Abstract
Environmental factors, in particular viral infections, are thought to have an important role in the pathogenesis of type 1 diabetes mellitus (T1DM). The COVID-19 pandemic reinforced this hypothesis as many observational studies and meta-analyses reported a notable increase in the incidence of T1DM following infection with SARS-CoV-2 as well as an association between SARS-CoV-2 infection and the risk of new-onset T1DM. Experimental evidence suggests that human β-cells express SARS-CoV-2 receptors and that SARS-CoV-2 can infect and replicate in β-cells, resulting in structural or functional alterations of these cells. These alterations include reduced numbers of insulin-secreting granules, impaired pro-insulin (or insulin) secretion, and β-cell transdifferentiation or dedifferentiation. The inflammatory environment induced by local or systemic SARS-CoV-2 infection might result in a set of signals (such as pro-inflammatory cytokines) that lead to β-cell alteration or apoptosis or to a bystander activation of T cells and disruption of peripheral tolerance that triggers autoimmunity. Other mechanisms, such as viral persistence, molecular mimicry and activation of endogenous human retroviruses, are also likely to be involved in the pathogenesis of T1DM following SARS-CoV-2 infection. This Review addresses the issue of the involvement of SARS-CoV-2 infection in the development of T1DM using evidence from epidemiological, clinical and experimental studies.
Key points
-
Epidemiological studies and meta-analyses reported an increase in the incidence of type 1 diabetes mellitus during the COVID-19 pandemic as well as an association between SARS-CoV-2 infection and the risk of new-onset type 1 diabetes mellitus.
-
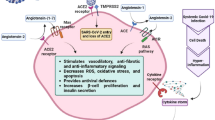
Angiotensin-converting enzyme 2 (ACE2), transmembrane serine protease 2 (TMPRSS2) and alternative receptors of SARS-CoV-2 have been identified in several human cell types, including pancreatic islet β-cells.
-
SARS-CoV-2 proteins are detected in both exocrine and endocrine cells (including β-cells) of post-mortem pancreas samples from patients with COVID-19 and in samples from non-human primates intranasally or intratracheally infected with SARS-CoV-2.
-
SARS-CoV-2 can infect and replicate in pancreatic β-cells ex vivo, resulting in structural and functional alterations of these cells.
-
Pro-inflammatory cytokines produced during SARS-CoV-2 infection can impair human pancreatic islet function.
-
SARS-CoV-2 infection might promote islet autoimmunity through various mechanisms, including bystander activation of T cells, disruption of peripheral tolerance, viral persistence, molecular mimicry and activation of endogenous human retroviruses.



Similar content being viewed by others
References
Atkinson, M. A., Eisenbarth, G. S. & Michels, A. W. Type 1 diabetes. Lancet 383, 69–82 (2014).
Roep, B. O. The role of T-cells in the pathogenesis of type 1 diabetes: from cause to cure. Diabetologia 46, 305–321 (2003).
Pociot, F. & Lernmark, Å. Genetic risk factors for type 1 diabetes. Lancet 387, 2331–2339 (2016).
Moltchanova, E. V., Schreier, N., Lammi, N. & Karvonen, M. Seasonal variation of diagnosis of type 1 diabetes mellitus in children worldwide. Diabet. Med. 26, 673–678 (2009).
Patterson, C. et al. Seasonal variation in month of diagnosis in children with type 1 diabetes registered in 23 European centers during 1989–2008: little short-term influence of sunshine hours or average temperature. Pediatr. Diabetes 16, 573–580 (2015).
Söderström, U., Aman, J. & Hjern, A. Being born in Sweden increases the risk for type 1 diabetes — a study of migration of children to Sweden as a natural experiment. Acta Paediatr. 101, 73–77 (2012).
Wang, Z., **e, Z., Lu, Q., Chang, C. & Zhou, Z. Beyond genetics: what causes type 1 diabetes. Clin. Rev. Allerg. Immunol. 52, 273–286 (2017).
Olmos, P. et al. The significance of the concordance rate for type 1 (insulin-dependent) diabetes in identical twins. Diabetologia 31, 747–750 (1988).
Peng, H. & Hagopian, W. Environmental factors in the development of type 1 diabetes. Rev. Endocr. Metab. Disord. 7, 149–162 (2007).
Patelarou, E. et al. Current evidence on the associations of breastfeeding, infant formula, and cow’s milk introduction with type 1 diabetes mellitus: a systematic review. Nutr. Rev. 70, 509–519 (2012).
Stene, L. C. & Gale, E. A. M. The prenatal environment and type 1 diabetes. Diabetologia 56, 1888–1897 (2013).
van der Werf, N., Kroese, F. G. M., Rozing, J. & Hillebrands, J.-L. Viral infections as potential triggers of type 1 diabetes. Diabetes Metab. Res. Rev. 23, 169–183 (2007).
