Morphological characteristics and mechanical robustness
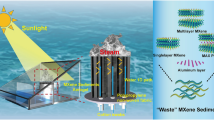
To comprehensively demonstrate the high strength and good transport ability of the wood-inspired metamaterial catalyst for water purification, we constructed a series of microlattice metamaterials with different strut diameters (0.30, 0.35, and 0.40 mm) and overlap rates (0, 30, 50, and 70%). 316 L SS, which is known to provide sufficient mechanical strength and corrosion resistance in water purification, was selected as the parent material. The experimental samples were prepared via SLM, which was characterized by high precision and high efficiency in the production of complex metallic structures43,44,45,46,47,48. SS-based microlattice metamaterials with different geometrical characteristics were successfully printed (Fig. 2a). The optical images of the 3D-printed microlattice metamaterials with 0.40 mm diameter and 0%, 30%, 50%, and 70% overlap rates are shown in Fig. 2b. Based on the Gibson-Ashby model of E/Es = C(RD)n (E and Es are Young’s modulus of microlattices and parent materials, respectively, C is within the ranges of 0.1–4.0, and RD is the relative density, n is 2 for bending-dominated structures)49,50,51, there is a positive correlation between the relative density and Young’s modulus of microlattice structures. The overlap design strategy was intended to increase relative density resulting from the increased node numbers and internal constraints of struts (Fig. 2c), which could significantly improve the structural stiffness to effectively withstand the flow impact. Meanwhile, the wood-inspired bimodal pores were intended to retard the fluid velocity of wastewater, thereby increasing the liquid-solid contact time in the matter transportation process for high-efficient catalytic purification. This is due to the wood-inspired overlap design increasing the surface area per volume, thereby increasing flow friction (Fig. 2c).
As shown in Fig. 2d, e, the traditional microlattice features uniform square pores with thin walls that were made up of inclined struts and robust nodes. After the overlap design of wood-inspired bimodal pores, the shaped pores sprout from staggered microlattice cells. Figure 2f, g show that numerous small pores are generated between neighboring microlattice cell walls (in the middle region) due to the overlap of space volume, resulting in decreased structural porosity. The comparison of the micro-CT-reconstructed and CAD models of traditional microlattices and wood-inspired metamaterials reveals that a surface deviation occurred on the downward surface of the struts, as indicated by the red nephogram, owing to the lack of sufficient holding force (Supplementary Figs. 2–3). Even if there are manufacturing deviations in 3D printing technology, the overall structure remains highly accurate with a surface peak deviation (Dp) of less than 30 μm and a dense solid part with small void defects (Fig. 2h, Supplementary Figs. 4, 5, and Supplementary Note 1). We have carefully considered the potential impact of manufacturing defects, such as void defects, cracks, and manufacturing accuracy, in the SLM process on the performance of metamaterial catalysts and discussed their influences in Supplementary Figs. 5–10 and Supplementary Notes 2–5. Given the maturity of SLM technology and the high additively manufacturability of 316 L SS metal powder, the few defects in both SLM-printed microlattices and wood-inspired metamaterials can be ignored. In addition, the small unevenness of the 3D-printed metamaterial surface facilitates the increase of surface area and the adhesion of Co atoms of the electrochemical deposition process. It can be found that there is a large amount of Co on the surface of the 3D-printed 316 L metamaterial surface (Fig. 2i and Supplementary Fig. 11).
