Abstract
Although the impact of the gut microbiome on health and disease is well established, there is controversy regarding the presence of microorganisms such as bacteria and their products in organs and tissues. However, recent contamination-aware findings of tissue-resident microbial signatures provide accumulating evidence in support of bacterial translocation in cardiometabolic disease. The latter provides a distinct paradigm for the link between microbial colonizers of mucosal surfaces and host metabolism. In this Perspective, we re-evaluate the concept of tissue-resident bacteria including their role in metabolic low-grade tissue and systemic inflammation. We examine the limitations and challenges associated with studying low bacterial biomass samples and propose experimental and analytical strategies to overcome these issues. Our Perspective aims to encourage further investigation of the mechanisms linking tissue-resident bacteria to host metabolism and their potentially actionable health implications for prevention and treatment.




Similar content being viewed by others
References
Hou, K. et al. Microbiota in health and diseases. Sig Transduct. Target Ther. 7, 135 (2022).
Fan, Y. & Pedersen, O. Gut microbiota in human metabolic health and disease. Nat. Rev. Microbiol. 19, 55–71 (2021).
Chakaroun, R. M., Massier, L. & Kovacs, P. Gut microbiome, intestinal permeability, and tissue bacteria in metabolic disease: perpetrators or bystanders? Nutrients 12, 1082 (2020).
Hotamisligil, G. S. Inflammation, metaflammation and immunometabolic disorders. Nature 542, 177–185 (2017).
Cani, P. D. et al. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes 57, 1470–1481 (2008).
Bleau, C., Karelis, A. D., St-Pierre, D. H. & Lamontagne, L. Crosstalk between intestinal microbiota, adipose tissue and skeletal muscle as an early event in systemic low-grade inflammation and the development of obesity and diabetes. Diabetes Metab. Res. Rev. 31, 545–561 (2015).
Raybould, H. E. Gut microbiota, epithelial function and derangements in obesity. J. Physiol. 590, 441–446 (2012).
Koppel, N., Rekdal, V. M. & Balskus, E. P. Chemical transformation of xenobiotics by the human gut microbiota. Science 356, eaag2770 (2018).
Amar, J. et al. Involvement of tissue bacteria in the onset of diabetes in humans: evidence for a concept. Diabetologia 54, 3055–3061 (2011).
Adami, J. G. On the bactericidal functions of the liver and the etiology of progressive hepatic cirrhosis. BMJ 2, 1215–1214 (1898).
Nicholls, A. G. A simple method of demonstrating the presence of bacteria in the mesentery of normal animals. J. Med. Res. 11, 455–468.1 (1904).
Lloyd-Price, J., Abu-Ali, G. & Huttenhower, C. The healthy human microbiome. Genome Med. 8, 51 (2016).
Bäckhed, F. Programming of host metabolism by the gut microbiota. Ann. Nutr. Metab. 58, 44–52 (2011).
Tan, C. C. S. et al. No evidence for a common blood microbiome based on a population study of 9,770 healthy humans. Nat. Microbiol. https://doi.org/10.1038/s41564-023-01350-w (2023).
Païssé, S. et al. Comprehensive description of blood microbiome from healthy donors assessed by 16S targeted metagenomic sequencing. Transfusion 56, 1138–1147 (2016).
Castillo, D. J., Rifkin, R. F., Cowan, D. A. & Potgieter, M. The healthy human blood microbiome: fact or fiction? Front. Cell. Infect. Microbiol. 9, 148 (2019).
McLaughlin, R. W. et al. Are there naturally occurring pleomorphic bacteria in the blood of healthy humans? J. Clin. Microbiol. 40, 4771–4775 (2002).
Villarroel, J., Donkin, I., Champion, C., Burcelin, R. & Barrès, R. Endurance training in humans modulates the bacterial DNA signature of skeletal muscle. Biomedicines 10, 64 (2021).
Ha, C. W. Y. et al. Translocation of viable gut microbiota to mesenteric adipose drives formation of cree** fat in humans. Cell 183, 666–683 (2020).
Balmer, M. L. et al. The liver may act as a firewall mediating mutualism between the host and its gut commensal microbiota. Sci. Transl. Med. 6, 237ra66 (2014).