Nekoua, M. P., Alid**ou, E. K. & Hober, D. Persistent coxsackievirus B infection and pathogenesis of type 1 diabetes mellitus. Nat. Rev. Endocrinol. 18, 503–516 (2022). This Review highlights evidence that persistent Coxsackievirus B infection triggers or accelerates islet autoimmunity and T1DM in susceptible individuals.
Soltesz, G., Patterson, C. & Dahlquist, G.; EURODIAB Study Group. Worldwide childhood type 1 diabetes incidence — what can we learn from epidemiology? Pediatr. Diabetes 8, 6–14 (2007).
Hober, D. & Sauter, P. Pathogenesis of type 1 diabetes mellitus: interplay between enterovirus and host. Nat. Rev. Endocrinol. 6, 279–289 (2010). The interplay between enterovirus infection with β-cells, the immune system and host genes in the pathogenesis of T1DM is discussed.
Lönnrot, M. et al. Respiratory infections are temporally associated with initiation of type 1 diabetes autoimmunity: the TEDDY study. Diabetologia 60, 1931–1940 (2017). A prospective study of 8,676 newborn babies reporting that viral respiratory infections are associated with an increased risk of islet autoimmunity.
Rasmussen, T., Witsø, E., Tapia, G., Stene, L. C. & Rønningen, K. S. Self-reported lower respiratory tract infections and development of islet autoimmunity in children with the type 1 diabetes high-risk HLA genotype: the MIDIA study. Diabetes Metab. Res. Rev. 27, 834–837 (2011).
Beyerlein, A., Wehweck, F., Ziegler, A.-G. & Pflueger, M. Respiratory infections in early life and the development of islet autoimmunity in children at increased type 1 diabetes risk: evidence from the BABYDIET study. JAMA Pediatr. 167, 800–807 (2013).
Mustonen, N. et al. Early childhood infections precede development of beta-cell autoimmunity and type 1 diabetes in children with HLA-conferred disease risk. Pediatr. Diabetes 19, 293–299 (2018).
Lee, H.-Y., Lu, C.-L., Chen, H.-F., Su, H.-F. & Li, C.-Y. Perinatal and childhood risk factors for early-onset type 1 diabetes: a population-based case-control study in Taiwan. Eur. J. Public Health 25, 1024–1029 (2015).
Ruiz, P. L. D. et al. Pandemic influenza and subsequent risk of type 1 diabetes: a nationwide cohort study. Diabetologia 61, 1996–2004 (2018).
Kordonouri, O. et al. Infections in the first year of life and development of beta cell autoimmunity and clinical type 1 diabetes in high-risk individuals: the TRIGR cohort. Diabetologia 65, 2098–2107 (2022).
Beyerlein, A., Donnachie, E., Jergens, S. & Ziegler, A.-G. Infections in early life and development of type 1 diabetes. JAMA 315, 1899–1901 (2016).
Wu, R. et al. Respiratory infections and type 1 diabetes: potential roles in pathogenesis. Rev. Med. Virol. 33, e2429 (2023).
Ho, D. et al. Identifying the lungs as a susceptible site for allele-specific regulatory changes associated with type 1 diabetes risk. Commun. Biol. 4, 1072 (2021).
Kim, D. et al. The architecture of SARS-CoV-2 transcriptome. Cell 181, 914–921 (2020).
Liu, Y.-C., Kuo, R.-L. & Shih, S.-R. COVID-19: the first documented coronavirus pandemic in history. Biomed. J. 43, 328–333 (2020).
Liu, J. et al. SARS-CoV-2 cell tropism and multiorgan infection. Cell Discov. 7, 17 (2021).
Unsworth, R. et al. New-onset type 1 diabetes in children during COVID-19: multicenter regional findings in the U.K. Diabetes Care 43, e170–e171 (2020).
Kamrath, C. et al. Incidence of type 1 diabetes in children and adolescents during the COVID-19 pandemic in Germany: results from the DPV registry. Diabetes Care 45, 1762–1771 (2022).
Gesuita, R. et al. Trends and cyclic variation in the incidence of childhood type 1 diabetes in two Italian regions over 33 years and during the COVID-19 pandemic. Diabetes Obes. Metab. 25, 1698–1703 (2023).
Rabbone, I. et al. Has COVID-19 delayed the diagnosis and worsened the presentation of type 1 diabetes in children? Diabetes Care 43, 2870–2872 (2020).
Rahmati, M. et al. The global impact of COVID‐19 pandemic on the incidence of pediatric new‐onset type 1 diabetes and ketoacidosis: a systematic review and meta‐analysis. J. Med. Virol. 94, 5112–5127 (2022).