The application of wood-inspired microlattice metamaterial catalysts requires evaluating the load-bearing capacity and its ability to withstand the impact of water flow. Damage-resistant and high-strength metamaterial catalysts are conducive to the long-term usability and stability of water purification systems. To explore whether the microlattice overlap process changes did impact the mechanical robustness, we used a quasi-static compression experiment to measure Young’s modulus of the wood-inspired metamaterial catalyst under compression (Fig. 2j, k). All of the 3D-printed microlattice metamaterial catalysts exhibited a stable plateau stage after the linear elastic stage of rapid stress increase under longitudinal external loads, and then the final densification stage was accompanied by a sharp rise in stress (Fig. 2j). We found that the larger overlap rate of microlattices, the higher strength as a same strain. The Young’s modulus of traditional microlattices of 0.40 mm strut diameter with a 0% overlap rate is 110.17 MPa, which sharply increased to 367.36 MPa as the overlap rate increased to 70%. In addition, the variation of strut parameters in the microlattice metamaterials does not change the robustness-enhanced attribution of the microlattice overlap (Fig. 2k and Supplementary Fig. 12). Due to the flexibility of the BCC-type microlattice and the plasticity of the 316 L material, metamaterial structures all failed as the buckling deformation Supplementary Figs. 13, 14). The finite element visualization results reveal that the stress was mainly concentrated near the node (Fig. 2l). The mechanical analysis of Euler’s buckling theory also shows that the decrease in equivalent strut length results in an increase in the buckling force, therefore leading to increased mechanical robustness (Supplementary Fig. 15). The proposed microlattice overlap design strategy could effectively disperse the stress under longitudinal external loading to improve the overall structural strength. With increasing overlap rate, the constraint between nodes in the wood-inspired microlattice metamaterials increased and the structural stiffness increased. Our results show that the overlap process and subsequently forming the bimodal pores greatly improves the robustness of microlattice metamaterials. At the same time, we should note that for different topological microlattices, the influence of this wood-inspired overlap** design strategy on their mechanical properties is distinctive. In addition to the BCC-type microlattice, we also considered the mechanical responses of other topological microlattices with different pore characteristics (Supplementary Fig. 16). Overall, the overlap** design strategy can enhance their mechanical robustness. However, not all microlattices follow the rule that the greater the overlap rate, the higher the strength improvement. This is attributed to the limitation of the force acting on the internal strut elements of the microlattice. In addition, it can be found that different topological microlattices can all be used for wood-inspired overlap** design, which verifies the flexibility of our design strategy and the universality of our method, and further enhances the design space of mechanical strength through microlattice topology transformation.
Transport performance
Transport performance includes air/fluid flow behavior and permeability, the latter of which directly indicates the capacity of microlattices to transport matter and affects matter exchange within structures52,53. Permeability is generally related to porosity, pore shape, and pore distribution. In the design and application of biological scaffolds, permeability, as one of the indicators for evaluating biological properties, can reflect the cell proliferation and differentiation of scaffolds20,21. The permeability of wood-inspired metamaterial catalysts affects the water purification efficiency of wastewater treatment systems, in which catalytic reaction occurs as wastewater goes through penetrating channels covered by Co/SS8. The transport performance of the wood-inspired microlattice metamaterial catalyst is determined mainly by its strut structure and pore characteristics. Although traditional periodic microlattices have multiple geometrical parameters for tailoring permeability, their tunability of transport properties is still limited due to their strong relationships with other performances, such as mechanical performance20,21,24. After microlattice overlap for forming the bimodal pores with staggered distribution patterns like the microstructure of Douglas fir, numerous millimeter-scale sub-pores were formed in the wood-inspired metamaterial catalyst. These sub-pores gain more flow channels, functioning as high-throughput transport origins. Besides, the microlattice overlap process generates a higher surface area and smaller pores (<D0, in the traditional periodic microlattices) within the metamaterial catalyst, which provides more contact time for high-efficiency catalysis.
To reveal the fundamental physics of the overlap** strategy on transport performances, we investigated fluid flow behavior insides both the traditional microlattice and wood-inspired metamaterial catalyst using computational fluid dynamics (CFD) finite element calculations, in which a fluid inlet is arranged on the upper surface of the structure perpendicular to the overlap** direction (Fig. 3a and Supplementary Fig. 17). We found a relatively large apparent permeability of traditional microlattices with a 0% overlap rate, which gradually decreased with the increases of the overlap rate. Besides, the lower the inlet velocity, the greater the permeability reduction (Fig. 3b). The permeability of wood-inspired metamaterial catalyst increased with increasing overlap rate and decreased with increasing strut diameter. The wood-inspired overlap design not only regulates the transport properties by adjusting strut diameter but also increases the design of freedom. This means that the strut diameter and overlap rate can synergistically increase the tunability of the transport performance (Fig. 3c). Figure 3d–i compares the distributions of the fluid velocity and shear rate within both traditional microlattices and wood-inspired metamaterial catalysts. Due to the large pore and periodic arrangement of traditional microlattices, the inlet fluid transports quickly without obstacle and induces very large-scale high-velocity (Fig. 3d) in the center of the multilayer pore channels. By contrast, due to the sub-pores and high surface area generated by the microlattice overlap** process, the inlet fluid can only slowly penetrate the wood-inspired metamaterial