Caly, W. R. & Strauss, E. A prospective study of bacterial infections in patients with cirrhosis. J. Hepatol. 18, 353–358 (1993).
Duan, Y. et al. Bacteriophage targeting of gut bacterium attenuates alcoholic liver disease. Nature 575, 505–511 (2019).
Anhê, F. F. et al. Type 2 diabetes influences bacterial tissue compartmentalisation in human obesity. Nat. Metab. 2, 233–242 (2020).
Sookoian, S. et al. Intrahepatic bacterial metataxonomic signature in non-alcoholic fatty liver disease. Gut 69, 1483–1491 (2020).
Udayan, S. et al. Identification of gut bacteria such as Lactobacillus johnsonii that disseminate to systemic tissues of wild type and MyD88–/– mice. Gut Microbes 14, 2007743 (2022).
Lluch, J. et al. The characterization of novel tissue microbiota using an optimized 16S metagenomic sequencing pipeline. PLoS ONE 10, e0142334 (2015).
Amar, J. et al. Intestinal mucosal adherence and translocation of commensal bacteria at the early onset of type 2 diabetes. EMBO Mol. Med. 3, 559–572 (2011).
Udayappan, S. D. et al. Intestinal Ralstonia pickettii augments glucose intolerance in obesity. PLoS ONE 12, e0181693 (2017).
Eisenhofer, R. et al. Contamination in low microbial biomass microbiome studies: issues and recommendations. Trends Microbiol. 27, 105–117 (2019).
He, Z. et al. Microbiota in mesenteric adipose tissue from Crohn’s disease promote colitis in mice. Microbiome 9, 228 (2021).
Massier, L. et al. Adipose tissue derived bacteria are associated with inflammation in obesity and type 2 diabetes. Gut 69, 1796–1806 (2020).
Sun, J. et al. The visceral adipose tissue bacterial microbiota provides a signature of obesity based on inferred metagenomic functions. Int. J. Obes. 47, 1008–1022 (2023).
Cicalese, L. et al. Acute pancreatitis and bacterial translocation. Dig. Dis. Sci. 46, 1127–1132 (2001).
Dervenis, C., Smailis, D. & Hatzitheoklitos, E. Bacterial translocation and its prevention in acute pancreatitis. J. Hepatobiliary Pancreat. Surg. 10, 415–418 (2003).
Miklossy, J., Martins, R., Darbinian, N., Khalili, K. & McGeer, P. L. Type 2 diabetes: local inflammation and direct effect of bacterial toxic components. Open Pathol. J. 2, 86–95 (2008).
Gao, H. et al. Accumulation of microbial DNAs promotes to islet inflammation and β cell abnormalities in obesity in mice. Nat. Commun. 13, 565 (2022).
Liu, X. -Y. et al. Kidney microbiota dysbiosis contributes to the development of hypertension. Gut Microbes 14, 2143220 (2022).
Errington, J. L-form bacteria, cell walls and the origins of life. Open Biol. 3, 120143 (2013).
Zulian, A. et al. Adipose tissue microbiota in humans. Int. J. Obes. 40, 1643–1648 (2016).
Giron, L. B. et al. Markers of fungal translocation are elevated during post-acute sequelae of SARS-CoV-2 and induce NF-κB signaling. JCI Insight 7, e160989 (2022).
Dominy, S. S. et al. Porphyromonas gingivalis in Alzheimer’s disease brains: Evidence for disease causation and treatment with small-molecule inhibitors. Sci. Adv. 5, eaau3333 (2019).
Schmidt, T. S. et al. Extensive transmission of microbes along the gastrointestinal tract. Elife 8, e42693 (2019).
Gummesson, A. et al. Intestinal permeability is associated with visceral adiposity in healthy women. Obesity 19, 2280–2282 (2011).
Teixeira, T. F. S. et al. Intestinal permeability parameters in obese patients are correlated with metabolic syndrome risk factors. Clin. Nutr. 31, 735–740 (2012).
Luther, J. et al. Hepatic injury in nonalcoholic steatohepatitis contributes to altered intestinal permeability. Cell. Mol. Gastroenterol. Hepatol. 1, 222–232 (2015).
