Abstract
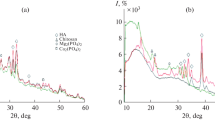
The CaCl2-(NH4)2HPO4-(C6H11NO4)n-NH3-H2O system at 25°C was studied by the solubility (Tananaev’s residual concentrations) technique and pH measurements. The parameters providing for the coprecipitation of nanocrystalline (12.5–18.7 nm) calcium and chitosan hydroxylapatites were found. Calcium-deficient chitosan hydroxylapatites Ca9.8(PO4)6(OH)1.6 · xC6H11NO4 · yH2O, where x = 0.075 or 0.37 and y = 5.8 or 6.2, and stoichiometric calcium hydroxylapatites Ca10(PO4)6(OH)2 · xC6H11NO4 · yH2O, where x = 0.075, 0.1, 0.2, 0.37, 0.5, or 0.75 and y = 5.7–7.5, were synthesized. Solid phases were characterized by chemical analysis, X-ray powder diffraction, thermogravimetric analysis, and IR spectroscopy.
Similar content being viewed by others
References
V. P. Orlovskii, Zh. A. Ezhova, G. V. Rodicheva, et al., Zh. Neorg. Khim. 37(5), 881 (1992).
G. Schwarzenbach and G. Flaschka, Die kompleksometrische Titration (Ferdinand Enke, Stuttgart, 1965; Khimiya, Moscow, 1970).
N. A. Chumaevskii, V. P. Orlovskii, Zh. A. Ezhova, et al., Zh. Neorg. Khim. 37(7), 1455 (1992).
L. Bellamy, The Infrared Spectra of Complex Molecules (London, 1954; Inostrannaya Literatura, Moscow, 1963).
JCPDC File, no. 9-432 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kalinnikov, V.T., Ezhova, Z.A., Zakharov, N.A. et al. Synthesis and Physicochemical Study of Chitosan-Containing Calcium Hydroxylapatites. Russ. J. Inorg. Chem. 53, 357–361 (2008). https://doi.org/10.1134/S0036023608030030
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023608030030