catalyst, causing much lower velocity in the pores (Fig. 3f). Comparison between Fig. 3g and 3h confirms that friction between the fluidic liquid and the strut surfaces increases the velocity, as well as causes the shear rate concentration in the liquid-solid interface (Fig. 3i). These make the wood-inspired metamaterial catalyst behave lower permeability, which is conducive to the contact between wastewater fluid and metamaterial catalyst, thus improving the reaction efficiency. In addition, we compared the transport permeability of the wood-inspired metamaterial catalysts with that of other structural types such as simple cubic (SC), face-center cubic (FCC), diamond, cuboctahedron, octahedron and octet-truss under the same inlet condition (Fig. 3j). We also calculated the pressure drop (Supplementary Fig. 18), which was the average pressure of the inlet surface at 1 mm/s inlet velocity. The wood-inspired overlap** design strategy facilitates increased pressure drop. Compared to traditional periodic microlattices, our developed wood-inspired metamaterials have higher strength and still good permeability. Besides, overlap** microlattice design enhances the tailoring space for strength and permeability. We also need to note that due to the increase in relative density, the permeability of the metamaterial structure is inevitably decreased. But for microlattice metamaterials used for structural catalysis, lower permeability means more complete catalytic processes. High permeability and high relative density are the design goals for robust and high-throughput metamaterials for the applications of high-efficient matter transportation without the reaction between liquid and solid. However, we consider that in the application of metamaterial catalysts, having relatively lower permeability and higher relative density within a certain permeability range is the ultimate goal direction.
The permeability of the traditional microlattices and different wood-inspired metamaterials have been experimentally tested, which is presented in Experiments and Methodologies. The experimental permeability results were compared with the corresponding computational permeability results of the traditional microlattice and different wood-inspired metamaterials, which was presented in Fig. 3k. There is usually a certain deviation in the simulated permeability and experimental permeability, but even if the microlattice topology is different, there is still a high linear relationship under different overlap** designs, indicating a good agreement between experiments and simulations. The permeability values of CFD simulations were well correlated (R2 = 0.80, 0.97, 1.00, 0.94, 0.99, 0.90, and 1.00, for SC, FCC, diamond, cuboctahedron, octahedron, octet-truss, and BCC, respectively) with experimental results. The discrepancies in experimental and computational permeability values could be attributed to the following reasons: First, the SLM-printed microlattices have slight surface roughness, leading to the overestimated permeability. Second, the fluid height could be a factor for the constant pressure permeability testing setup, while the height of the pressure head in the laboratory is lower than the optimal value20. Finally, the small gap between the permeability testing setup and the periphery of the tested samples also results in permeability discrepancies.
Water purification capacity and mechanism
In the process of verifying the wood-inspired metamaterial catalyst (mainly Co/SS) activated peroxymonosulfate (PMS) for water purification, the metamaterial catalyst was assembled into a fixed-bed water purification system. The results of the degradation process of sulfamethoxazole (SMX) were obtained via high-performance liquid chromatography (HPLC). The traditional microlattice with 0% overlap rate and wood-inspired metamaterial catalyst with 30, 50, and 70% have all completed the degradation process of SMX within 15 min (Fig. 4a). The kinetics of 20 ppm SMX degradation was presented (Supplementary Fig. 19) and analyzed by a pseudo-first-order reaction model expressed as follows:
$$-{{{{{{\mathrm{ln}}}}}}}\left(\frac{\it{C}}{\it{C}_{0}}\right)={\it{kt}}$$
(1)
where C0 is the initial concentration of SMX, C is the concentration of SMX after treatment, and k is the kinetic constant of the reaction. Figure 4b summarizes the dynamic constant results normalized by the occupied space volume, which shows a positive correlation with the overlap rate. These results are all consistent with the transport simulation results described above. The 70% overlap metamaterial catalyst showed the highest reaction kinetic constant per unit volume after normalization (0.191 cm−3). Meanwhile, the kinetic constants of wood-inspired metamaterial catalyst activation PMS with different strut diameters were also tested and normalized (Supplementary Fig. 20). With the increase in the strut diameter, the surface area of the metamaterial catalyst was increased, which was accompanied by the increase in the PMS activation rate. To confirm the organic pollutant removal rate, we characterized the organic mineralization rate in the 70% overlap metamaterial catalyst water purification system by measuring the total organic carbon (TOC). The unmineralized organic pollutants decreased to 24.1% at 15 min and decreased to 18.8% at 30 min, proving that most organic matter is mineralized into non-toxic inorganic matter (Fig. 4c). Meanwhile, high-performance liquid chromatography-mass spectrometry (HPLC-MS) was used to detect intermediates in the degradation process. The mass spectrometry of the possible intermediate products is depicted in Supplementary Fig. 21a. Intermediate products with m/z values of 97, 137, 179, 181, 207, 228, 252, 266, 268, and 282 were detected by the negative ionization mode of the MS spectra. Based on previous reports54,55, the possible degradation pathways of SMX are depicted in Supplementary Fig. 21b. The toxicity of SMX pollutants and possible degradation intermediates was assessed by a toxicity estimation software tool (T. E. S. T.). The fathead minnow LC50-96 h was investigated by the mode of action method56. As shown in Supplementary Fig. 22, SMX with a fathead minnow LC50-96 h value of 40.23 mg L−1 was considered “harmful”, while the toxicity of intermediates produced in pathway I was increased, while the toxicity of intermediates in pathway II was decreased. Although some toxic intermediates were produced during the degradation process, considering that the Co/SS metamaterial catalyst /PMS system has a strong oxidation and mineralization capacity for SMX, it should be considered as an environmentally friendly water treatment system.
