Abstract
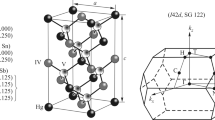
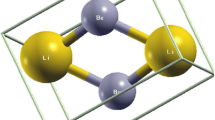
Total, difference, and deformation electron densities are calculated from the first principles using the density functional theory and the sublattice method for LiBX (B = Mg, Ca, Zn; X = N, P, As) crystals with the sphalerite structure. The nature and formation features of the chemical bonding caused by a change in the chemical composition are revealed. A weak bond between Li+ ions with X anions enables their displacements in the space between crystal-forming tetrahedral (BX)– groups. It is found that Ca–X bonds are mainly ionic and in a series of crystals the ionic covalent Li–B bond is traced.
Similar content being viewed by others
References
D. Kieven and R. Klenk, Phys. Rev. B, 81, No. 7, 075208 (2010).
T. Gruhn, Phys. Rev. B, 82, No. 12, 125210 (2010).
D. M. Wood, A. Zunger, and R. de Groot, Phys. Rev. B, 31, No. 4, 2570 (1985).
E. Carlsson, A. Zunger, and D. M. Wood, Phys. Rev. B, 32, No. 2, 1386 (1985).
S.-H. Wie and A. Zunger, Phys. Rev. Lett., 56, No. 5, 528 (1986).
K. Kuriyama and T. Katoh, Phys. Rev. B, 37, No. 12, 7140 (1988).
K. Kuriyama, K. Tomoharu, and T. Tanaka, Phys. Rev. B, 49, No. 7, 4511 (1994).
K. Kuriyama, T. Kato, and K. Kawada, Phys. Rev. B, 49, No. 16, 11452 (1994).
K. Kuriyama, K. Kushida, and R. Taguchi, Solid State Commun., 108, No. 7, 429 (1998).
K. Kuriyama, R. Taguchi, K. Kushida, and K. Ushiyama, J. Cryst. Growth, 198/199, No. 1, 802 (1999).
K. Kuriyama, T. Ishikawa, and K. Kushida, Phys. Rev. B, 72, No. 23, 233201 (2005).
K. Kuriyama, Y. Yamashita, T. Ishikawa, and K. Kushida, Phys. Rev. B, 75, No. 23, 233204 (2007).
S. Kacimi, H. Mehnane, and A. Zaoui, J. Alloys Compd., 587, No. 2, 451 (2014).
M. V. Starodubtseva and Yu. M. Basalaev, Fundam. Probl. Sovrem. Materialoved., 9, No. 3, 349 (2012).
M. V. Starodubtseva and Yu. M. Basalaev, Topical Problems of Solid State Physics, Proc. Int. Sci. Conf., vol. 2, Minsk (2013), p. 159.
Yu. M. Basalaev and M. V. Starodubtseva, Izv. Vyssh. Uchebn. Zaved., Fiz., 57, No. 1, 124 (2014).
R. Dovesi et al., Z. Kristallogr., 220, No. 5/6, 571 (2005).
Yu. M. Basalaev and A. S. Poplavnoi, J. Struct. Chem., 50, No. 6, 1229 (2009).
Yu. N. Zhuravlev and A. S. Poplavnoi, J. Struct. Chem., 42, No. 5, 711 (2001).
Yu. M. Basalaev, Yu. N. Zhuravlev, A. V. Kosobutskii, and A. S. Poplavnoi, Phys. Solid State, 46, No. 5, 848 (2004).
H. J. Monkhorst and J. D. Pack, Phys. Rev. B, 13, No. 12, 5188 (1976).
F. Casper et al., Semicond. Sci. Technol., 27, No. 6, 063001 (2012).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © 2015 Yu. M. Basalaev, M. V. Starodubtseva.
Translated from Zhurnal Strukturnoi Khimii, Vol. 56, No. 6, pp. 1088–1094, November–December, 2015.
Rights and permissions
About this article
Cite this article
Basalaev, Y.M., Starodubtseva, M.V. Features of the valence electron charge distribution in LiBIIXV crystals. J Struct Chem 56, 1036–1042 (2015). https://doi.org/10.1134/S0022476615060025
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476615060025