D’Souza, D. et al. Incidence of diabetes in children and adolescents during the COVID-19 pandemic: a systematic review and meta-analysis. JAMA Netw. Open 6, e2321281 (2023).
Ruiz, P. L. D. et al. SARS-CoV-2 infection and subsequent risk of type 1 diabetes in 1.2 million children [abstract 233]. EASD 2022. https://www.easd.org/media-centre/home.html#!resources/sars-cov-2-infection-and-subsequent-risk-of-type-1-diabetes-in-1-2-million-children-3e68c8b2-dde1-43aa-8d75-af7356502994 (2022).
Kendall, E. K., Olaker, V. R., Kaelber, D. C., Xu, R. & Davis, P. B. Association of SARS-CoV-2 infection with new-onset type 1 diabetes among pediatric patients from 2020 to 2021. JAMA Netw. Open 5, e2233014 (2022).
Qeadan, F. et al. The associations between COVID-19 diagnosis, type 1 diabetes, and the risk of diabetic ketoacidosis: a nationwide cohort from the US using the Cerner Real-World Data. PLoS One 17, e0266809 (2022).
Lugar, M. et al. SARS-CoV-2 infection and development of islet autoimmunity in early childhood. JAMA 330, 1151–1160 (2023).
McKeigue, P. M. et al. Relation of incident type 1 diabetes to recent COVID-19 infection: cohort study using e-health record linkage in Scotland. Diabetes Care 46, 921–928 (2023).
Noorzae, R., Junker, T. G., Hviid, A. P., Wohlfahrt, J. & Olsen, S. F. Risk of type 1 diabetes in children is not increased after SARS-CoV-2 infection: a nationwide prospective study in Denmark. Diabetes Care 46, 1261–1264 (2023).
Messaaoui, A., Hajselova, L. & Tenoutasse, S. Anti-SARS-CoV-2 antibodies in new-onset type 1 diabetes in children during pandemic in Belgium. J. Pediatr. Endocrinol. Metab. 34, 1319–1322 (2021).
Jia, X. et al. Prevalence of SARS-CoV-2 antibodies in children and adults with type 1 diabetes. Diabetes Technol. Ther. 23, 517–521 (2021).
Rewers, M. et al. SARS-CoV-2 infections and presymptomatic type 1 diabetes autoimmunity in children and adolescents from Colorado, USA, and Bavaria, Germany. JAMA 328, 1252–1255 (2022).
Krischer, J. P. et al. SARS-CoV-2 — no increased islet autoimmunity or type 1 diabetes in teens. N. Engl. J. Med. 389, 474–475 (2023).
Pietropaolo, M., Hotez, P. & Giannoukakis, N. Incidence of an insulin-requiring hyperglycemic syndrome in SARS-CoV-2-infected young individuals: is it type 1 diabetes? Diabetes 71, 2656–2663 (2022).
Rahmati, M. et al. New-onset type 1 diabetes in children and adolescents as postacute sequelae of SARS-CoV-2 infection: a systematic review and meta-analysis of cohort studies. J. Med. Virol. 95, e28833 (2023).
Kamrath, C., Eckert, A. J., Holl, R. W. & Rosenbauer, J. Impact of the COVID-19 pandemic on children and adolescents with new-onset type 1 diabetes. Pediatr. Diabetes 2023, e7660985 (2023). This meta-analysis, including 40 studies carried out worldwide, reports an increased incidence of T1DM during the COVID-19 pandemic as well as an increased risk of T1DM following SARS-CoV-2 infection.
Zhang, T. et al. Risk for newly diagnosed diabetes after COVID-19: a systematic review and meta-analysis. BMC Med. 20, 444 (2022).
Lai, H. et al. Risk of incident diabetes after COVID-19 infection: a systematic review and meta-analysis. Metabolism 137, 155330 (2022).
Rubino, F. et al. New-onset diabetes in covid-19. N. Engl. J. Med. 383, 789–790 (2020).
Eslami, N. et al. SARS-CoV-2: receptor and co-receptor tropism probability. Curr. Microbiol. 79, 133 (2022).
Xu, H. et al. High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. Int. J. Oral. Sci. 12, 8 (2020).
Shang, J. et al. Structural basis of receptor recognition by SARS-CoV-2. Nature 581, 221–224 (2020).
Heurich, A. et al. TMPRSS2 and ADAM17 cleave ACE2 differentially and only proteolysis by TMPRSS2 augments entry driven by the severe acute respiratory syndrome coronavirus spike protein. J. Virol. 88, 1293–1307 (2014).