Michielan, A. & D’Incà, R. Intestinal permeability in inflammatory bowel disease: pathogenesis, clinical evaluation, and therapy of leaky gut. Mediators Inflamm. 2015, 628157 (2015).
Bruewer, M. et al. Proinflammatory cytokines disrupt epithelial barrier function by apoptosis-independent mechanisms. J. Immunol. 171, 6164–6172 (2003).
Kanaya, T., Williams, I. R. & Ohno, H. Intestinal M cells: tireless samplers of enteric microbiota. Traffic 21, 34–44 (2020).
Regoli, M., Borghesi, C., Bertelli, E. & Nicoletti, C. Uptake of a gram-positive bacterium (Streptococcus pneumoniae R36a) by the M cells of rabbit Peyer’s patches. Ann. Anat. 177, 119–124 (1995).
Guerville, M. & Boudry, G. Gastrointestinal and hepatic mechanisms limiting entry and dissemination of lipopolysaccharide into the systemic circulation. Am. J. Physiol. Gastrointest. Liver Physiol. 311, G1–G15 (2016).
Dean, P. & Kenny, B. Intestinal barrier dysfunction by enteropathogenic Escherichia coli is mediated by two effector molecules and a bacterial surface protein. Mol. Microbiol. 54, 665–675 (2004).
Caron, T. J., Scott, K. E., Fox, J. G. & Hagen, S. J. Tight junction disruption: Helicobacter pylori and dysregulation of the gastric mucosal barrier. World J. Gastroenterol. 21, 11411–11427 (2015).
Rossi, O., van Baarlen, P. & Wells, J. M. Host-recognition of pathogens and commensals in the mammalian intestine. in Between Pathogenicity and Commensalism (eds Dobrindt, U. et al.) 291–321 (Springer, 2013). https://doi.org/10.1007/82_2011_191
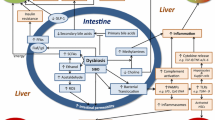
Massier, L., Blüher, M., Kovacs, P. & Chakaroun, R. M. Impaired intestinal barrier and tissue bacteria: pathomechanisms for metabolic diseases. Front. Endocrinol. 12, 616506 (2021).
Brandl, K., Kumar, V. & Eckmann, L. Gut-liver axis at the frontier of host–microbial interactions. Am. J. Physiol. Gastrointest. Liver Physiol. 312, G413–G419 (2017).
Broadley, S. P. et al. Dual-track clearance of circulating bacteria balances rapid restoration of blood sterility with induction of adaptive immunity. Cell Host Microbe 20, 36–48 (2016).
Zeng, Z. et al. CRIg functions as a macrophage pattern recognition receptor to directly bind and capture blood-borne gram-positive bacteria. Cell Host Microbe 20, 99–106 (2016).
Levels, J. H., Abraham, P. R., van den Ende, A. & van Deventer, S. J. Distribution and kinetics of lipoprotein-bound endotoxin. Infect. Immun. 69, 2821–2828 (2001).
Takeshita, S., Nakatani, K., Tsujimoto, H., Kawamura, Y. & Sekine, I. Detection of circulating lipopolysaccharide-bound monocytes in children with gram-negative sepsis. J. Infect. Dis. 182, 1549–1552 (2000).
Roth, R. I., Levin, F. C. & Levin, J. Distribution of bacterial endotoxin in human and rabbit blood and effects of stroma-free hemoglobin. Infect. Immun. 61, 3209–3215 (1993).
Poschl, J. M. B., Leray, C., Ruef, P., Cazenave, J. P. & Linderkamp, O. Endotoxin binding to erythrocyte membrane and erythrocyte deformability in human sepsis and in vitro. Crit. Care Med. 31, 924–928 (2003).
Stahl, A. et al. Lipopolysaccharide from enterohemorrhagic Escherichia coli binds to platelets through TLR4 and CD62 and is detected on circulating platelets in patients with hemolytic uremic syndrome. Blood 108, 167–176 (2006).
Yao, Z. et al. Blood-borne lipopolysaccharide is rapidly eliminated by liver sinusoidal endothelial cells via high-density lipoprotein. J. Immunol. 197, 2390–2399 (2016).
