Abstract
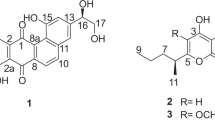
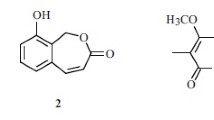
Two new chlorinated bis-indole alkaloids, dionemycin (1) and 6-OMe-7′,7″-dichorochromopyrrolic acid (2), along with seven known analogs 3–9, were isolated from the deep-sea derived Streptomyces sp. SCSIO 11791. Their structures were elucidated by extensive HRESIMS, and 1D and 2D NMR data analysis. In vitro antibacterial and cytotoxic assays revealed that, compound 1, shows anti-staphylococcal activity with an MIC range of 1–2 μg/mL against six clinic strains of methicillin-resistant Staphylococcus aureus (MRSA) isolated from human and pig. Additionally, compound 1 displayed cytotoxic activity against human cancer cell lines NCI-H460, MDA-MB-231, HCT-116, HepG2, and noncancerous MCF10A with an IC50 range of 3.1–11.2 μM. Analysis of the structure-activity relationship reveals that the chlorine atom at C-6″ could be pivotal for conferring their bioactivities, thus providing hints on chemical modifications on bis-indole alkaloid scaffold in drug design.


Similar content being viewed by others
References
Kam TS, Choo YM. Bisindole alkaloids. Alkaloids Chem Biol. 2006;63:181–337.
Veale CG, Davies-Coleman MT. Marine bi-, bis-, and trisindole alkaloids. Alkaloids Chem Biol. 2014;73:1–64.
Cordell GA. The bisindole alkaloids. In: Saxton JE editor. chemistry of heterocyclic compounds: indoles, part four, the monoterpenoid indole alkaloids. Vol 25. New York, NY: John Wiley & Sons, Inc.; 1983.
Rahman MT, Tiruveedhula VV, Cook JM. Synthesis of bisindole alkaloids from the apocynaceae which contain a macroline or sarpagine unit: a review. Molecules. 2016;21:1525–65.
Neuss N, Gorman M, Svoboda G, Maciak G, Beer C. Vinca alkaloids. III. 1. Characterization of leurosine and vincaleukoblastine, new alkaloids from Vinca rosea. J Am Chem Soc. 1959;81:4754–5.
Neuss N, Neuss MN. Therapeutic use of bisindole alkaloids from catharanthus. Alkaloids Chem Pharmacol. 1990;37:229–40.
Yasuzawa T, et al. The structures of the novel protein kinase C inhibitors K-252a, b, c and d. J Antibiot. 1986;39:1072–8.
Bush JA, Long BH, Catino JJ, Bradner WT, Tomita K. Production and biological activity of rebeccamycin, a novel antitumor agent. J Antibiot. 1987;40:668–78.
Omura S, Sasaki Y, Iwai Y, Takeshima H. Staurosporine, a potentially important gift from a microorganism. J Antibiot. 1995;48:535–48.
Long BH, Rose WC, Vyas DM, Matson JA, Forenza S. Discovery of antitumor indolocarbazoles: rebeccamycin, NSC 655649, and fluoroindolocarbazoles. Curr Med Chem Anticancer Agents. 2002;2:255–66.
Lien WC, et al. 7-hydroxy-staurosporine, UCN-01, induces DNA damage response, and autophagy in human osteosarcoma U2-OS cells. J Cell Biochem. 2018;119:4729–41.
Gribble GW. In: Knöelker H-J, editor. Occurrence of halogenated alkaloids. The alkaloids; Academic Press: London, 2012. pp. 1–165.
Salucci S, et al. Marine bisindole alkaloid: a potential apoptotic inducer in human cancer cells. Eur J Histochem. 2018;62:2881.
Bifulco G, Bruno I, Riccio R, Lavayre J, Bourdy G. Further brominated bis-Indole and bris-Indole alkaloids from the deep-water new-caledonian marine sponge Orina sp. J Nat Prod. 1995;58:1254–60.
Veale CG, et al. Synthetic analogues of the marine bisindole deoxytopsentin: potent selective inhibitors of MRSA pyruvate kinase. J Nat Prod. 2015;78:355–62.
