Abstract
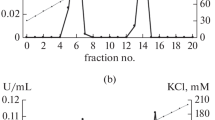
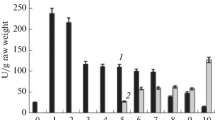
The use of a modified, four-stage purification scheme allowed us to obtain electrophoretically homogeneous preparations of constitutive forms of succinate dehydrogenase from maize (Zea mays L.) scutellum in the late stages of seed germination. It has been established that the isoenzymes differed significantly in their quaternary structure. Thus, succinate dehydrogenase 1 turned out to be a heterotetramer, and succinate dehydrogenase 2 turned out to be a heteroterodimer. It was found that their main catalytic and kinetic characteristics also differed, in particular, by their affinity to the substrate (succinate) and the value of pH optimum of the catalyzed reaction. Succinate dehydrogenase 2 was characterized by enhanced resistance to a specific inhibitor—malonate.





Similar content being viewed by others
REFERENCES
Eprintsev, A.T., Popov, V.N., and Shevchenko, M.Yu., Ekspressiya i regulyatsiya fermentov glioksilatnogo tsikla (Expression and Regulation of Glyoxylate Cycle Enzymes), Voronezh: Tsentr.-Chern. Knizh. Izd., 2005.
Eprintsev, A.T., Fedorin, D.N., Sazonova, O.V., and Igamberdiev, A.U., Plant Physiol. Biochem., 2016, vol. 102, pp. 161–166.
Figueroa, P., Leon, G., Elorza, A., Holuigue, L., Araya, A., and Jordana, X., Plant. Mol. Biol., 2002, vol. 50, nos. 4–5, pp. 725–734.
Millar, A.H., Eubel, H., Jansch, L., Kruft, V., Heazlewood, J.L., and Braun, H-P., J. Plant Mol. Biol. Biotech., vol. 56, no. 1, pp. 77–90.
Eprintsev, A.T., Fedorin, D.N., Selivanova, N.V., Akhmad, Dzh.A., and Popov, V.N., Biol. Bull. (Moscow), 2010, vol., no. 3, pp. 324–332.
Eprintsev, A.T., Fedorin, D.N., Karabutova, L.A., and Igamberdiev, A.U., J. Plant Physiol., 2016, vol. 205, no. 1, pp. 33–40.
Eprintsev, A.T., Popov, V.N., and Fedorin, D.N., Suktsinatdegidrogenaza vysshikh rastenii (Higher Plant Succinate Dehydrogenase), Voronezh: Tsentr.-Chern. Knizh. Izd., 2010.
Popov, V.N., Eprintsev, A.T., and Fedorin, D.N., Russ. J. Plant Physiol., 2007, vol. 54, no. 3, pp. 360–365.
Eprintsev, A.T., Fedorin, D.N., Selivanova, N.V., Vu, T.L., Makhmud, A.S., and Popov, V.N., Russ. J. Plant Physiol., 2012, vol. 59, no. 3, pp. 299–306.
Elorza, A., Roschzttardtz, H., Go’mez, I., Mouras, A., Holuigue, L., Araya, A., and Jordana, X., Plant Cell Physiol., 2006, vol. 47, no. 1, pp. 14–21.
Eprintsev, A.T., Fedorin, D.N., Nikitina, M.V., and Igamberdiev, A.U., J. Plant Physiol., 2015, vol. 181, no. 1, pp. 14–19.
Eprintsev, A.T., Glioksilatnyi tsikl. Universal’nyi mekhanizm adaptatsii? (Glyoxylate Cycle: A Universal Adaptation Mechanism?), Moscow: Akademkniga, 2007.
Pineiru, deKarval’yuM.A.A., Zemlyanukhin, A.A., and Eprintsev, A.T., Malatdegidrogenaza vysshikh rastenii (Higher Plant Malate Dehydrogenase), Voronezh: Voronezh. Gos. Univ., 1991.
Lowry, O.H., Rosebrough, N.J., Farr, A.L., and Randall, R.J., J. Biol. Chem., 1951, vol. 193, no. 1, pp. 265–275.
Davis, B.J., Ann. N. Y. Acad. Sci., 1964, vol. 121, no. 2, pp. 404–427.
Shevchenko, A., Wilm, M., Vorm, O., and Mann, M., Anal. Chem., 1996, vol. 68, no. 5, pp. 850–858.
Lakin, G.F., Biometriya (Biometry), Moscow: Vysshaya Shkola, 1990.
Igamberdiev, A.U. and Eprintsev, A.T., Front. Plant Sci., 2016, vol. 15, no. 7, p. 1042.
Day, D.A., Dry, I.B., Soole, K.L., Wiskich, J.T., and Moore, A.L., Plant Physiol., 1991, vol. 95, no. 3, pp. 948–953.
Eprintsev, A.T. and Popov, V.N., Fermentativnaya regulyatsiya metabolizma di- i trikarbonovykh kislot v rasteniyakh (Enzymatic Regulation of the Metabolism of Di- and Tricarboxylic Acids in Plants), Voronezh: Voronezh. Gos. Univ., 1999.
Valls-Lacalle, L., Barba, I., Miro-Casas, E., Alburquerque-Bejar, J.J., Ruiz-Meana, M., Fuertes-Agudo, M., Rodriguez-Sinovas, A., and Garcia-Dorado, D., Cardiovasc. Res., 2016, vol. 109, no. 3, pp. 374–384.
Belt, K., Huang, S., Thatcher, L.F., Casarotto, H., Singh, K.B., Van Aken, O., and Millar, A.H., Plant Physiol., 2017, vol. 173, no. 4, pp. 2029–2040.
Muller, F.L., Liu, Y., Abdul-Ghani, M.A., Lustgarten, M.S., Bhattacharya, A., Jang, Y.C., and Van Remmen, H., Biochem. J., 2008, vol. 409, no. 2, pp. 491–499.
Funding
The work was financially supported by the Russian Science Foundation (project no. 19-14-00150).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest. This article does not contain any studies involving animals or human participants performed by any of the authors.
Additional information
Translated by T. Borisova
Rights and permissions
About this article
Cite this article
Eprintsev, A.T., Fedorin, D.N. Purification of Constitutive Isoenzymes of Succinate Dehydrogenase from Zea mays Scutellum in the Homogeneous State and the Study of Their Characteristics. Appl Biochem Microbiol 56, 179–184 (2020). https://doi.org/10.1134/S0003683820020064
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0003683820020064