Compared with some other PMS catalytic degradation systems, this degree of mineralization of Co/SS metamaterial catalysts is significant (Supplementary Table 1). The Co/SS metamaterial catalyst/PMS catalyzed SMX degradation process is beneficial in reducing the toxicity of SMX antibiotics. Most intermediates showed a trend of decreasing toxicity, although a small percentage remained toxic. Therefore, Co/SS metamaterial catalyst as a persulfate catalyst that can highly mineralize SMX should be an effective means of detoxification.
Furthermore, the mechanism of PMS activation by the wood-inspired metamaterial catalyst was demonstrated by using active substance quenching experiments. In this experiment, methanol (MeOH), tert-butyl alcohol (TBA), and furfuryl alcohol (FFA) were equipped as quenching agents. The experimental results of active substance quenching are shown in Supplementary Fig. 23a. The addition of each of the three quenching agents significantly reduced the catalytic rate, confirming that ‧OH, ‧SO4−, and non-radical active substances occurred in the degradation system. To quantify the contribution of different radicals, we fitted the reaction kinetic constants in the presence of different quenching agents using Eq. (1) (Supplementary Fig. 23b). When added in sufficient abundance, the quenching agent will immediately deplete the corresponding active substance; thus, the contributions of different active substances to the degradation process can be quantified. The results showed that ‧OH, ‧SO4−, and non-radical active substances accounted for 46.3, 25.5, and 28.6% of the degradation process, respectively (Fig. 4d). Therefore, the degradation process was a free radical pathway dominated by ‧OH.
Electron paramagnetic resonance (EPR) spectroscopy is widely used to verify the existence of free radicals. The technique can directly detect the active components in the degradation system. The reagents 5,5-dimethyl-1-pyrroline-N-oxide (DMPO) and 2,2,6,6-tetramethylpiperidine (TEMP) were added to the degradation system to detect potential free radicals. The EPR results are shown in Fig. 4e. In the system with DMPO as the collector, the EPR signal of the DMPO-OH adduct (αN = αH = 14.8 G) was detected. The EPR signal of the DMPO-SO4 adduct (αN = 13.2 G, αH = 9.6 G, αH = 1.48 G, αH = 0.78 G) was not detected because of the low concentration of ‧SO4−. In the system with TEMP as the trap** agent, TEMPO (α = 16.9 G) exhibited a characteristic threefold signal of 1:1:1, indicating generation. Therefore, the EPR results demonstrate that the non-free radical pathway includes 1O2 as the active substance. To verify electron transfer during degradation, a time-current curve was performed under the open circuit voltage (Fig. 4f). When only a metamaterial catalyst exists in the system, there is no obvious current response. With the addition of PMS, the current density increased significantly, indicating that the interaction between PMS and Co/SS metamaterial catalyst formed a metastable complex. In the system with both PMS and SMX, the current density was further enhanced, implying electron transfer from SMX to Co/SS metamaterial catalyst/PMS complex, which proves that direct electron transfer also exists in the degradation process of the non-radical pathway.
