Abstract
Purpose
The pathogenesis of pterygium is not well known, and controversy exists about the cell origins and the nature of initial trigger required for its development. We investigated whether endothelial progenitor cells (EPCs) are involved in pathogenesis of pterygium and the mechanism underlying the selective recruitment of EPCs during this process.
Methods
We studied 13 normal controls and 28 pterygium patients (primary (n=15), recurrent (n=13)). Substance-P, vascular endothelial growth factor (VEGF), and stem cell factor (SCF) were measured in plasma and tears using ELISA, and circulating CD34+ and c-kit+ mononuclear cells (MNCs) by flow cytometry. Anterior segment fluorescein angiography (FAG) was performed to evaluate hypoxic conditions in the early stage of pterygium. Surgically removed pterygial tissues were analyzed immunohistochemically using the progenitor cell markers, CD34, c-kit, VEGFR-1, and VEGFR-2.
Results
Anterior segment FAG findings showed an increase in non-perfusion areas and attenuated vessels in the nasal limbus during early-stage pterygium. Circulating CD34+ MNCs and c-kit+ MNCs were increased in pterygium groups compared with normal controls. Systemic and local cytokines including SP, VEGF, and SCF in pterygium groups were also elevated and showed positive correlations with CD34+ and c-kit+ MNC numbers. Immunohistochemical analysis of pterygium showed strong progenitor cell marker immunoreactivities.
Conclusions
EPCs might be involved in pterygium development, and ocular hypoxia triggers this neovascularization by recruiting EPCs derived from the bone marrow via the production of systemic and local cytokines.
Similar content being viewed by others
Introduction
Pterygium is a chronic disease that is characterized by an aggressive invasion of fibrovascular tissue into the cornea, and is one of the most common ocular surface diseases in tropical and subtropical zones.1 Many suffer from this aggressive disease and its prevalence in the general population has been estimated to be 7–13%.2, 3 Several environmental factors that may induce ocular inflammation and various cytokines have been reported to be involved in the development of pterygium.4, 5, 6 However, the precise mechanism of pterygium pathogenesis is unclear.
New vessel formation in adults was initially believed to result exclusively from the proliferation and migration of fully differentiated endothelial cells derived from pre-existing vessels.7, 8 However, recent studies suggest that circulating endothelial progenitor cells (EPCs) derived from the bone marrow also contribute to postnatal neovascularization.9, 10 EPCs are characterized by CD34, VEGFR-2, and CD133 antigen-positive cells, and are known to be stimulated by several modulators, such as vascular endothelial growth factor (VEGF), stem cell factor (SCF), and substance-P (SP).11, 12, 13, 14
Evidence suggests that VEGF is a potent endothelial cell-specific factor, and VEGF has been demonstrated to have a chemotactic effect on EPCs, whereas SCF (also called c-kit ligand) is a multi-potent growth factor that targets diverse haematopoietic cells including stem cells.13, 15 Another modulating factor, SP (a neurotransmitter), has been reported to be involved in angiogenesis and neuroimmuno-haematopoietic interactions,16, 17 and in corneal wound healing.18, 19 Previous reports indicate that these stem cell-modulating factors are expressed in pterygium.20, 21 In addition, we previously reported that bone marrow-derived stem and progenitor cells are detected in the pterygial tissue.22 However, these reports are limited to studies of the local effects on pterygium, and little is known of the physiologic mechanism underlying the localized expressions of these stem cell-modulating factors.
Given the above background, we hypothesized that new vessel formation in pterygium is a process of postnatal vasculogenesis, and that stem cell modulators are likely to be involved in its pathogenesis. To identify the factor that triggers postnatal vasculogenesis, we used anterior segment fluorescein angiography (FAG) to investigate the vasculature of pterygium and hypoxic conditions in early pterygium. We also investigated the local and systemic expressions of stem cell-modulating factors and circulating EPCs to characterize the mobilization and migration of EPCs under the influence of chemotactic factors. Finally, we immunostained for angiogenic-related factors and infiltrating EPCs in pterygial tissues to elucidate the development of postnatal vasculogenesis during the pathogenesis of pterygium.
Materials and methods
The protocol and consent form for the study were approved by the Institutional Review Board at Chung-Ang University, Seoul, Korea. All procedures were performed under the tenets of the Helsinki declaration and informed consent was obtained from all patients.