Li, Y. et al. The MERS-CoV receptor DPP4 as a candidate binding target of the SARS-CoV-2 spike. iScience 23, 101160 (2020).
Wang, S. et al. AXL is a candidate receptor for SARS-CoV-2 that promotes infection of pulmonary and bronchial epithelial cells. Cell Res. 31, 126–140 (2021).
Wang, K. et al. CD147-spike protein is a novel route for SARS-CoV-2 infection to host cells. Signal. Transduct. Target. Ther. 5, 283 (2020).
Cantuti-Castelvetri, L. et al. Neuropilin-1 facilitates SARS-CoV-2 cell entry and infectivity. Science 370, 856–860 (2020).
Shin, J., Toyoda, S., Fukuhara, A. & Shimomura, I. GRP78, a novel host factor for SARS-CoV-2: the emerging roles in COVID-19 related to metabolic risk factors. Biomedicines 10, 1995 (2022).
Lempp, F. A. et al. Lectins enhance SARS-CoV-2 infection and influence neutralizing antibodies. Nature 598, 342–347 (2021).
Kearns, F. L. et al. Spike-heparan sulfate interactions in SARS-CoV-2 infection. Curr. Opin. Struct. Biol. 76, 102439 (2022).
Amraei, R. et al. Extracellular vimentin is an attachment factor that facilitates SARS-CoV-2 entry into human endothelial cells. Proc. Natl Acad. Sci. USA 119, e2113874119 (2022).
Liao, Z. et al. Human transferrin receptor can mediate SARS-CoV-2 infection. Proc. Natl Acad. Sci. USA 121, e2317026121 (2024).
Wei, C. et al. HDL-scavenger receptor B type 1 facilitates SARS-CoV-2 entry. Nat. Metab. 2, 1391–1400 (2020).
Hoffmann, M. & Pöhlmann, S. Novel SARS-CoV-2 receptors: ASGR1 and KREMEN1. Cell Res. 32, 1–2 (2022).
Li, W. et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature 426, 450–454 (2003).
Lau, Y. L. & Peiris, J. M. Pathogenesis of severe acute respiratory syndrome. Curr. Opin. Immunol. 17, 404–410 (2005).
Hamming, I. et al. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 203, 631–637 (2004).
Roca-Ho, H., Riera, M., Palau, V., Pascual, J. & Soler, M. J. Characterization of ACE and ACE2 expression within different organs of the NOD mouse. Int. J. Mol. Sci. 18, 563 (2017).
Hikmet, F. et al. The protein expression profile of ACE2 in human tissues. Mol. Syst. Biol. 16, e9610 (2020).
Qi, J. et al. The scRNA-seq expression profiling of the receptor ACE2 and the cellular protease TMPRSS2 reveals human organs susceptible to SARS-CoV-2 infection. Int. J. Env. Res. Public Health 18, 284 (2021).
Fignani, D. et al. SARS-CoV-2 receptor angiotensin i-converting enzyme type 2 (ACE2) is expressed in human pancreatic β-cells and in the human pancreas microvasculature. Front. Endocrinol. 11, 596898 (2020).
Qadir, M. M. F. et al. SARS-CoV-2 infection of the pancreas promotes thrombofibrosis and is associated with new-onset diabetes. JCI Insight 6, e151551 (2023). SARS-CoV-2 infection is associated with thrombofibrosis of the pancreas in non-human primates and humans and is also associated with new-onset diabetes mellitus.
Kusmartseva, I. et al. Expression of SARS-CoV-2 entry factors in the pancreas of normal organ donors and individuals with COVID-19. Cell Metab. 32, 1041–1051 (2020).
Mawla, A. M. & Huising, M. O. Navigating the depths and avoiding the shallows of pancreatic islet cell transcriptomes. Diabetes 68, 1380–1393 (2019).
Wu, C.-T. et al. SARS-CoV-2 infects human pancreatic β cells and elicits β cell impairment. Cell Metab. 33, 1565–1576 (2021).
Liu, F. et al. ACE2 expression in pancreas may cause pancreatic damage after SARS-CoV-2 infection. Clin. Gastroenterol. Hepatol. 18, 2128–2130 (2020).
Müller, J. A. et al. SARS-CoV-2 infects and replicates in cells of the human endocrine and exocrine pancreas. Nat. Metab. 3, 149–165 (2021). SARS-CoV-2 can infect human pancreatic endocrine cells and impair β-cell function.
Kharchenko, P. V. The triumphs and limitations of computational methods for scRNA-seq. Nat. Methods 18, 723–732 (2021).
Aigha, I. I. & Abdelalim, E. M. NKX6.1 transcription factor: a crucial regulator of pancreatic β cell development, identity, and proliferation. Stem Cell Res. Ther. 11, 459 (2020).