Topchiy, E. et al. Lipopolysaccharide is cleared from the circulation by hepatocytes via the low density lipoprotein receptor. PLoS ONE 11, e0155030 (2016).
Hanslin, K. et al. The impact of the systemic inflammatory response on hepatic bacterial elimination in experimental abdominal sepsis. Intensive Care Med. Exp. 7, 52 (2019).
Queck, A. et al. Role of portal venous platelet activation in patients with decompensated cirrhosis and TIPS. Gut 69, 1535–1536 (2020).
Chelakkot, C., Ghim, J. & Ryu, S. H. Mechanisms regulating intestinal barrier integrity and its pathological implications. Exp. Mol. Med. 50, 1–9 (2018).
Bischoff, S. C. et al. Intestinal permeability—a new target for disease prevention and therapy. BMC Gastroenterol. 14, 189 (2014).
Cortez-Gonzalez, X. et al. TLR9-independent activation of B lymphocytes by bacterial DNA. DNA Cell Biol. 25, 253–261 (2006).
Alvarez, M. E. et al. Neutrophil signaling pathways activated by bacterial DNA stimulation. J. Immunol. 177, 4037–4046 (2006).
Trevani, A. S. et al. Bacterial DNA activates human neutrophils by a CpG-independent pathway. Eur. J. Immunol. 33, 3164–3174 (2003).
Lin, Y. et al. The lipopolysaccharide-activated Toll-like receptor (TLR)-4 induces synthesis of the closely related receptor TLR-2 in adipocytes. J. Biol. Chem. 275, 24255–24263 (2000).
Seki, E., Park, E. & Fujimoto, J. Toll-like receptor signaling in liver regeneration, fibrosis and carcinogenesis. Hepatol. Res. 41, 597–610 (2011).
Garay-Malpartida, H. M. et al. Toll-like receptor 4 (TLR4) expression in human and murine pancreatic beta-cells affects cell viability and insulin homeostasis. BMC Immunol. 12, 18 (2011).
Reyna, S. M. et al. Elevated Toll-like receptor 4 expression and signaling in muscle from insulin-resistant subjects. Diabetes 57, 2595–2602 (2008).
Liang, H., Hussey, S. E., Sanchez-Avila, A., Tantiwong, P. & Musi, N. Effect of lipopolysaccharide on inflammation and insulin action in human muscle. PLoS ONE 8, e63983 (2013).
Frisard, M. I. et al. Toll-like receptor 4 modulates skeletal muscle substrate metabolism. Am. J. Physiol. Endocrinol. Metab. 298, E988–E998 (2010).
Frisard, M. I. et al. Low levels of lipopolysaccharide modulate mitochondrial oxygen consumption in skeletal muscle. Metab. Clin. Exp. 64, 416–427 (2015).
Amyot, J., Semache, M., Ferdaoussi, M., Fontes, G. & Poitout, V. Lipopolysaccharides impair insulin gene expression in isolated islets of Langerhans via Toll-like receptor-4 and NF-κB signalling. PLoS ONE 7, e36200 (2012).
He, W. et al. TLR4 triggered complex inflammation in human pancreatic islets. Biochim. Biophys. Acta Mol. Basis Dis. 1865, 86–97 (2019).
Vives-Pi, M. et al. Evidence of expression of endotoxin receptors CD14, Toll-like receptors TLR4 and TLR2 and associated molecule MD-2 and of sensitivity to endotoxin (LPS) in islet beta cells. Clin. Exp. Immunol. 133, 208–218 (2003).
Vila, I. K. et al. Immune cell Toll-like receptor 4 mediates the development of obesity- and endotoxemia-associated adipose tissue fibrosis. Cell Rep. 7, 1116–1129 (2014).
Chiadak, J. D. et al. Lipopolysaccharide modifies glycerol permeability and metabolism in 3T3-L1 adipocytes. Int. J. Mol. Sci. 18, 2566 (2017).
Choi, Y. et al. Gut microbe-derived extracellular vesicles induce insulin resistance, thereby impairing glucose metabolism in skeletal muscle. Sci. Rep. 5, 15878 (2015).