Patients and sample collection
We studied 15 primary cases, 13 recurrent pterygia cases, and 13 controls that planned to undergo cataract surgery. Patients with a history of previous ocular surgery (except pterygium removal), ischaemic cardiovascular disease, haematologic disorder, or malignant disease were excluded to prevent confusions with other systemic hypoxic diseases. All patients underwent a detailed bio-microscopic examination preoperatively.
Blood and tear samples were collected before pterygium excision. Tear samples for enzyme-linked immunosorbent assays (ELISA) were collected from the inferior tear meniscus of affected eyes after 5 min after topical anaesthesia (Alcaine®, Alcon, Ft Worth, TX, USA) to minimize irritation, using 8 mm polyester wicks (Transorb rods®, American Filtrona, Richmond, VA, USA).23 Wicks were then placed into the end of a micropipette tip located within a 1.5 ml tube and centrifuged at 10 000 r.p.m. for 5 min. Venous blood was collected from an antecubital vein, and divided into two EDTA-coated tubes for ELISA and flow cytometric analysis. For ELISA, samples were centrifuged at 3000 g for 5 min to get plasma and then stored in a deep freezer at −70°C.
Consecutive pterygial tissues were obtained after surgical removal, and normal conjunctiva tissues (n=7) were obtained at the time of cataract surgery. All tissue specimens were paraffin-embedded and snap-frozen. Serial cross-sections were made along the longitudinal axis.
Anterior segment FAG
To evaluate relative ischaemic state of bulbar conjunctivae, we performed anterior segment FAG. Ten patients with early pterygium and 13 normal controls participated in this study. We defined early pterygium as a microscopic fibrovascular proliferation, not grossly invading the cornea, with punctate epithelial erosion, and microaneurysm. Sodium fluorescein (10%, 5 ml) was injected into an antecubital vein and conjunctival fluorescence was photographed using a fundus camera (Topcon TRC-50IA, Topcon, Tokyo).24
Measurement of chemotactic factors
To quantify local and systemic chemotactic factors, SP, VEGF, and SCF levels in tears and plasma were examined by double-sandwiched ELISA using commercial ELISA kits. Competitive ELISAs were used to quantify SP immunoreactivity, as previously reported,25 using a commercial ELISA kit (R&D Systems, Minneapolis, MN, USA). VEGF and SCF (R&D Systems, Minneapolis, MN) were measured in a similar manner. Complete blood cell counts and c-ESR were checked to establish base line levels.
Quantification of circulating mononuclear cells
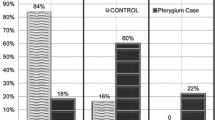
To determine percentages of CD34+ cells, peripheral white blood cells were collected and stained with phycoerythrin-conjugated anti-CD34 monoclonal antibody (for specific staining) and with anti-CD45 antibody (for nonspecific staining) (Becton-Dickinson, San Jose, CA, USA). The reagent kit (ProCOUNT™ Becton-Dickinson Immunocytometry Systems (BDIS), San Jose, CA, USA) was used for progenitor cell enumeration (Figure 1). FACS™ Lysing Solution was used to lyse erythrocytes before the sample analysis on the flow cytometer (FACScan, Becton-Dickinson, San Jose, CA, USA). Peripheral blood c-kit+MNCs were also identified using anti-c-kit mouse monoclonal antibody (Becton-Dickinson, San Jose, CA, USA). After appropriate gating, cell-associated immunofluorescence was analysed using a FACscan flow cytometer system (Becton-Dickinson, San Jose, CA, USA).
Representative histogram profile of CD34+ MNCs (a) with matching negative control antibody (b). Phenotypic characterization of circulating CD34+ MNCs was performed using the ProCOUNT™ reagent kit. The CD34 reagent contains a nucleic acid and phycoerythrin (PE)-labelled murine monoclonal anti-45, clone 2D1 (a). The control reagent is used to assess the amount of nonspecific antibody binding, particularly that caused by Fc receptors (b).
