Abstract
Introduction
Phospholipids are possible favorable agents for colorectal cancer (CRC). Choline has been inversely related to CRC risk but findings are inconsistent. We assessed the effect of dietary sphingomyelin (SM) choline moiety and total choline intake on risk of CRC.
Method
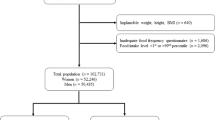
This analysis is based on a multicenter case–control study conducted between 1992 and 1996 in Italy. A total of 6107 subjects were enrolled, including 1225 colon cancer cases, 728 rectal cancer cases and 4154 hospital-based controls. We applied data on the composition of foods in terms of SM choline moiety and choline intake on dietary information collected through a validated food-frequency questionnaire. Odds ratio (OR) for energy-adjusted tertiles of SM choline moiety and choline were estimated through logistic regression models adjusted for sex, age, center, education, alcohol consumption, body mass index, family history of CRC, and physical activity.
Results
Choline was inversely related to CRC risk (OR for the highest versus the lowest tertile: 0.85; 95% confidence interval [CI]: 0.73–0.99), with a significant trend in risk. The OR for an increment of one standard deviation of energy-adjusted choline intake was 0.93 (95% CI: 0.88–0.98). The association was consistent in colon and rectal cancer and also across colon subsites. SM choline moiety was not associated with CRC risk (OR for the highest versus the lowest tertile: 0.96, 95% CI 0.84–1.11).
Conclusion
This study shows an inverse association between choline intake and CRC but not with SM choline moiety.
Similar content being viewed by others
Data availability
Data are available upon reasonable request from the corresponding author.
References
Hannun YA, Obeid LM. Sphingolipids and their metabolism in physiology and disease. Nat Rev Mol Cell Biol. 2018;19:175–91.
Milhas D, Clarke CJ, Hannun YA. Sphingomyelin metabolism at the plasma membrane: implications for bioactive sphingolipids. FEBS Lett. 2010;584:1887–94.
Dudeja PK, Dahiya R, Brasitus TA. The role of sphingomyelin synthetase and sphingomyelinase in 1,2-dimethylhydrazine-induced lipid alterations of rat colonic plasma membranes. Biochim Biophys Acta. 1986;863:309–12.
Dillehay DL, Webb SK, Schmelz EM, Merrill AH Jr. Dietary sphingomyelin inhibits 1,2-dimethylhydrazine-induced colon cancer in CF1 mice. J Nutr. 1994;124:615–20.
Schmelz EM, Dillehay DL, Webb SK, Reiter A, Adams J, Merrill AH Jr. Sphingomyelin consumption suppresses aberrant colonic crypt foci and increases the proportion of adenomas versus adenocarcinomas in CF1 mice treated with 1,2-dimethylhydrazine: implications for dietary sphingolipids and colon carcinogenesis. Cancer Res. 1996;56:4936–41.
Schmelz EM, Bushnev AS, Dillehay DL, Liotta DC, Merrill AH Jr. Suppression of aberrant colonic crypt foci by synthetic sphingomyelins with saturated or unsaturated sphingoid base backbones. Nutr Cancer. 1997;28:81–5.
Symolon H, Schmelz EM, Dillehay DL, Merrill AH Jr. Dietary soy sphingolipids suppress tumorigenesis and gene expression in 1,2-dimethylhydrazine-treated CF1 mice and ApcMin/+ mice. J Nutr. 2004;134:1157–61.
Ahn EH, Schroeder JJ. Sphingoid bases and ceramide induce apoptosis in HT-29 and HCT-116 human colon cancer cells. Exp Biol Med. 2002;227:345–53.
Sugawara T, Zaima N, Yamamoto A, Sakai S, Noguchi R, Hirata T. Isolation of sphingoid bases of sea cucumber cerebrosides and their cytotoxicity against human colon cancer cells. Biosci Biotechnol Biochem. 2006;70:2906–12.
Schmelz EM, Bushnev AS, Dillehay DL, Sullards MC, Liotta DC, Merrill AH Jr. Ceramide-beta-D-glucuronide: synthesis, digestion, and suppression of early markers of colon carcinogenesis. Cancer Res. 1999;59:5768–72.
Symolon H, Bushnev A, Peng Q, Ramaraju H, Mays SG, Allegood JC, et al. Enigmol: a novel sphingolipid analogue with anticancer activity against cancer cell lines and in vivo models for intestinal and prostate cancer. Mol Cancer Ther. 2011;10:648–57.
Zhang P, Li B, Gao S, Duan RD. Dietary sphingomyelin inhibits colonic tumorigenesis with an up-regulation of alkaline sphingomyelinase expression in ICR mice. Anticancer Res. 2008;28:3631–5.
Vesper H, Schmelz EM, Nikolova-Karakashian MN, Dillehay DL, Lynch DV, Merrill AH Jr. Sphingolipids in food and the emerging importance of sphingolipids to nutrition. J Nutr. 1999;129:1239–50.