Liu, W., Yang, G., Liu, P., Jiang, X. & **n, Y. Modulation of adipose tissue metabolism by microbial-derived metabolites. Front. Microbiol. 13, 1031498 (2022).
Spivak, I., Fluhr, L. & Elinav, E. Local and systemic effects of microbiome-derived metabolites. EMBO Rep. 23, e55664 (2022).
Lauder, A. P. et al. Comparison of placenta samples with contamination controls does not provide evidence for a distinct placenta microbiota. Microbiome 4, 29 (2016).
Kennedy, K. M. et al. Questioning the fetal microbiome illustrates pitfalls of low-biomass microbial studies. Nature 613, 639–649 (2023).
Olomu, I. N. et al. Elimination of ‘kitome’ and ‘splashome’ contamination results in lack of detection of a unique placental microbiome. BMC Microbiol. 20, 157 (2020).
Read, S. J. Recovery efficiencies of nucleic acid extraction kits as measured by quantitative LightCyclerTM PCR. Mol. Pathol. 54, 86–90 (2001).
Polz, M. F. & Cavanaugh, C. M. Bias in template-to-product ratios in multitemplate PCR. Appl. Environ. Microbiol 64, 3724–3730 (1998).
Marchesi, J. R. et al. Design and evaluation of useful bacterium-specific PCR primers that amplify genes coding for bacterial 16S rRNA. Appl. Environ. Microbiol. 64, 795–799 (1998).
Novitsky, T. J., Schmidt-Gengenbach, J. & Remillard, J. F. Factors affecting recovery of endotoxin adsorbed to container surfaces. J. Parenter. Sci. Technol. 40, 284–286 (1986).
Boutagy, N. E., McMillan, R. P., Frisard, M. I. & Hulver, M. W. Metabolic endotoxemia with obesity: is it real and is it relevant? Biochimie 124, 11–20 (2016).
Hurley, J. C. Endotoxemia: methods of detection and clinical correlates. Clin. Microbiol. Rev. 8, 268–292 (1995).
Erb-Downward, J. R. et al. Critical relevance of stochastic effects on low-bacterial-biomass 16S rRNA gene analysis. mBio 11, e00258-20 (2020).
Karstens, L. et al. Controlling for contaminants in low-biomass 16S rRNA gene sequencing experiments. mSystems https://doi.org/10.1128/msystems.00290-19 (2019).
Berry, D. et al. Phylotype-level 16S rRNA analysis reveals new bacterial indicators of health state in acute murine colitis. ISME J. 6, 2091–2106 (2012).
Batani, G., Bayer, K., Böge, J., Hentschel, U. & Thomas, T. Fluorescence in situ hybridization (FISH) and cell sorting of living bacteria. Sci. Rep. 9, 18618 (2019).
Young, A. P., Jackson, D. J. & Wyeth, R. C. A technical review and guide to RNA fluorescence in situ hybridization. PeerJ 8, e8806 (2020).
Ng, K. M. & Tropini, C. Visualization of gut microbiota-host interactions via fluorescence in situ hybridization, lectin staining, and imaging. J. Vis. Exp. https://doi.org/10.3791/62646 (2021).
Amann, R. & Fuchs, B. M. Single-cell identification in microbial communities by improved fluorescence in situ hybridization techniques. Nat. Rev. Microbiol. 6, 339–348 (2008).
Morono, Y. & Inagaki, F. Chapter three - analysis of low-biomass microbial communities in the deep biosphere. in Advances in Applied Microbiology (eds Sariaslani, S. et al.) vol. 95, 149–178 (Academic Press, 2016).
Schippers, A. et al. Prokaryotic cells of the deep sub-seafloor biosphere identified as living bacteria. Nature 433, 861–864 (2005).
Hoshino, T. et al. Atribacteria from the subseafloor sedimentary biosphere disperse to the hydrosphere through submarine mud volcanoes. Front. Microbiol. 8, 1135 (2017).
Chakaroun, R. M. et al. Circulating bacterial signature is linked to metabolic disease and shifts with metabolic alleviation after bariatric surgery. Genome Med. 13, 105 (2021).