Immunohistochemical study
Sectioned (2–4 μm) specimens were processed immunohistochemically, as previously described.26 Briefly, paraffin sections were deparaffinized in xylene, and rehydrated, and quenched for endogenous peroxidase. Cryostat sections were placed on gelatinized slides, and fixed in cold acetone. Tissue sections were equilibrated in Tris-buffered saline (TBS), blocked in non-immuno serum (Zymed Laboratories, South San Francisco, CA, USA). Sections were then incubated with antibodies to mouse anti-human CD34 (1 : 100), c-Kit (1 : 100; all from Neo Markers, Fremont, CA, USA), hypoxia-inducible factor-1α (HIF-1α; 1 : 200; Novus Biologicals, Littleton, CO, USA), to rabbit anti-human VEGF (1 : 100), VEGFR-1 (1 : 100), VEGFR-2 (1 : 100), SP (1 : 100; all from Neo Markers, Fremont, CA, USA) overnight at 4°C. Sections were extensively washed in TBS before adding a biotinylated secondary antibody, rewashed, incubated for 1 h with peroxidase-conjugated streptavidin, and the presence of peroxidase was revealed by adding substrate-chromogen (3-amino-9-ethycarbazole) solution. The sections were then counterstained with haematoxylin. Tissues were examined under an optical microscope and photo-documented.
Statistical analysis
All data are expressed as means±SD. Statistical analysis was performed using SPSS statistical software (Ver. 10.0). Differences between the normal control, primary, and recurrent pterygium groups were assessed using multiple analysis of variance (MANOVA). Correlation between mobilizing EPCs and local and systemic chemotactic factors was analyzed by multiple correlation analyses. The probability level for statistical significance was set at 5% and high-level significance was set at 1%.
Results
Clinical findings and anterior segment FAG
Ages and sex ratios are shown as means in Table 1, but no significant difference was observed between the three groups in terms of mean age or gender. All pterygia were nasal and unilateral. In both primary and recurrent pterygium, local ocular surface inflammatory signs such as punctate keratopathy were observed.
In early pterygium, the invasion of small, coarse new vessels with an overlying translucent epithelium, and punctate epithelial erosions were found (Figure 2a and b). Anterior FAG findings of early pterygium showed delayed fluorescein dye filling, increased non-perfusion areas, and attenuated and obscured vessels at nasal limbi (Figure 2c).
Hypoxic and inflammatory conditions during early-stage pterygium. Biomicroscopic finding (a, b), anterior segment FAG of early-stage pterygium (c), and of normal conjunctiva (d). The invasion of small vessels beyond the cornea with underlying translucent epithelium was observed (a, arrow). Ocular surface inflammatory signs, such as superficial punctate keratopathy, were observed (b, arrowhead). Delayed perfusion of fluorescent dye and vessel attenuation were observed in early-stage pterygium but not normal conjunctival tissue samples at the same time (c, d). (*perfusion time).
Local and systemic chemotactic factors
SP and VEGF levels in tears were significantly elevated in the two pterygium groups as compared with the normal controls (Table 2), and these levels were more prominent in the recurrent pterygium group than in the primary pterygium group (P<0.01). SCF was not detected in tears.
In plasma, changes in chemotactic factors were similar to those observed in tears. SP levels were higher in the two pterygium groups than in the normal controls, and were more prominent in the recurrent pterygium group than in the primary pterygium group (P<0.05, P<0.01 respectively). VEGF and SCF showed patterns similar to that of SP. c-ESR levels were 1.65 and 2.66 times higher in the primary and recurrent pterygium groups than in the control group (P<0.01).
Circulating MNCs
The circulating CD34+ MNCs were higher in the primary and recurrent pterygium groups than in the normal control group (P<0.05, P<0.01, respectively). CD34+ MNCs counts in the primary and recurrent groups were also significantly different (P<0.01). C-kit+ MNCs, indicating haematopoietic and endothelial stem cells, were also significantly higher in the primary and recurrent pterygium groups than in normal controls (P<0.05), but the difference between the two pterygial groups was not significant. Table 2 summarizes the results of flow cytometric analyses for circulating CD34 positive and c-kit positive MNCs.