Tang, X. et al. SARS-CoV-2 infection induces beta cell transdifferentiation. Cell Metab. 33, 1577–1591 (2021). SARS-CoV-2 antigens are detected in β-cells from pancreas necropsies of patients with COVID-19, and SARS-CoV-2 infection induces eIF2 pathway-mediated human β-cell transdifferentiation ex vivo.
van der Heide, V. et al. Limited extent and consequences of pancreatic SARS-CoV-2 infection. Cell Rep. 38, 110508 (2022).
Eizirik, D. L., Szymczak, F. & Mallone, R. Why does the immune system destroy pancreatic β-cells but not α-cells in type 1 diabetes? Nat. Rev. Endocrinol. 19, 425–434 (2023).
Ben Nasr, M. et al. Indirect and direct effects of SARS-CoV-2 on human pancreatic islets. Diabetes 71, 1579–1590 (2022). Inflammatory cytokines that are upregulated during COVID-19 in serum samples from patients can induce islet apoptosis.
Hodik, M. et al. Enterovirus infection of human islets of Langerhans affects β-cell function resulting in disintegrated islets, decreased glucose stimulated insulin secretion and loss of Golgi structure. BMJ Open Diabetes Res. Care 4, e000179 (2016).
Yao, Y. & Wang, C. Dedifferentiation: inspiration for devising engineering strategies for regenerative medicine. NPJ Regen. Med. 5, 14 (2020).
Bensellam, M., Jonas, J.-C. & Laybutt, D. R. Mechanisms of β-cell dedifferentiation in diabetes: recent findings and future research directions. J. Endocrinol. 236, R109–R143 (2018).
Talchai, C., Xuan, S., Lin, H. V., Sussel, L. & Accili, D. Pancreatic β cell dedifferentiation as a mechanism of diabetic β cell failure. Cell 150, 1223–1234 (2012).
Jopling, C., Boue, S. & Belmonte, J. C. I. Dedifferentiation, transdifferentiation and reprogramming: three routes to regeneration. Nat. Rev. Mol. Cell Biol. 12, 79–89 (2011).
Spaeth, J. M. et al. Defining a novel role for the Pdx1 transcription factor in islet β-cell maturation and proliferation during weaning. Diabetes 66, 2830–2839 (2017).
Fajgenbaum, D. C. & June, C. H. Cytokine storm. N. Engl. J. Med. 383, 2255–2273 (2020).
Jarczak, D. & Nierhaus, A. Cytokine storm — definition, causes, and implications. Int. J. Mol. Sci. 23, 11740 (2022).
Zanza, C. et al. Cytokine storm in COVID-19: immunopathogenesis and therapy. Medicina 58, 144 (2022).
Anindya, R., Rutter, G. A. & Meur, G. New-onset type 1 diabetes and severe acute respiratory syndrome coronavirus 2 infection. Immunol. Cell Biol. 101, 191–203 (2023).
Montefusco, L., Bolla, A. M. & Fiorina, P. Should we expect a wave of type 1 diabetes following SARS‐CoV‐2 pandemic? Diabetes Metab. Res. Rev. 38, e3529 (2022).
Sathish, T., Tapp, R. J., Cooper, M. E. & Zimmet, P. Potential metabolic and inflammatory pathways between COVID-19 and new-onset diabetes. Diabetes Metab. 47, 101204 (2021).
Shao, S., Yang, Q., Pan, R., Yu, X. & Chen, Y. Interaction of severe acute respiratory syndrome coronavirus 2 and diabetes. Front. Endocrinol. 12, 731974 (2021).
Coomans de Brachène, A. et al. Interferons are key cytokines acting on pancreatic islets in type 1 diabetes. Diabetologia 67, 908–927 (2024).
Millette, K. et al. SARS-CoV2 infects pancreatic beta cells in vivo and induces cellular and subcellular disruptions that reflect beta cell dysfunction. Preprint at Res. Sq. https://doi.org/10.21203/rs.3.rs-592374/v1 (2021). The effects of SARS-CoV-2 on β-cells are investigated in vivo using a rhesus macaque model.
Moudgil, K. D. & Sercarz, E. E. Crypticity of self antigenic determinants is the cornerstone of a theory of autoimmunity. Discov. Med. 5, 378–382 (2009).
Op de Beeck, A. & Eizirik, D. L. Viral infections in type 1 diabetes mellitus — why the β cells? Nat. Rev. Endocrinol. 12, 263–273 (2016).
Desimmie, B. A. et al. Insights into SARS-CoV-2 persistence and its relevance. Viruses 13, 1025 (2021).
Gousseff, M. et al. Clinical recurrences of COVID-19 symptoms after recovery: viral relapse, reinfection or inflammatory rebound? J. Infect. 81, 816–846 (2020).