Aagaard, K. et al. The placenta harbors a unique microbiome. Sci. Transl. Med. 6, 237ra65 (2014).
He, Q. et al. The meconium microbiota shares more features with the amniotic fluid microbiota than the maternal fecal and vaginal microbiota. Gut Microbes 12, 1794266 (2020).
Stinson, L. et al. Comparison of bacterial DNA profiles in mid-trimester amniotic fluid samples from preterm and term deliveries. Front. Microbiol. 11, 415 (2020).
Bolte, E. E., Moorshead, D. & Aagaard, K. M. Maternal and early life exposures and their potential to influence development of the microbiome. Genome Med. 14, 4 (2022).
Prince, A. L. et al. The placental membrane microbiome is altered among subjects with spontaneous preterm birth with and without chorioamnionitis. Am. J. Obstet. Gynecol. 214, 627.e1–627.e16 (2016).
Mishra, A. et al. Microbial exposure during early human development primes fetal immune cells. Cell 184, 3394–3409 (2021).
Rackaityte, E. et al. Viable bacterial colonization is highly limited in the human intestine in utero. Nat. Med. 26, 599–607 (2020).
Salter, S. J. et al. Reagent and laboratory contamination can critically impact sequence-based microbiome analyses. BMC Biol. 12, 87 (2014).
de Goffau, M. C. et al. Human placenta has no microbiome but can contain potential pathogens. Nature 572, 329–334 (2019).
Kennedy, K. M. et al. Fetal meconium does not have a detectable microbiota before birth. Nat. Microbiol. 6, 865–873 (2021).
Theis, K. R. et al. Does the human placenta delivered at term have a microbiota? Results of cultivation, quantitative real-time PCR, 16S rRNA gene sequencing, and metagenomics. Am. J. Obstet. Gynecol. 220, 267.e1–267.e39 (2019).
Blaser, M. J. et al. Lessons learned from the prenatal microbiome controversy. Microbiome 9, 8 (2021).
Bushman, F. D. De-discovery of the placenta microbiome. Am. J. Obstet. Gynecol. 220, 213–214 (2019).
Perez-Muñoz, M. E., Arrieta, M. -C., Ramer-Tait, A. E. & Walter, J. A critical assessment of the ‘sterile womb’ and ‘in utero colonization’ hypotheses: implications for research on the pioneer infant microbiome. Microbiome 5, 48 (2017).
Mirzayi, C. et al. Reporting guidelines for human microbiome research: the STORMS checklist. Nat. Med. 27, 1885–1892 (2021).
Davis, N. M., Proctor, D. M., Holmes, S. P., Relman, D. A. & Callahan, B. J. Simple statistical identification and removal of contaminant sequences in marker-gene and metagenomics data. Microbiome 6, 226 (2018).
Musat, N., Musat, F., Weber, P. K. & Pett-Ridge, J. Tracking microbial interactions with NanoSIMS. Curr. Opin. Biotechnol. 41, 114–121 (2016).
Berry, D. & Loy, A. Stable-isotope probing of human and animal microbiome function. Trends Microbiol. 26, 999–1007 (2018).
Hatzenpichler, R., Krukenberg, V., Spietz, R. L. & Jay, Z. J. Next-generation physiology approaches to study microbiome function at single cell level. Nat. Rev. Microbiol. 18, 241–256 (2020).
Lechene, C. P., Luyten, Y., McMahon, G. & Distel, D. L. Quantitative imaging of nitrogen fixation by individual bacteria within animal cells. Science 317, 1563–1566 (2007).
Berry, D. et al. Host-compound foraging by intestinal microbiota revealed by single-cell stable isotope probing. Proc. Natl Acad. Sci. USA 110, 4720–4725 (2013).
Guillermier, C., Poczatek, J. C., Taylor, W. R. & Steinhauser, M. L. Quantitative imaging of deuterated metabolic tracers in biological tissues with nanoscale secondary ion mass spectrometry. Int. J. Mass Spectrom. 422, 42–50 (2017).
Steinhauser, M. L. & Lechene, C. P. Quantitative imaging of subcellular metabolism with stable isotopes and multi-isotope imaging mass spectrometry. Semin. Cell Dev. Biol. 24, 661–667 (2013).
