tungstate ion solutions is determined in the NH4OH concentration ranges 0.01–6 and 0.01–0.2 mol/L (based on the metal). For all solutions, the activation energy decreases substantially with increasing temperature and decreases insignificantly with increasing tungsten content in a solution at a constant ammonium hydroxide concentration. An increase in the NH4OH concentration at a constant tungsten content in a solution is found to cause an increase in the activation energy. All calculated activation energies suggest that the charge transfer in the system under study is determined by diffusional restraints.





Similar content being viewed by others
REFERENCES
A. A. Palant and V. A. Bryukvin, “Electrochemical alternating current–assisted processing of metallic tungsten and molybdenum wastes in ammonia electrolytes,” Russ. Metall. (Metally), No. 2, 171–173 (2004).
A. A. Palant and V. A. Pavlovskii, “Physicochemical and technological foundations of electrochemical processing of metallic tungsten wastes,” Tekhnologiya Metallov, No. 11, 3–7 (2003).
A. A. Palant, R. A. Apriamov, and V. A. Pavlovskii, “Electrochemical processing of metallic tungsten wastes,” Tsvetn. Met., No. 10, 47–49 (1995).
G. A. Gadiev, M. S. Kasaeva, and V. M. Alkatseva, “Electrical conductivity of alkaline solutions of sodium tungstate,” in Trudy Molod. Uchen. Vladikavkaz Nauch. Tsentr RAN (2002), Vol. 2, pp. 16–21.
V. M. Alkatseva, “Electrical conductivity of alkaline solutions of sodium tungstate and molybdate,” Izv. Vyssh. Uchebn. Zaved., Tsvetn. Metall., No. 4, 28–31 (2005).
Chemistry and Technology of Rare and Disseminated Elements. Part III: Textbook for Higher Educational Institutes, Ed. by K. A. Bol’shakov, (Vysshaya Shkola, Moscow, 1976).
V. M. Alkatseva, “Electrical conductivity of ammonia solutions of ammonium molybdate,” in Proceedings of V International Conference on Stable Development of Mountain Territories: Problems and Prospects of the Integration of Science and Education (Vladikavkaz, 2004), pp. 288–290.
V. M. Alkatseva, “Electrical conductivity of ammonia solutions of ammonium tungstate,” in Proceedings of International Scientific Technical Conference on Technical Sciences: Tendencies, Prospects, and Technology of Development (Volgograd, 2015), pp. 81–84.
V. V. Shcherbakov, Yu. M. Artemkina, T. N. Ponomareva, and A. D. Kirillov, “Electrical conductivity of an ammonia–water system,” Zh. Neorg. Khim. 54 (2), 321–323 (2009).
Manual on Electrochemistry, Ed. by A. M. Sukhotin (Khimiya, Leningrad, 1981).
D. Dobosh, Electrochemical Constants. A Handbook for Specialists in Electrochemistry (Mir, Moscow, 1980).
O. M. Levchuk, A. M. Levin, and V. A. Bryukvin, “Effect the tungsten(VI) and molybdenum(VI) ions on the electrical conductivity of ammonium hydroxide solutions,” Russ. Metall. (Metally), No. 3, 258–261 (2016).
O. M. Levchuk and A. M. Levin, “Electrical conductivity of ammonia solutions containing rhenium(VII),” Russ. Metall. (Metally), No. 9, 712–715 (2014).
O. G. Kuznetsova, A. M. Levin, M. A. Sevost’yanov, O. V. Tsybin, and A. O. Bol’shikh, “Electrical conductivity of ammonia–alkali solutions and its activation energy,” Russ. Metall. (Metally), No. 9, 921–925 (2019). https://doi.org/10.1134/S0036029519090064
A. A. Palant and G. I. Anufrieva, “Electrical conductivity of ammonium hydroxide aqueous solutions containing tungsten and molybdenum ions,” Hydrometallurgy 42 (3), 435–439 (1996).
A. A. Palant, V. A. Bryukvin, and A. M. Levin, “Electrochemical processing of tungsten-containing carbide wastes of solid alloys,” Tsvetn. Metall., No. 8, 42 (1999).
A. A. Palant, V. A. Bryukvin, and O. M. Gracheva, “Electrochemical processing of metallic tungsten, molybdenum, and rhenium wastes in ammonia electrolytes by alternating current,” Tsvetn. Metallurg., No. 11, 7–9 (2006).
A. Afifi and C. Eizen, Statistical Analysis. Computer Approach (Mir, Moscow, 1982).
V. V. Shcherbakov, Z. Salem, V. I. Ermakov, and A. F. Vorob’ev, “Electrical conductivity and dielectric permeability of aqueous solutions of ammonia,” Elektrokhimiya 28, 283–286 (1992).
A. M. Levin, O. G. Kuznetsova, and M. A. Sevost’yanov, “Calculation of the activation energy of the electrical conductivity of solutions of a NaOH–NH4OH system,” in Proceedings of International Scientific Practical Conference on Innovative Technologies (AMI, Sterlitamak, 2018), pp. 46–50.
L. T. Vlaev and V. G. Georgieva, “Activation energy of the electrical conductivity of aqueous solutions of sulfuric acid, selenic acid, and potassium tellurate,” Elektrokhimiya 40 (6), 768–772 (2004).
A. N. Mashina, Yu. M. Artemkina, and V. V. Shcherbakov, “Temperature dependence of the activation energy of the electrical conductivity of aqueous solutions of strong electrolytes,” Usp. Khim. Khim. Technol. XXXI (4), 49–51 (2017).
A. A. Ivanov, “Electrical conductivity of aqueous solutions of acids in binary and ternary water–electrolyte systems,” Zh. Neorg. Khim. 53 (12), 2081–2097 (2008).
V. V. Shcherbakov. Yu. M. Artemkina, and T. N. Ponomareva, “Electrical conductivity of concentrated aqueous solutions of propionic acids, sodium propionates, and their mixture,” Elektrokhimiya 44 (10), 1275–1280 (2008).
Funding
This work was carried out in terms of state assignment no. 075-00746-19-00.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by E. Yablonskaya
Rights and permissions
About this article
Cite this article
Kuznetsova, O.G., Levin, A.M., Sevost’yanov, M.A. et al. Electrical Conductivity of Tungsten-Containing Ammonia Solutions and Calculation of Its Activation Energy. Russ. Metall. 2020, 1343–1348 (2020). https://doi.org/10.1134/S0036029520110099
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036029520110099