Immunohistochemical expression of angiogenic-related factors and EPCs
Routine histological sections with H&E staining revealed chronic inflammatory cells infiltration in the stroma of pterygium tissues, and this was more prominent in recurrent than in primary pterygium. Immunohistochemical analysis with angiogenic-related factors showed positively stained cells in the pterygial epithelium and stroma. HIF-1α was expressed in the epithelial cells, and VEGF was coexpressed in the same cells (Figure 3a and b). SP was expressed in the basal epithelium and in round stromal cells (Figure 3c). Compared with the primary pterygium, HIF-1α- and VEGF-positive cells were more numerous and staining densities were higher in recurrent pterygial tissues (Figure 3d and e). In normal conjunctiva, HIF-1α- and VEGF-positive cells were not observed in pterygial epithelium (Figure 3f).
Detection and localization of HIF-1α, VEGF, and substance-P in pterygium and normal conjunctiva. HIF-1α and VEGF were stained in primary pterygial epithelium (a, b, arrow) and in stromal vascular endothelium. Substance-P was expressed in the basal epithelium (c, arrow) and in round stromal cells (c, arrowhead). In recurrent pterygial tissue, expressions of HIF-1α (d) and VEGF (e) were stronger than in primary pterygium. In normal conjunctiva, HIF-1 α was not expressed (f). Original magnification: × 200.
Progenitor cell markers, such as CD34, c-kit, VEGFR-1, and VEGFR-2, were expressed strongly in epithelial and stromal layers, particularly around small blood vessels in stromal layers (Figure 4a–d). The expressions of these markers were also stronger in recurrent pterygium (data not shown). In normal conjunctiva, CD34-, VEGFR-1-, and VEGFR-2-positive cells were focally but rarely observed in vascular endothelium (Figure 4e–g).
Immunohistochemical staining of endothelial progenitor cell markers in primary pterygium and normal conjunctiva. Expressions of CD34 (a), VEGFR-1 (b), VEGFR-2 (c), and c-kit (d) were noted in cells clustered around small stromal blood vessels of pterygium. Immunohistochemical staining of a normal conjunctiva shows rare expression of CD34 (e), VEGFR-1 (f), and VEGFR-2 (g). Original magnification: × 400.
Correlation between circulating EPCs and chemotactic factors
Multiple correlation analyses were performed to determine the correlations between levels of both local and systemic chemotactic factors and circulating CD34+ and c-kit+ MNCs. Circulating CD34+ MNCs were present at low levels in normal controls, and were weakly correlated with the levels of local and systemic chemotactic factors. In contrast, systemic and local chemotactic cytokines, including SP, VEGF, and SCF, in the pterygium groups were found to be strongly and positively correlated with CD34+ MNC levels (r: 0.735–0.846). These correlations were more prominent in the recurrent pterygium group (r: 0.787–0.926).
C-kit+ MNC levels were not found to be related to any chemotactic cytokine expression level in normal controls. In primary pterygium, VEGF in tears and SP and SCF in plasma were found to be moderately correlated with c-kit+ MNC levels (r: 0.585–0.650). However, in recurrent pterygium, c-kit+ MNCs were closely associated with all systemic and local chemotactic factors except systemic VEGF (r: 0.767–0.972).
Discussion
The present study demonstrates that bone marrow-derived EPCs are involved in the pathogenesis of pterygium. We found that the number of cells expressing CD34 and c-kit (markers of bone marrow-derived circulating progenitor cells) in peripheral blood was higher in the primary and recurrent pterygium groups than in the normal controls. Moreover, we observed cells expressing progenitor cell markers, such as CD34, c-kit, VEGFR-1, and VEGFR-2, in the pterygium, particularly around small blood vessels. Previous reports have suggested that CD34+ cells coexpressing AC133 and VEGFR-2 are functional endothelial cells that have the capacity to migrate and differentiate into mature endothelial cells.11 The colocalizations of these markers and the perivascular infiltration of these cells suggest that EPCs might be involved in the vasculogenesis needed for pterygium development. Although we did not confirm this directly by inducing endothelial phenotype from the isolated putative EPC in peripheral blood using in vitro study, our data have the significance for demonstration of progenitor cells elevation in the pterygium patients in vivo.