Decelle, J. et al. Subcellular chemical imaging: new avenues in cell biology. Trends Cell Biol. 30, 173–188 (2020).
Decelle, J. et al. Subcellular architecture and metabolic connection in the planktonic photosymbiosis between Collodaria (radiolarians) and their microalgae. Environ. Microbiol. 23, 6569–6586 (2021).
Gyngard, F., Trakimas, L. & Steinhauser, M. L. High-fidelity quantification of cell cycle activity with multi-isotope imaging mass spectrometry. in Cardiac Regeneration: Methods and Protocols (eds Poss, K. D. et al.) 257–268 (Springer, 2021). https://doi.org/10.1007/978-1-0716-0668-1_19
Musat, N. et al. A single-cell view on the ecophysiology of anaerobic phototrophic bacteria. Proc. Natl Acad. Sci. USA 105, 17861–17866 (2008).
Chakaroun, R., Massier, L., Musat, N. & Kovacs, P. New paradigms for familiar diseases: lessons learned on circulatory bacterial signatures in cardiometabolic diseases. Exp. Clin. Endocrinol. Diabetes 130, 313–326 (2022).
Hülpüsch, C. et al. Benchmarking MicrobIEM – a user-friendly tool for decontamination of microbiome sequencing data. BMC Biol. 21, 269 (2023).
Zhong, Z. -P. et al. Clean low-biomass procedures and their application to ancient ice core microorganisms. Front. Microbiol. 9, 1094 (2018).
Clokie, B. G. J. et al. Optimization of low-biomass sample collection and quantitative PCR-Based titration impact 16S rRNA microbiome resolution. Microbiol. Spectr. 10, e0225522 (2022).
Hasrat, R. et al. Benchmarking laboratory processes to characterise low-biomass respiratory microbiota. Sci. Rep. 11, 17148 (2021).
Prudent, E. & Raoult, D. Fluorescence in situ hybridization, a complementary molecular tool for the clinical diagnosis of infectious diseases by intracellular and fastidious bacteria. FEMS Microbiol Rev. 43, 88–107 (2019).
Narendra, D. P. & Steinhauser, M. L. Metabolic analysis at the nanoscale with multi-isotope imaging mass spectrometry (MIMS). Curr. Protoc. Cell Biol. 88, e111 (2020).
Morono, Y. Accessing the energy-limited and sparsely populated deep biosphere: achievements and ongoing challenges of available technologies. Prog. Earth Planet. Sci. 10, 18 (2023).
Vester, J. K., Glaring, M. A. & Stougaard, P. Improved cultivation and metagenomics as new tools for bioprospecting in cold environments. Extremophiles 19, 17–29 (2015).
Busch, W. Aus der Sitzung der medicinischen Section vom 13 November 1867. Berl. Klin. Wochenschr. 5, 137 (1868).
Nejman, D. et al. The human tumor microbiome is composed of tumor type-specific intracellular bacteria. Science 368, 973–980 (2020).
Poore, G. D. et al. Microbiome analyses of blood and tissues suggest cancer diagnostic approach. Nature 579, 567–574 (2020).
Ghaddar, B. et al. Tumor microbiome links cellular programs and immunity in pancreatic cancer. Cancer Cell 40, 1240–1253 (2022).
Narunsky-Haziza, L. et al. Pan-cancer analyses reveal cancer-type-specific fungal ecologies and bacteriome interactions. Cell 185, 3789–3806 (2022).
Dohlman, A. B. et al. A pan-cancer mycobiome analysis reveals fungal involvement in gastrointestinal and lung tumors. Cell 185, 3807–3822 (2022).
Debelius, J. W. et al. The local tumor microbiome is associated with survival in late-stage colorectal cancer patients. Microbiol. Spectr. 11, e0506622 (2023).
Chowdhury, S. et al. Programmable bacteria induce durable tumor regression and systemic antitumor immunity. Nat. Med. 25, 1057–1063 (2019).
Chen, Y. E. et al. Engineered skin bacteria induce antitumor T cell responses against melanoma. Science 380, 203–210 (2023).