Buonsenso, D. et al. Viral persistence in children infected with SARS-CoV-2: current evidence and future research strategies. Lancet Microbe 4, e745–e756 (2023).
Davis, H. E., McCorkell, L., Vogel, J. M. & Topol, E. J. Long COVID: major findings, mechanisms and recommendations. Nat. Rev. Microbiol. 21, 133–146 (2023).
de Melo, G. D. et al. COVID-19-related anosmia is associated with viral persistence and inflammation in human olfactory epithelium and brain infection in hamsters. Sci. Transl. Med. 13, eabf8396 (2021).
Yazdanpanah, N. & Rezaei, N. Autoimmune complications of COVID‐19. J. Med. Virol. 94, 54–62 (2022). This review highlights the development of several autoimmune diseases following SARS-CoV-2 infection.
Mehta, P. et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet 395, 1033–1034 (2020).
Montefusco, L. et al. Acute and long-term disruption of glycometabolic control after SARS-CoV-2. Infect. Nat. Metab. 3, 774–785 (2021).
Ehrenfeld, M. et al. Covid-19 and autoimmunity. Autoimmun. Rev. 19, 102597 (2020).
Swiecki, M. & Colonna, M. The multifaceted biology of plasmacytoid dendritic cells. Nat. Rev. Immunol. 15, 471–485 (2015).
Boddu, S. K., Aurangabadkar, G. & Kuchay, M. S. New onset diabetes, type 1 diabetes and COVID-19. Diabetes Metab. Syndr. 14, 2211–2217 (2020).
Pacheco, Y. et al. Bystander activation and autoimmunity. J. Autoimmun. 103, 102301 (2019).
Rahimi, K. Guillain-Barre syndrome during COVID-19 pandemic: an overview of the reports. Neurol. Sci. 41, 3149–3156 (2020).
Toubiana, J. et al. Kawasaki-like multisystem inflammatory syndrome in children during the covid-19 pandemic in Paris, France: prospective observational study. BMJ 369, m2094 (2020).
Mahévas, M. et al. Clinical characteristics, management and outcome of COVID-19-associated immune thrombocytopenia: a French multicentre series. Br. J. Haematol. 190, e224–e229 (2020).
Chen, M., Zhou, W. & Xu, W. Thyroid function analysis in 50 patients with COVID-19: a retrospective study. Thyroid 31, 8–11 (2021).
Galeotti, C. & Bayry, J. Autoimmune and inflammatory diseases following COVID-19. Nat. Rev. Rheumatol. 16, 413–414 (2020).
Akkuş, G. Newly-onset autoimmune diabetes mellitus triggered by COVID 19 infection: a case-based review. Endocr. Metab. Immune Disord. Drug Targets 23, 887–893 (2023).
Mishra, R., Elshimy, G., Kannan, L., Jacob, A. & Raj, R. Case report: SARS-CoV-2 infection as a trigger for diabetic ketoacidosis and newly detected pancreatic autoantibodies. Front. Endocrinol. 13, 983206 (2022).
Genç, S. et al. Could COVID-19 trigger type 1 diabetes? Presentation of covid-19 case presented with diabetic ketoacidosis. Acta Endocrinol. 17, 532–536 (2021).
Alfishawy, M., Nassar, M., Mohamed, M., Fatthy, M. & Elmessiery, R. M. New-onset type 1 diabetes mellitus with diabetic ketoacidosis and pancreatitis in a patient with COVID-19. Sci. Afr. 13, e00915 (2021).
Nielsen-Saines, K., Li, E., Olivera, A. M., Martin-Blais, R. & Bulut, Y. Case report: insulin-dependent diabetes mellitus and diabetic keto-acidosis in a child with COVID-19. Front. Pediatrics 9, 628810 (2021).
Marchand, L., Pecquet, M. & Luyton, C. Type 1 diabetes onset triggered by COVID-19. Acta Diabetol. 57, 1265–1266 (2020).
Gao, Z., Zhang, H., Liu, C. & Dong, K. Autoantibodies in COVID-19: frequency and function. Autoimmun. Rev. 20, 102754 (2021).
Wang, E. Y. et al. Diverse functional autoantibodies in patients with COVID-19. Nature 595, 283–288 (2021).
Coppieters, K. T., Wiberg, A. & Von Herrath, M. G. Viral infections and molecular mimicry in type 1 diabetes. APMIS 120, 941–949 (2012).
Lucchese, G. & Flöel, A. SARS-CoV-2 and Guillain-Barré syndrome: molecular mimicry with human heat shock proteins as potential pathogenic mechanism. Cell Stress. Chaperones 25, 731–735 (2020).