Wang X, Wang Y, Xu J, Xue C. Sphingolipids in food and their critical roles in human health. Crit Rev Food Sci Nutr. 2021;61:462–91.
Yonemori KM, Lim U, Koga KR, Wilkens LR, Au D, Boushey CJ, et al. Dietary choline and betaine intakes vary in an adult multiethnic population. J Nutr. 2013;143:894–9.
Lu MS, Fang YJ, Pan ZZ, Zhong X, Zheng MC, Chen YM, et al. Choline and betaine intake and colorectal cancer risk in Chinese population: a case-control study. PLoS One. 2015;10:e0118661.
Lee JE, Giovannucci E, Fuchs CS, Willett WC, Zeisel SH, Cho E. Choline and betaine intake and the risk of colorectal cancer in men. Cancer Epidemiol Biomarkers Prev. 2010;19:884–7.
Zeisel S, Corbin K Choline. Present Knowledge in Nutrition. 10th ed: John Wiley & Sons, Inc.; 2012:405–. https://lpi.oregonstate.edu/mic/other-nutrients/choline. 2012.
Zeisel SH, Blusztajn JK. Choline and human nutrition. Annu Rev Nutr. 1994;14:269–96.
Kondo Y, Issa JP. DNA methylation profiling in cancer. Expert Rev Mol Med. 2010;12:e23.
Laird PW, Jaenisch R. DNA methylation and cancer. Hum Mol Genet. 1994;3:1487–95.
Davis CD, Uthus EO. DNA methylation, cancer susceptibility, and nutrient interactions. Exp Biol Med. 2004;229:988–95.
Patterson KY, Bhagwat S, Williams JR, Howe JC, Holden JM, Zeisel SH, et al. USDA Database for the Choline Content of Common Foods, Release 2 (2008). Nutrient Data Laboratory, Beltsville Human Nutrition Research Center, ARS, USDA. https://doi.org/10.15482/USDAADC/1178141.
Stoffel W, LeKim D, Tschung TS. A simple chemical method for labelling phosphatidylcholine and sphingomyelin in the choline moiety. Hoppe Seylers Z Physiol Chem. 1971;352:1058–64.
Rossi M, Mascaretti F, Parpinel M, Serraino D, Crispo A, Celentano E, et al. Dietary intake of branched-chain amino acids and colorectal cancer risk. Br J Nutr. 2021;126:22–7.
Franceschi S, Negri E, Salvini S, Decarli A, Ferraroni M, Filiberti R, et al. Reproducibility of an Italian food frequency questionnaire for cancer studies: results for specific food items. Eur J Cancer. 1993;29A:2298–305.
Decarli A, Franceschi S, Ferraroni M, Gnagnarella P, Parpinel MT, La Vecchia C, et al. Validation of a food-frequency questionnaire to assess dietary intakes in cancer studies in Italy. Results for specific nutrients. Ann Epidemiol. 1996;6:110–8.
U.S. Department of Agriculture (USDA) ARSFC. Foundation Foods. Version Current: October 2020. Internet: fdc.nal.usda.gov. Accessed Feb 2021. 2020.
U.S. Department of Agriculture (USDA) ARSFC. Agricultural Research Service. FoodData Central, Agricultural Research Service. USDA Food and Nutrient Database for Dietary Studies 2017-2018.. Food Surveys Research Group Home Page, http://www.arsusdagov/nea/bhnrc/fsrg 2020.
US Department of Agriculture (USDA) ARS, Nutrient Data Laboratory. USDA National Nutrient Database for Standard Reference, Legacy. 2018.
Patterson KY, Bhagwat S, Williams JR, Howe JC, Holden JM, Zeisel SH, et al. USDA Database for the Choline Content of Common Foods, Release 2 (2008). 2015.
Lewis ED, Kosik SJ, Zhao YY, Jacobs RL, Curtis JM, Field CJ. Total choline and choline-containing moieties of commercially available pulses. Plant Foods Hum Nutr. 2014;69:115–21.
Richard C, Lewis E, Zhao Y-, Asomaning J, Jacobs R, Field C. Measurement of the total choline content in 48 commercial dairy products or dairy alternatives. J Food Compos Anal. 2016;45:1–8.
FAO/INFOODS. Guidelines for food Matching Version 1.2. Rome: FAO; 2012.
Gnagnarella P, Salvini S, Parpinel M. Food Composition Database for Epidemiological Studies in Italy by Version 1.2015 Website http://www.bda-ieo.it/. 2015.
Willett W, Stampfer MJ. Total energy intake: implications for epidemiologic analyses. Am J Epidemiol. 1986;124:17–27.
Cheng S, Long JS. Testing for IIA in the multinomial logit model. Sociol Method Res. 2007;35:583–600.
Cho E, Willett WC, Colditz GA, Fuchs CS, Wu K, Chan AT, et al. Dietary choline and betaine and the risk of distal colorectal adenoma in women. J Natl Cancer Inst. 2007;99:1224–31.
Youn J, Cho E, Lee JE. Association of choline and betaine levels with cancer incidence and survival: a meta-analysis. Clin Nutr. 2019;38:100–9.