Riquelme, E. et al. Tumor microbiome diversity and composition influence pancreatic cancer outcomes. Cell 178, 795–806 (2019).
Gihawi, A., Cooper, C. S. & Brewer, D. S. Caution regarding the specificities of pan-cancer microbial structure. Micro. Genom. 9, mgen001088 (2023).
Gihawi, A. et al. Major data analysis errors invalidate cancer microbiome findings. mBio 14, e0160723 (2023).
Offord, C. ‘Major errors’ alleged in landmark study that used microbes to identify cancers. https://www.science.org/content/article/major-errors-alleged-landmark-study-used-microbes-identify-cancers (2023).
Komiya, Y. et al. Patients with colorectal cancer have identical strains of Fusobacterium nucleatum in their colorectal cancer and oral cavity. Gut 68, 1335–1337 (2019).
Kartal, E. et al. A faecal microbiota signature with high specificity for pancreatic cancer. Gut 71, 1359–1372 (2022).
Chakaroun, R. M., Olsson, L. M. & Bäckhed, F. The potential of tailoring the gut microbiome to prevent and treat cardiometabolic disease. Nat. Rev. Cardiol. 20, 217–235 (2023).
Woo, A. Y. M. et al. Targeting the human gut microbiome with small-molecule inhibitors. Nat. Rev. Chem. https://doi.org/10.1038/s41570-023-00471-4 (2023).
Aldubayan, M. A. et al. Supplementation with inulin-type fructans affects gut microbiota and attenuates some of the cardiometabolic benefits of a plant-based diet in individuals with overweight or obesity. Front. Nutr. 10, 1108088 (2023).
Pedersen, S. S. et al. Targeted delivery of butyrate improves glucose homeostasis, reduces hepatic lipid accumulation and inflammation in db/db mice. Int. J. Mol. Sci. 24, 4533 (2023).
Horowitz, A., Chanez-Paredes, S. D., Haest, X. & Turner, J. R. Paracellular permeability and tight junction regulation in gut health and disease. Nat. Rev. Gastroenterol. Hepatol. https://doi.org/10.1038/s41575-023-00766-3 (2023).
Jensen, S. K. et al. Rewiring host-microbe interactions and barrier function during gastrointestinal inflammation. Gastroenterol. Rep. 10, goac008 (2022).
Huang, Z. et al. Antibody neutralization of microbiota-derived circulating peptidoglycan dampens inflammation and ameliorates autoimmunity. Nat. Microbiol. 4, 766–773 (2019).
Wheeler, R. et al. Microbiota-induced active translocation of peptidoglycan across the intestinal barrier dictates its within-host dissemination. Proc. Natl Acad. Sci. USA 120, e2209936120 (2023).
Ohto, U. Activation and regulation mechanisms of NOD-like receptors based on structural biology. Front Immunol. 13, 953530 (2022).
Acknowledgements
L.M. was supported by a postdoctoral grant from the Swedish Society for Medical Research (SSMF) and a starting grant from the Swedish Research Council (2023-02839). N.M. was funded by the Novo Nordisk Foundation through an NNF Young Investigator Award, grant NNF22OC0071609 ReFuel. R.C.’s research work was supported by a Walter Benjamin Fellowship grant from the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation). P.K.’s research work was supported by a grant from the DFG (Projektnummer 209933838 – SFB 1052; B3). We thank T.S. Schmidt (EMBL, Heidelberg, Germany) for rewarding scientific discussions and providing scientific material relating to the tumour microbiome section. We thank D.R. Marquez (@darwid_ilustration) for the contribution to our graphical illustrations.
Author information
Authors and Affiliations
Contributions
L.M., R.C. and P.K. designed the manuscript; L.M. drafted the figures; L.M., R.C., P.K. and N.M. contributed to the literature review; L.M., R.C., P.K. and N.M. wrote the manuscript; M.S. and V.T. edited and discussed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Metabolism thanks Andre Marette and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Massier, L., Musat, N., Stumvoll, M. et al. Tissue-resident bacteria in metabolic diseases: emerging evidence and challenges. Nat Metab (2024). https://doi.org/10.1038/s42255-024-01065-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42255-024-01065-0
- Springer Nature Limited