Manivasagan P, Kang KH, Sivakumar K, Li-Chan EC, Oh HM, Kim SK. Marine actinobacteria: an important source of bioactive natural products. Environ Toxicol Pharm. 2014;38:172–88.
Zhang W, et al. Spiroindimicins A-D: new bisindole alkaloids from a deep-sea-derived actinomycete. Org Lett. 2012;14:3364–7.
Zhang W, et al. Indimicins A-E, bisindole alkaloids from the deep-sea-derived Streptomyces sp. SCSIO 03032. J Nat Prod. 2014;77:1887–92.
Barth H, et al. Preparation of 3,4-bis(indol-3-yl) maleimides as protein kinase C inhibitors. Germany, Patent 90108468.1, 1990.
McArthur KA, et al. Lynamicins A-E, chlorinated bisindole pyrrole antibiotics from a novel marine actinomycete. J Nat Prod. 2008;71:1732–7.
Du YL, Ryan KS. Expansion of bisindole biosynthetic pathways by combinatorial construction. Acs Synth Biol. 2015;4:682–8.
Mitchell SS, et al. Nereus Pharmaceuticals, Inc., USA. Bis-indole pyrroles useful as antimicrobial agents. USA, PTC 002039, 2005.
Onaka H, Taniguchi S, Igarashi Y, Furumai T. Characterization of the biosynthetic gene cluster of rebeccamycin from Lechevalieria aerocolonigenes ATCC 39243. Biosci Biotechnol Biochem. 2003;67:127–38.
Song Y, et al. Cytotoxic and antibacterial angucycline- and prodigiosin-analogues from the deep-sea derived Streptomyces sp. SCSIO 11594. Mar Drugs. 2015;13:1304–16.
Song Y, et al. Cytotoxic and antibacterial marfuraquinocins from the deep South China Sea-derived Streptomyces niveus SCSIO 3406. J Nat Prod. 2013;76:2263–8.
Ding B, et al. New dimeric members of the phomoxanthone family: phomolactonexanthones A, B and deacetylphomoxanthone C isolated from the fungus Phomopsis sp. Mar Drugs. 2013;11:4961–72.
CLSI. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; approved standard—ninth edition. CLSI document M07-A9. Wayne, PA: clinical and laboratory standards institute; 2012.
Acknowledgements
This study was supported, in part, by the Program of Guangzhou Science and Technology Plan (201707010454), the National Natural Science Foundation of China (41676151), Key Special Project for Introduced Talents Team of Southern Marine Science and Engineering Guangdong Laboratory (Guangzhou) (GML2019ZD0406), the National Key Research and Development Program of China (2017YFD0201401) and the Special Fund for Strategic Pilot Technology of Chinese Academy of Sciences (XDA13020302-2). We thank the analytical facility center of the South China Sea Institute of Oceanology, Dr. Zhihui **ao and Mr. Chuanrong Li for recording NMR data and Ms. Aijun Sun, and Ms. Yun Zhang for acquisition of ESIMS and HRESIMS data. Additionally, we appreciate Professor Jianhua Liu from South China Agricultural University and our colleague Professor Chang Chen for their gift of pathogenic bacteria MRSA GDQ6P012P and GDE4P037P and four Vibrio sp. strains, respectively.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Dedicated to Professor William Fenical in recognition of his contributions to marine derived secondary metabolites.
Supplementary information
Rights and permissions
About this article
Cite this article
Song, Y., Yang, J., Yu, J. et al. Chlorinated bis-indole alkaloids from deep-sea derived Streptomyces sp. SCSIO 11791 with antibacterial and cytotoxic activities. J Antibiot 73, 542–547 (2020). https://doi.org/10.1038/s41429-020-0307-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41429-020-0307-4
- Springer Japan KK
This article is cited by
-
Repurposing of Strychnine as the Potential Inhibitors of Aldo–keto Reductase Family 1 Members B1 and B10: Computational Modeling and Pharmacokinetic Analysis
The Protein Journal (2024)
-
Sulfoxanthicillin from the deep-sea derived Penicillium sp. SCSIO sof101: an antimicrobial compound against Gram-positive and -negative pathogens
The Journal of Antibiotics (2023)
-
Bioactive Microbial Metabolites in Cancer Therapeutics: Mining, Repurposing, and Their Molecular Targets
Current Microbiology (2022)