The basic mechanism of how bone marrow-derived progenitor cells migrate from the bone marrow to the limbus via the vascular system to participate in pterygium development is unclear. Hypoxia is a well-known trigger factor of this stem cell recruitment. Our clinical observations show a highly developed limbal vascular arcade and the invasion of small vessels during the earliest stages of pterygium. In addition, we found delayed perfusion and attenuation of coarsened small vessels in the early stage of pterygium by anterior segment FAG. These findings are in accordance with those of a previous report, which suggested the importance of ocular surface hypoxia in pterygium development.27 In our study, the immunostaining of pterygium tissues revealed the expressions of angiogenic-related factor, such as HIF-1α, VEGF, and SP, supporting the initiatory role of ocular hypoxia in postnatal vasculogenesis. Environmental factors, such as UV, heat, and wind, might cause chronic inflammation and sublethal hypoxia, which in turn could induce proinflammatory signals to bone marrow. In the present study, increased leucocyte and c-ESR levels in the primary and recurrent pterygium groups suggested chronic inflammation in the ocular surface of pterygium patients.
The selective migration and recruitment of EPCs into pterygial tissues may be mediated via their interactions with specific chemokines. VEGF is a well-known chemoattractant for CD34+ MNCs, and plays an important role in the haematopoietic stem cell trafficking between the peripheral circulation and bone marrow.28 In the present study, we found that systemic and local VEGF levels were increased in pterygium groups, and statistical analysis revealed that plasma and tear VEGF levels were strongly correlated with circulating CD34+ MNCs in the pterygium groups. Subsequent histological evaluations showed a strong positive reaction to VEGF in pterygial tissue, which implies that VEGF has a chemoattractant effect. It seems likely that in addition to VEGF, SP might stimulate the homing and recruitment of EPCs from bone marrow. A previous report suggested that SP acts as a proangiogenic signal, and found that when SP levels are attenuated, neovascularization does not occur.29 In our study, local and systemic SP levels were found to be higher in the pterygium groups, and to share a close relationship with CD34+ and c-kit+ MNCs. Thus, based on these observations, SP appears to affect vasculogenesis.
It is unclear whether the upregulation of circulating CD34+ and c-kit+ MNCs in our study occurred secondary to the direct stimulation of these chemotactic factors, such as VEGF and SP. Probably, the possibility that those MNCs are already at ocular tissue without these chemoattractants cannot be excluded. However, it is known that VEGF plays an important role in bone marrow reconstitution and upregulates stromal cell-derived factor-1 (SDF-1).30, 31 SDF-1 is known to mediate the mobilization and migration of bone marrow-derived progenitor cells in vivo.32 SP has also been shown to have a stimulatory effect on human bone marrow stem cells in vitro by interacting with bone marrow supporting stromal cells.33 Therefore, it is possible to speculate that an increase in VEGF and SP levels may stimulate bone marrow, and enhance progenitor cell mobilization.
Our findings also explain features of pterygium recurrence after surgical excision. CD34+ MNC counts were elevated in the pterygium groups, and were significantly higher in the recurrent pterygium group than in the primary pterygium group. Thus, it is possible to speculate that in recurrent pterygium, systemic and local influences on EPCs are stronger than in primary pterygium. Moreover, circulating c-kit+ MNC counts were increased, although they were less prominent than those of CD34+ MNCs in the recurrent pterygium group. C-kit+ MNCs reflect all stem cells including mesenchymal stem cells. Thus, it might be speculated that an imbalance between endothelial and mesenchymal progenitor cells causes excessive endothelial sprouting without mesenchymal architectural support.
To the best of our knowledge, this is the first study to demonstrate EPC involvement and to identify the systemic and local chemokines involved in the pathogenesis of pterygium. Our findings show that an ischaemic signal from the ocular surface, involving proinflammatory chemokines which are diffused into systemic circulation, may stimulate bone marrow, and that subsequently CD34 and c-kit+ MNCs are mobilized into peripheral blood and differentiate into pterygial endothelial cells on ischaemic foci. Moreover, this finding may describe a more general cellular response to ocular surface damage. Although many questions remain regarding the quantitative participation and the contribution made by bone marrow-derived progenitor cells to pterygial endothelial cells, our results indicate that the identification of such vasculogenic factors and EPCs may be of therapeutic utility in pterygium.
References
Coroneo MT, Di Girolamo N, Wakefield D . The pathogenesis of pterygia. Curr Opin Ophthalmol 1999; 10: 282–288.
Gazzard G, Saw SM, Farook M, Koh D, Widjaja D, Chia SE et al. Pterygium in Indonesia: prevalence, severity and risk factors. Br J Ophthalmol 2002; 86: 1341–1346.