To find out the change in the valence state of metamaterial catalyst surface elements during degradation, X-ray photoelectron spectroscopy (XPS) was investigated. Before the reaction, the Co/SS metamaterial catalyst exhibited Co 2p3/2 peaks at 781.3, and 783.1 eV, corresponding to Co(III) and Co(II), respectively, and the peak area ratio ICoII:ICoIII = 0.32. After the reaction, the peak area of Co(III) increased significantly, and ICoII:ICoIII = 0.21 (Fig. 4g). These results indicate the conversion of Co(II) and Co(III) on the Co/SS metamaterial catalyst surface during PMS activation and the catalytic reaction. Moreover, the Co/SS metamaterial catalyst before the reaction exhibited Fe 2p3/2 peaks at 710.6 eV, indicating that the frame material had surface oxidation before the water treatment (Fig. 4h). After the water treatment, the Co/SS metamaterial catalyst featured a Fe 2p3/2 peak at 712.5 eV, indicating the generation of Fe(III). According to the changes in the valence state, we propose the following mechanism of PMS activation by Co/SS metamaterial catalyst (Fig. 4i):
$$\equiv {{{{{{{\rm{C}}}}}}}}{{{{{{{\rm{o}}}}}}}}(0)+{{2{{{{{{{\rm{HSO}}}}}}}}}_{5}^{-}}\to \equiv {{{{{{{\rm{Co}}}}}}}}({{{{{{{\rm{II}}}}}}}})+{{2{{{{{{{\rm{HSO}}}}}}}}}_{4}^{-}}+2{}^{\cdot }{{{{{{{\rm{OH}}}}}}}}$$
(2)
$$\equiv {{{{{{{\rm{Co}}}}}}}}({{{{{{{\rm{II}}}}}}}})+{{{{{{{{\rm{HSO}}}}}}}}_{5}^{-}}\to \equiv {{{{{{{\rm{Co}}}}}}}}({{{{{{{\rm{III}}}}}}}})+{}^{\cdot }{{{{{{{{\rm{SO}}}}}}}}}_{4}^{-}+{{{{{{{{\rm{OH}}}}}}}}}^{-}$$
(3)
$$\equiv {{{{{{{\rm{Co}}}}}}}}({{{{{{{\rm{III}}}}}}}})+{{{{{{{{\rm{HSO}}}}}}}}_{5}^{-}}\to \equiv {{{{{{{\rm{Co}}}}}}}}({{{{{{{\rm{II}}}}}}}})+{}^{\cdot }{{{{{{{{\rm{SO}}}}}}}}}_{5}^{-}+{{{{{{{{\rm{H}}}}}}}}}^{+}$$
(4)
$$\equiv {{{{{{{\rm{Co}}}}}}}}\left({{{{{{{\rm{III}}}}}}}}\right)+{}^{\cdot }{{{{{{{{\rm{SO}}}}}}}}}_{5}^{-}+{{{{{{{{\rm{H}}}}}}}}}^{+}\to \equiv {{{{{{{\rm{Co}}}}}}}}\left({{{{{{{\rm{II}}}}}}}}\right)+{}^{\cdot }{{{{{{{{\rm{SO}}}}}}}}}_{4}^{-}+{}^{\cdot }{{{{{{{\rm{OH}}}}}}}}$$
(5)
$$\equiv {{{{{{{\rm{Fe}}}}}}}}(0)+\equiv {{{{{{{\rm{Co}}}}}}}}({{{{{{{\rm{II}}}}}}}})\to {{{{{{{\rm{Co}}}}}}}}(0)+\equiv {{{{{{{\rm{Fe}}}}}}}}({{{{{{{\rm{II}}}}}}}})$$
(6)
$$\equiv {{{{{{{\rm{Fe}}}}}}}}\left({{{{{{{\rm{II}}}}}}}}\right)+\equiv {{{{{{{\rm{Co}}}}}}}}\left({{{{{{{\rm{III}}}}}}}}\right)\to 2{{{{{{{\rm{Co}}}}}}}}\left({{{{{{{\rm{II}}}}}}}}\right)+\equiv {{{{{{{\rm{Fe}}}}}}}}\left({{{{{{{\rm{III}}}}}}}}\right)$$
(7)
$${{{{{{{{{\rm{HSO}}}}}}}}}_{5}^{-}+{}^{\cdot }{{{{{{{{\rm{SO}}}}}}}}}_{5}^{-}}\to {}^{1}{{{{{{{\rm{O}}}}}}}}_{2}+{{{{{{{{\rm{H}}}}}}}}}^{+}+{2{{{{{{{\rm{SO}}}}}}}}}_{4}^{2-}$$
(8)
The corrosion resistance of Co/SS metamaterial catalysts is the basement for long-term use. The corrosion resistance of the Co/SS framework has been reduced to a certain extent; this is the response for Co as a reactive substance is not as resistant to corrosion of PMS as Fe (Supplementary Fig. 24a–c and Supplementary Note 6). According to the standard electrode potential EΘ of Fe/Fe2+ is −0.44 V and EΘ of Co/Co2+ is −0.28 V, which means that galvanic corrosion occurs when Co/SS contact, and electrons are transferred from Fe to Co. This result hurts the corrosion resistance of SS-based metamaterials as shown in the above corrosion data. However, electron injection into Co is conducive to the circulation of Co species in the catalytic process (Supplementary Fig. 24d).