Moin, A. S. M., Nandakumar, M., Diane, A., Dehbi, M. & Butler, A. E. The role of heat shock proteins in type 1 diabetes. Front. Immunol. 11, 612584 (2021).
Quintana, F. J. & Cohen, I. R. The HSP60 immune system network. Trends Immunol. 32, 89–95 (2011).
Brudzynski, K. Insulitis-caused redistribution of heat-shock protein HSP60 inside β-cells correlates with induction of HSP60 autoantibodies. Diabetes 42, 908–913 (1993).
Raz, I. et al. Treatment of new-onset type 1 diabetes with peptide DiaPep277® is safe and associated with preserved beta-cell function: extension of a randomized, double-blind, phase II trial. Diabetes Metab. Res. Rev. 23, 292–298 (2007).
Cappello, F., Marino Gammazza, A., Dieli, F., Conway de Macario, E. & Macario, A. J. Does SARS-CoV-2 trigger stress-induced autoimmunity by molecular mimicry? A hypothesis. J. Clin. Med. 9, 2038 (2020).
Vahabi, M., Ghazanfari, T. & Sepehrnia, S. Molecular mimicry, hyperactive immune system, and SARS-COV-2 are three prerequisites of the autoimmune disease triangle following COVID-19 infection. Int. Immunopharmacol. 112, 109183 (2022).
Devaux, C. A. & Camoin-Jau, L. Molecular mimicry of the viral spike in the SARS-CoV-2 vaccine possibly triggers transient dysregulation of ACE2, leading to vascular and coagulation dysfunction similar to SARS-CoV-2 infection. Viruses 15, 1045 (2023).
Patrizio, A., Ferrari, S. M., Antonelli, A. & Fallahi, P. A case of Graves’ disease and type 1 diabetes mellitus following SARS-CoV-2 vaccination. J. Autoimmun. 125, 102738 (2021).
Sasaki, H. et al. Newly developed type 1 diabetes after coronavirus disease 2019 vaccination: a case report. J. Diabetes Investig. 13, 1105–1108 (2022).
Tang, X., He, B., Liu, Z., Zhou, Z. & Li, X. Fulminant type 1 diabetes after COVID-19 vaccination. Diabetes Metab. 48, 101324 (2022).
**ong, X. et al. Incidence of diabetes following COVID-19 vaccination and SARS-CoV-2 infection in Hong Kong: a population-based cohort study. PLoS Med. 20, e1004274 (2023).
Coate, K. C. et al. SARS-CoV-2 cell entry factors ACE2 and TMPRSS2 are expressed in the microvasculature and ducts of human pancreas but are not enriched in β cells. Cell Metab. 32, 1028–1040 (2020).
Garcia-Montojo, M. & Nath, A. HERV-W envelope expression in blood leukocytes as a marker of disease severity of COVID-19. eBioMedicine 67, 103363 (2021).
Levet, S. et al. An ancestral retroviral protein identified as a therapeutic target in type-1 diabetes. JCI Insight 2, e94387 (2017).
Levet, S. et al. Human endogenous retroviruses and type 1 diabetes. Curr. Diab Rep. 19, 141 (2019).
Balestrieri, E. et al. Evidence of the pathogenic HERV-W envelope expression in T lymphocytes in association with the respiratory outcome of COVID-19 patients. eBioMedicine 66, 103341 (2021).
Temerozo, J. R. et al. Human endogenous retrovirus K in the respiratory tract is associated with COVID-19 physiopathology. Microbiome 10, 65 (2022).
Halasa, N. B. et al. Maternal vaccination and risk of hospitalization for covid-19 among infants. N. Engl. J. Med. 387, 109–119 (2022).
Laguila Altoé, A. et al. Vaccine protection through placenta and breastfeeding: the unmet topic in COVID-19 pandemic. Front. Immunol. 13, 910138 (2022).
Rick, A.-M. et al. Impact of maternal SARS-CoV-2 booster vaccination on blood and breastmilk antibodies. PLoS One 18, e0287103 (2023).
**e, Y. & Al-Aly, Z. Risks and burdens of incident diabetes in long COVID: a cohort study. Lancet Diabetes Endocrinol. 10, 311–321 (2022).
Barrett, C. E. et al. Risk for newly diagnosed diabetes >30 days after SARS-CoV-2 infection among persons aged <18 years — United States, March 1, 2020–June 28, 2021. MMWR Morb. Mortal. Wkly Rep. 71, 59–65 (2022).
Naveed, Z. et al. Association of COVID-19 infection with incident diabetes. JAMA Netw. Open 6, e238866 (2023).
Reges, O. et al. Time-varying association of acute and post-acute COVID-19 with new-onset diabetes mellitus among hospitalized and non-hospitalized patients. BMJ Open Diabetes Res. Care 11, e003052 (2023).