Guertin KA, Li XS, Graubard BI, Albanes D, Weinstein SJ, Goedert JJ, et al. Serum Trimethylamine N-oxide, Carnitine, Choline, and Betaine in Relation to Colorectal Cancer Risk in the Alpha Tocopherol, Beta Carotene Cancer Prevention Study. Cancer Epidemiol Biomarkers Prev. 2017;26:945–52.
Myte R, Gylling B, Schneede J, Ueland PM, Haggstrom J, Hultdin J, et al. Components of One-carbon Metabolism Other than Folate and Colorectal Cancer Risk. Epidemiology. 2016;27:787–96.
Nitter M, Norgard B, de Vogel S, Eussen SJ, Meyer K, Ulvik A, et al. Plasma methionine, choline, betaine, and dimethylglycine in relation to colorectal cancer risk in the European Prospective Investigation into Cancer and Nutrition (EPIC). Ann Oncol. 2014;25:1609–15.
Bae S, Ulrich CM, Neuhouser ML, Malysheva O, Bailey LB, **ao L, et al. Plasma choline metabolites and colorectal cancer risk in the Women’s Health Initiative Observational Study. Cancer Res. 2014;74:7442–52.
Thomas AM, Manghi P, Asnicar F, Pasolli E, Armanini F, Zolfo M, et al. Metagenomic analysis of colorectal cancer datasets identifies cross-cohort microbial diagnostic signatures and a link with choline degradation. Nat Med. 2019;25:667–78.
Zhang CX, Pan MX, Li B, Wang L, Mo XF, Chen YM, et al. Choline and betaine intake is inversely associated with breast cancer risk: a two-stage case-control study in China. Cancer Sci. 2013;104:250–8.
Xu X, Gammon MD, Zeisel SH, Lee YL, Wetmur JG, Teitelbaum SL, et al. Choline metabolism and risk of breast cancer in a population-based study. FASEB J. 2008;22:2045–52.
Zeng FF, Xu CH, Liu YT, Fan YY, Lin XL, Lu YK, et al. Choline and betaine intakes are associated with reduced risk of nasopharyngeal carcinoma in adults: a case-control study. Br J Cancer. 2014;110:808–16.
Ying J, Rahbar MH, Hallman DM, Hernandez LM, Spitz MR, Forman MR, et al. Associations between dietary intake of choline and betaine and lung cancer risk. PLoS One. 2013;8:e54561.
Kotsopoulos J, Hankinson SE, Tworoger SS. Dietary betaine and choline intake are not associated with risk of epithelial ovarian cancer. Eur J Clin Nutr. 2010;64:111–4.
Richman EL, Kenfield SA, Stampfer MJ, Giovannucci EL, Zeisel SH, Willett WC, et al. Choline intake and risk of lethal prostate cancer: incidence and survival. Am J Clin Nutr. 2012;96:855–63.
Schwingshackl L, Hoffmann G. Adherence to Mediterranean diet and risk of cancer: a systematic review and meta-analysis of observational studies. Int J Cancer. 2014;135:1884–97.
Rosato V, Guercio V, Bosetti C, Negri E, Serraino D, Giacosa A, et al. Mediterranean diet and colorectal cancer risk: a pooled analysis of three Italian case-control studies. Br J Cancer. 2016;115:862–5.
Randi G, Edefonti V, Ferraroni M, La Vecchia C, Decarli A. Dietary patterns and the risk of colorectal cancer and adenomas. Nutr Rev. 2010;68:389–408.
Mahmoud AM, Ali MM. Methyl Donor Micronutrients that Modify DNA Methylation and Cancer Outcome. Nutrients. 2019;11:608.
Acknowledgements
The authors would like to thank Dr. Alfred H. Merrill Jr for his expert advice during the initial conceptualization of the study.
Funding
This work was supported by the Italian Foundation for Cancer Research (AIRC).
Author information
Authors and Affiliations
Contributions
MR and PB conceived the study; DS, EN, AG, AC, and CLV collected the original data; MR, FF, and MP developed the assessment of exposure, with input from YH and CL; MR analyzed the data, with input from MK, CLV, CP and PB; MR, MK and GC drafted the manuscript, with input from YH, CL, CLV and PB; all authors reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study protocol was submitted to the Board of Ethics of the participating hospitals and received the approval required at the time of data collection. The Ethics Committees of the Hospital “Niguarda Ca’ Granda”, Milan, provided the study approval (1125/194). Informed consent was obtained from all individual participants included in the study. This study was performed in line with the principles of the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rossi, M., Khalifeh, M., Fiori, F. et al. Dietary choline and sphingomyelin choline moiety intake and risk of colorectal cancer: a case-control study. Eur J Clin Nutr 77, 905–910 (2023). https://doi.org/10.1038/s41430-023-01298-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41430-023-01298-4
- Springer Nature Limited
This article is cited by
-
Dietary choline intake and health outcomes in U.S. adults: exploring the impact on cardiovascular disease, cancer prevalence, and all-cause mortality
Journal of Health, Population and Nutrition (2024)