Wong TY, Foster PJ, Johnson GJ, Seah SK, Tan DT . The prevalence and risk factors for pterygium in an adult Chinese population in Singapore: the Tanjong Pagar survey. Am J Ophthalmol 2001; 131: 176–183.
Kennedy M, Kim KH, Harten B, Brown J, Planck S, Meshul C et al. Ultraviolet irradiation induces the production of multiple cytokines by human corneal cells. Invest Ophthalmol Vis Sci 1997; 38: 2483–2491.
Nick G, Rakesb K, Minas T, Dennis W . UVB-mediated induction of IL-6 and 8 in pterygia and cultured human pterygium epithelial cells. Invest Ophthalmol Vis Sci 2002; 43: 3430–3477.
Kria L, Ohira A, Amemiya T . Growth factors in cultured pterygium fibroblasts: immunohistochemical and ELISA analysis. Graef Arch Clin Exp Ophthalmol 1998; 236: 702–708.
Risau W . Differentiation of endothelium. FASEB J 1999; 9: 926–933.
Shintani S, Murohara T, Ikeda H, Ueno T, Honma T, Katoh A et al. Mobilization of endothelial progenitor cells in patients with acute myocardial infarction. Circulation 2001; 103: 2776–2779.
Peters BA, Diaz LA, Polyak K, Meszler L, Romans K, Guinan EC et al. Contribution of bone marrow-derived endothelial cells to human tumor vasculature. Nat Med 2005; 11: 261–262.
Hess DC, Hill WD, Martin-Studdard A, Carroll J, Brailer J, Carothers J . Bone marrow as a source of endothelial cells and NeuN-expressing cells after stroke. Stroke 2002; 33: 1362–1368.
Peichev M, Naiyer AJ, Pereira D, Zhu Z, Lane WJ, Williams M et al. Expression of VEGFR-2 and AC133 by circulating human CD34+ cells identifies a population of functional endothelial precursors. Blood 2000; 95: 952–958.
Asahara T, Masuda H, Takahashi T, Kalka C, Pastore C, Silver M et al. Bone marrow origin of endothelial progenitor cells responsible for postnatal vasculogenesis in physiological and pathological neovascularization. Cir Res 1999; 85: 221–228.
de Vries P, Brasel KA, Eisenman JR, Alpert AR, Williams DE . The effect of recombinant mast cell growth factor on purified murine hematopoietic stem cells. J Exp Med 1991; 173: 1205–1211.
Rameshwar P, Ganea D, Gascon P . In vitro stimulatory effect of substance P on hematopoiesis. Blood 1993; 81: 391–398.
Noack F, Sotlar K, Thorns C, Smrcek J, Diedrich K, Feller AC et al. VEGF-, KIT protein- and neutral endopeptidase (NEP/CD10)-positive myofibroblasts—precursors of angiogenesis in chorioangiomas? Placenta 2003; 24: 758–766.
Rameshwar P, Zhu G, Donnelly RJ, Qian J, Ge H, Goldstein KR et al. The dynamics of bone marrow stromal cells in the proliferation of multipotent hematopoietic progenitors by substance P: an understanding of the effects of a neurotransmitter on the differentiating hematopoietic stem cell. J Neuroimmunol 2001; 121: 22–31.
Pelletier L, Angonin R, Regnard J, Fellmann D, Charbord P . Human bone marrow angiogenesis: in vitro modulation by substance P and neurokinin A. Br J Haematol 2002; 119: 1083–1089.
Nagano T, Nakamura M, Nakata K, Yamaguchi T, Takase K, Okahara A et al. Effects of substance P and IGF-1 in corneal epithelial barrier function and wound healing in a rat model of neurotrophic keratopathy. Invest Ophthalmol Vis Sci 2003; 44: 3810–3815.
Murphy CJ, Marfurt CF, McDermott A, Bentley E, Abrams GA, Reid TW et al. Spontaneous chronic corneal epithelial defects (SCCED) in dogs: clinical features, innervation, and effects of topical substance-P with or without IGF-1. Invest Ophthalmol Vis Sci 2001; 42: 2252–2261.