The long-term stability of the wood-inspired metamaterial catalyst is also an important factor in practical utilization. To understand the maximum stability of Co/SS metamaterial catalysts, a water treatment system as shown in Fig. 4j, was assembled, which consisted of two peristaltic pumps mixing 20 ppm SMX and 0.4 g L−1 PMS at a flow rate of 0.5 mL min−1 respectively into a fixed-bed processor equipped with Co/SS metamaterial catalysts, and a third bucket was used to collect the treated liquid. It was found that the Co/SS metamaterial catalysts could run continuously for more than 96 h and the degradation efficiency remained above 90%, as shown in Fig. 4k. After 108 h treatment, the degradation efficiency decreased, which may be due to the occurrence of surface deactivation and passivation. This deactivation is attributed to the lack of active Co species, and the degradation efficiency can be restored to 98% by Ar/H2 reduction at 300 °C for 3 h. It should be noted that a vast of AOP-activated materials have only studied the service life of five cycles, and this stability is far from enough for practical water treatment materials. Our Co/SS metamaterial catalysts show the stability of the day level, which has a certain positive significance for the practical application of water treatment materials. Compared with other high-stability water treatment systems, the stability of our system has a considerable advantage (Supplementary Table 2).
Practicality, designability, and applicability
Actual water bodies contain numerous kinds of inorganic anions and other organic substances, many of which will inhibit PMS activation. We verified the resistance of the wood-inspired metamaterial catalyst to 200 ppm NaHCO3, Na2SO4, NaNO3, NaCl, and 20 ppm humic acid (HA) in the PMS activation process. Although impurities influenced the ‧OH-dominated activation process (Fig. 5a), the Co/SS metamaterial catalyst still achieved over 93% treatment efficiency within 15 min in 200 ppm Na2SO4, NaNO3, and NaCl. To verify the effect of pH, the wood-inspired metamaterial catalyst was placed in environments of various pH, from 3 to 9, and they maintained excellent SMX treatment efficiency under both acidic and neutral conditions (pH = 3, 5, and 7, Fig. 5b). The treatment efficiency was inhibited under alkaline conditions (pH = 9), but a 75% degradation rate was still achieved after 10 min. It could be that OH- covers the surface of the catalyst, resulting in unfavorable adsorption of PMS and pollutants. After that, we used actual water samples as the solvent to test the activation degradation of SMX by PMS. The results showed that the degradation rate of the wood-inspired metamaterial catalyst reached more than 95% after 15 min in tap water and Yangtze River water as the solvent (Supplementary Fig. 25), which indicated the application potential of Co/SS metamaterial catalyst in the real environment. The wood-inspired metamaterial catalyst-activated PMS system can not only be used for SMX degradation but also bisphenol A (BPA) and norfloxacin (NOR) can achieve 95.3 and 98.2% efficiency within 15 min, respectively (Fig. 5c). This indicates that the wood-inspired metamaterial catalyst-activated PMS water purification system can be used in water samples with complex contaminants.