Shrestha, D. B. et al. New-onset diabetes in COVID-19 and clinical outcomes: a systematic review and meta-analysis. World J. Virol. 10, 275–287 (2021).
Sathish, T., Kapoor, N., Cao, Y., Tapp, R. J. & Zimmet, P. Proportion of newly diagnosed diabetes in COVID-19 patients: a systematic review and meta-analysis. Diabetes Obes. Metab. 23, 870–874 (2021).
Banerjee, M., Pal, R. & Dutta, S. Risk of incident diabetes post-COVID-19: a systematic review and meta-analysis. Prim. Care Diabetes 16, 591–593 (2022).
Ssentongo, P., Zhang, Y., Witmer, L., Chinchilli, V. M. & Ba, D. M. Association of COVID-19 with diabetes: a systematic review and meta-analysis. Sci. Rep. 12, 20191 (2022).
Li, J. et al. Increased risk of new-onset diabetes in patients with COVID-19: a systematic review and meta-analysis. Front. Public Health 11, 1170156 (2023).
Salmi, H. et al. New-onset type 1 diabetes in Finnish children during the COVID-19 pandemic. Arch. Dis. Child. 107, 180–185 (2022).
Shulman, R., Cohen, E., Stukel, T. A., Diong, C. & Guttmann, A. Examination of trends in diabetes incidence among children during the COVID-19 pandemic in Ontario, Canada, from March 2020 to September 2021. JAMA Netw. Open 5, e2223394 (2022).
Tittel, S. R. et al. Did the COVID-19 lockdown affect the incidence of pediatric type 1 diabetes in Germany? Diabetes Care 43, e172–e173 (2020).
Van Belle, T. L., Coppieters, K. T. & Von Herrath, M. G. Type 1 diabetes: etiology, immunology, and therapeutic strategies. Physiol. Rev. 91, 79–118 (2011).
Singh, S. et al. Impact of COVID-19 and lockdown on mental health of children and adolescents: a narrative review with recommendations. Psychiatry Res. 293, 113429 (2020).
Karavanaki, K. et al. Psychological stress as a factor potentially contributing to the pathogenesis of type 1 diabetes mellitus. J. Endocrinol. Invest. 31, 406–415 (2008).
Hirani, D., Salem, V., Khunti, K. & Misra, S. Newly detected diabetes during the COVID-19 pandemic: what have we learnt? Best Pract. Res. Clin. Endocrinol. Metab. 37, 101793 (2023).
Boboc, A. A. et al. SARS-CoV-2 positive serology and islet autoantibodies in newly diagnosed pediatric cases of type 1 diabetes mellitus: a single-center cohort study. Int. J. Mol. Sci. 24, 8885 (2023).
Kamrath, C. et al. Frequency of autoantibody-negative type 1 diabetes in children, adolescents, and young adults during the first wave of the COVID-19 pandemic in Germany. Diabetes Care 44, 1540–1546 (2021).
Zhou, L., Qu, H., Zhang, Q., Hu, J. & Shou, L. Case report: fulminant type 1 diabetes following paucisymptomatic SARS-CoV-2 infection during late pregnancy. Front. Endocrinol. 14, 1168927 (2023).
Pan, Y., Zhong, X., Qiu, W., Zhao, F. & Yao, J. New-onset fulminant type 1 diabetes after SARS-CoV-2 infection. Diabetes Care 46, e140–e142 (2023).
Martinez, A. H., Hicks, K. A., Moorjani, T. P., Bell, J. & Lin, Y. A case of autoantibody negative pediatric diabetes mellitus with marked insulin resistance concomitant with COVID-19: a novel form of disease? J. Endocr. Soc. 5, A690–A691 (2021).
Hollstein, T. et al. Autoantibody-negative insulin-dependent diabetes mellitus after SARS-CoV-2 infection: a case report. Nat. Metab. 2, 1021–1024 (2020).
Acknowledgements
The authors thank the team of the Laboratory of Virology ULR3610 and all their collaborators. The authors’ research was supported by Ministère de l’Education Nationale, de la Recherche et de la Technologie, Université de Lille, CHU Lille.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Endocrinology thanks Malin Flodström Tullberg and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
CoviDIAB project: https://covidiab.e-dendrite.com
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Debuysschere, C., Nekoua, M.P., Alid**ou, E.K. et al. The relationship between SARS-CoV-2 infection and type 1 diabetes mellitus. Nat Rev Endocrinol (2024). https://doi.org/10.1038/s41574-024-01004-9
Accepted:
Published:
DOI: https://doi.org/10.1038/s41574-024-01004-9
- Springer Nature Limited