Abstract
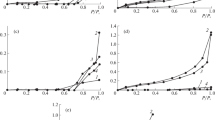
New data are obtained on the thermodynamics and molecular mechanisms of the interaction between dioxane and nanocluster polyoxomolybdates of the keplerate and toroidal types, with their composites containing water-soluble nonionic polymers—polyvinyl alcohol and polyvinylpyrrolidone, and between the components of such composites. The studies are performed using a gravimetric variant of equilibrium interval sorption with thermodynamic cycle calculations, and by IR spectroscopy. The effect the nature of the sorbates and the sorbents has on the parameters of sorption is analyzed using the constructed sorption isotherms and the concentration dependences of the Gibbs energy, particularly by comparing dioxane to other low-molecular compounds that differ appreciably in polarity and their ability to form hydrogen bonds: methanol, benzene, acetone, and ethyl acetate.





Similar content being viewed by others
Change history
03 December 2020
erratum
REFERENCES
A. Müller, E. Krickemeyer, H. Bogge, et al., Angew. Chem., Int. Ed. Engl. 37, 3360 (1998).
A. Müller, V. P. Fedin, C. Kuhlmann, et al., Chem. Commun., No. 10, 927 (1999).
A. Müller, S. Sarkar, S. Q. N. Shah, et al., Angew. Chem., Int. Ed. Engl. 38, 3238 (1999).
A. Müller, R. Maiti, M. Schmidtmann, et al., Chem. Commun., 2126 (2001).
T. Liu, E. Diemann, and A. Müller, J. Chem. Educ. 84, 526 (2007).
D. G. Kurth, P. Lehmann, D. Volkmer, et al., J. Chem. Soc., Dalton Trans., No. 21, 3989 (2000).
A. Müller and P. Gouzerh, Chem. Soc. Rev. 41, 7431 (2012).
C. Besson, S. Schmitz, K. M. Capella, et al., J. Chem. Soc., Dalton Trans. 41, 9852 (2012).
S. Kopilevich, A. Gil, M. Garcia-Ratés, et al., J. Am. Chem. Soc. 134, 13082 (2012).
J. Elistratova, B. Akhmadeev, A. Gubaidullin, et al., New J. Chem. 41, 5271 (2017).
A. A. Ostroushko, I. G. Danilova, I. F. Gette, and M. O. Tonkushina, Russ. J. Inorg. Chem. 60, 500 (2015).
A. A. Ostroushko, I. D. Gagarin, M. O. Tonkushina, K. V. Grzhegorzhevskii, I. G. Danilova, I. F. Gette, and G. A. Kim, Russ. J. Phys. Chem. A 91, 1811 (2017).
A. M. Popa, L. Hu, D. Crespy, et al., J. Membr. Sci. 373, 196 (2011).
A. A. Ostroushko, L. V. Adamova, E. V. Eremina, and K. V. Grzhegorzhevskii, Russ. J. Phys. Chem. A 89, 1439 (2015).
A. A. Ostroushko, L. V. Adamova, and E. V. Eremina, Russ. J. Phys. Chem. A 91, 1536 (2017).
K. V. Grzhegorzhevskii, L. V. Adamova, E. V. Eremina, and A. A. Ostroushko, Russ. J. Phys. Chem. A 91, 561 (2017).
A. A. Ostroushko, L. V. Adamova, and E. V. Eremina, Russ. J. Phys. Chem. A 91, 1536 (2017).
A. A. Ostroushko, L. V. Adamova, and E. V. Koveza, Russ. J. Phys. Chem. A 92, 2242 (2018).
A. A. Ostroushko, L. V. Adamova, E. V. Eremina, K. V. Grzhegorzhevskii, E. V. Velichko, S. G. Bogdanov, and A. N. Pirogov, Russ. J. Phys. Chem. A 91, 1313 (2017).
A. A. Ostroushko, A. P. Safronov, M. O. Tonkushina, V. Yu. Korotaev, and A. Yu. Barkov, Russ. J. Phys. Chem. A 88, 2179 (2014).
A. A. Ostroushko, M. O. Tonkushina, and N. A. Martynova, Russ. J. Phys. Chem. A 84, 1022 (2010).
A. A. Ostroushko, V. A. Vazhenin, and M. O. Tonkushina, Russ. J. Inorg. Chem. 62, 483 (2017).
Experimental Methods in Adsorption and Molecular Chromatography, Ed. by Yu. S. Nikitin and R. S. Petrova (Mosk. Gos. Univ., Moscow, 1990) [in Russian].
K. V. Grzhegorzhevskii, P. S. Zelenovskiy, O. V. Koryakova, and A. A. Ostroushko, Inorg. Chim. Acta 489, 287 (2019).
The Chemist’s Handbook, Ed. by B. P. Nikol’skii (Khimiya, Moscow, Leningrad, 1982), Vol. 1, p. 963 [in Russian].
A. V. Karyakin and G. A. Kriventsova, The State of Water in Organic and Inorganic Compounds (Nauka, Moscow, 1973) [in Russian].
K. Mizuno, S. Imafuji, T. Fujiwara, et al., J. Phys. Chem. B 107, 3972 (2003).
O. A. Mel’nikova, I. A. Samkova, M. Yu. Mel’nikov, et al., Usp. Sovrem. Estestvozn., No. 8, 42 (2016).
ACKNOWLEDGMENTS
The authors thank M.Yu. Kornev for his help in performing our spectroscopic studies of samples.
Funding
This work was supported by the RF Ministry of Education and Science, project no. 4.6653.2017/8.9; and by the Competitiveness Enhancement Program of Yeltsin Ural Federal University, agreement no. 02.A03.21.0006.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by V. Glyanchenko
Rights and permissions
About this article
Cite this article
Ostroushko, A.A., Adamova, L.V., Grzhegorzhevskii, K.V. et al. Interaction between Dioxane and Nanocluster Polyoxomolybdates and Their Composites Containing Water-Soluble Polymers. Russ. J. Phys. Chem. 94, 762–771 (2020). https://doi.org/10.1134/S0036024420040159
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024420040159