The above results demonstrated the practicality of the wood-inspired metamaterial catalyst. On this basis, we elucidated the geometrical characteristics of wood-inspired metamaterial catalysts (Fig. 5d). The structural design (mechanical and transport properties) and catalytic performances (treatment duration) of the wood-inspired metamaterial catalyst can be leveraged to obtain a large-scale, continuous-flow, and high-throughput water treatment, demonstrating its application potential in flow-related engineering (Fig. 5e). We can also achieve desirable throughput and water quality by flexibly and precisely tuning the structure and catalytic performances. The stiffness (E) of the wood-inspired metamaterial catalyst relies on the strut diameter (d) and overlap rate (δ). The water flow time (t1) depends on the surface porosity (SA) and the overall length (N × a), while the catalytic time (t2) is related to the permeability and the type of catalyzer (x). In addition, the permeability and surface porosity are both influenced by geometrical characteristics, including strut diameter and overlap rate. This relationship network involves two constraints. First, the water flow time needs to be shorter than the catalytic time to ensure complete purification of the sewage. Second, the impact strength of water flow needs to be less than the rigidity of the wood-inspired metamaterial catalyst. A reverse engineering-guided design was conducted according to the established relationship and known constraints related to geometric topology, and physical and chemical properties. The stiffness, transport property, and catalytic capacity of the wood-inspired metamaterial catalyst were tunable, which indicates that the structure-function integrated manufacturing of water purification systems can be easily realized through metamaterial structural design and 3D printing. Microlattices or porous structures have strong structural designability and additively manufacturability57, while also exhibiting significant mechanical robustness for metal-based materials in 3D printing component manufacturing. However, due to the low selectivity of metal-based materials in the SLM process, many materials with good catalytic performance are difficult to manufacture as components, which greatly limits their catalytic applications in the fields of environment and energy. On the contrary, metal-organic frameworks or carbon-based synthetic materials have ultra-high surface area and permanent porosity, making them ideal candidates for fuel (hydrogen and methane) storage, carbon dioxide capture, and catalytic applications58. However, they are still full of challenges for large-scale engineering applications in terms of the current manufacturable sizes of only nanometers/micrometers and poor chemical stability. Compared with the microlattice/porous structure and synthesis catalyst, the wood-inspired metamaterial catalyst, which was fabricated by the simple preparation through the combined process of metal 3D printing and electrochemical deposition, integrates the dual functions of structural strength and functional catalysis, has strong manufacturability, higher macro- and micro- structural designability, stronger controllability of geometry and performance, higher strength and high-throughput water purification capability (Fig. 5f).
The wood-inspired metamaterial catalyst composed of assembled microlattices can be realized at different length scales (Fig. 5g), because of the advances in 3D printing technologies, which could, in principle, scale the pore size from the micrometer to the meter scales, and use different constitutive materials, targeting different applications (such as different water purification scenes). Meanwhile, it is even possible to consume harmful metabolic substances by implanting tiny metamaterial scaffolds for high-throughput catalytic reactions in organisms (Fig. 5g). Our work focuses on high-throughput structural design to achieve robust and efficient metamaterial catalysts. There are certain limitations in the manufacturing process selection and material selection. However, the design strategy of imitating wood bimodal pores and the strategy of coupling 3D printing with electrochemical deposition to form high-throughput metamaterial catalysts can be used for reference in other flow-catalytic fields. Besides, compared with the original performance of Douglas fir wood, the 3D-printed wood-like overlap** microlattice metamaterial still exhibits moderate mechanical and excellent transport properties in the macroscale (Supplementary Note 7 and Supplementary Figs. 26–28).
In summary, we report a structure-function integrated wood-inspired metamaterial catalyst water purification system based on metal 3D printing technology and an electrochemical deposition process. The wood-inspired metamaterial structure was developed by a microlattice overlap strategy, which allows the creation of numerous sub-pores between the microlattice units with increased robustness and surface area per volume. We presented that a wood-inspired metamaterial catalyst with a 70% overlap rate has three times more strength, three times surface area per unit volume, and four times normalized reaction kinetics than traditional microlattices. The overlap strategy and subsequently formed bimodal pores greatly improve the robustness of wood-inspired metamaterial catalysts. The overlap-generated numerous sub-pores and high surface area make the inlet fluid slowly penetrate the structure, causing much higher velocity and adequate contact reaction in the pores within wood-inspired metamaterial catalysts. Compared with multi-process and time-consuming water purification systems, the metamaterial catalysts possess higher efficiency, low cost, and scalability. These wood-inspired metamaterial catalysts with superior mechanical robustness, high-throughput flow, and high-efficiency catalysis can replace traditional water purification systems, as well as inspire unprecedented development in the fields of flow catalysis and other structure-function integrated applications.