Kria L, Ohira A, Amemiya T . Immunohistochemical localization of basic fibroblast growth factor, platelet derived growth factor, transforming growth factor-beta and tumor necrosis factor-alpha in the pterygium. Acta Histochem 1996; 98: 195–201.
Nakagami T, Watanabe I, Murakami A, Okisaka S, Ebihara N . Expression of stem cell factor in pterygium. Jpn J Ophthalmol 2000; 44: 193–197.
Song YS, Ryu YH, Choi SR, Kim JC . The involvement of adult stem cells originated from bone marrow in the pathogenesis of pterygia. Yonsei Med J 2005; 46: 687–692.
Jones DT, Monroy D, Pflugfelder SC . A novel method of tear collection: comparison of transorb TM polyster rods and glass capillary micropipettes. Invest Ophthalmol Vis Sci 1996; 37: 3935, S851.
Chan CM, Chew PW, Alsagoff Z, Wong JS, Tan DT . Vascular patterns in pterygium and conjunctival autografting: a pilot study using indocyanine green anterior segment angiography. Br J Ophthalmol 2001; 85: 350–353.
Maloof PB, Joshi DD, Qian J, Gascón P, Singh D, Rameshwar P . Induction of preprotachykinin-I and neurokinin-1 by adrenocorticotropin and prolactin: implication for neuroendocrine-immune-hematopoietic axis. J Neuroimmunol 2001; 112: 188–196.
Pereira HA, Shafer WM, Pohl J, Martin LE, Spitznagel JK . CAP37, a human neutrophil-derived chemotactic factor with monocyte specific activity. J Clin Invest 1990; 85: 1468–1476.
Seifert P, Sekundo W . Capillaries in the epithelium of pterygium. Br J Ophthalmol 1998; 82: 77–81.
Gill M, Dias S, Hattori K, Rivera ML, Hicklin D, Witte L et al. Vascular trauma induces rapid but transient mobilization of VEGFR2(+)AC133(+) endothelial precursor cells. Circ Res 2001; 88: 167–174.
Troger J, Neyer S, Heufler C, Huemer H, Schmid E, Griesser U et al. Substance P and vasoactive intestinal polypeptide in the streptozotocin-induced diabetic rat retina. Invest Ophthalmol Vis Sci 2001; 42: 1045–1050.
Salcedo R, Wasserman K, Young HA, Grimm MC, Howard OM, Anver MR et al. Vascular endothelial growth factor and basic fibroblast growth factor induce expression of CXCR4 on human endothelial cells: in vivo neovascularization induced by stromal-derived factor-1alpha. Am J Pathol 1999; 154: 1125–1135.
Grunewald M, Avraham I, Dor Y, Bachar-Lustig E, Itin A, Yung S et al. VEGF-induced adult neovascularization: recruitment, retention, and role of accessory cells. Cell 2006; 124: 175–189.
Sweeney EA, Lortat-Jacob H, Priestley GV, Nakamoto B, Papayannopoulou T . Sulfated polysaccharides increase plasma levels of SDF-1 in monkeys and mice: involvement in mobilization of stem/progenitor cells. Blood 2002; 99: 44–51.
Hiramoto M, Aizawa S, Iwase O, Nakano M, Toyama K, Hoque M et al. Stimulatory effects of substance P on CD34 positive cell proliferation and differentiation in vitro are mediated by the modulation of stromal cell function. Int J Mol Med 1998; 1: 347–354.
Acknowledgements
This research was partly supported by research funds from the Research Institute for Biomedical and Pharmaceutical Science at Chung-Ang University, Seoul, Republic of Korea, and by research funds (code: stem cell 13132) from the Stem Cell Research Center of the 21C frontier research program funded by the Korean Ministry of Science and Technology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, J., Song, Y., Ha, H. et al. Endothelial progenitor cells in pterygium pathogenesis. Eye 21, 1186–1193 (2007). https://doi.org/10.1038/sj.eye.6702433
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.eye.6702433
- Springer Nature Limited
Keywords
This article is cited by
-
The effect of cyclosporine a in pterygium surgery using fibrin glue
International Ophthalmology (2024)
-
Does systemic inflammation play a role in patients with pterygium?
International Ophthalmology (2020)
-
Management of primary pterygium with intra-lesional injection of 5 flurouracil and bevacizumab (Avastin)
Eye (2019)