Abstract
Background
Central venous access devices (CVAD) are associated with central line associated bloodstream infection (CLABSI) and venous thromboembolism (VTE). We identified trends in non-intensive care unit (ICU) CVAD utilization, described complication rates, and compared resources between low and high CVAD sites.
Methods
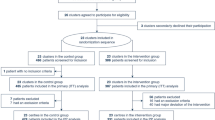
We combined data from the Pediatric Health Information System (PHIS) database and surveys from included hospitals. We analyzed 10-year trends in CVAD encounters for non-ICU children between 01/2012-12/2021 and described variation and complication rates between 01/2017-12/2021. Using Fisher’s exact test, we compared resources between low and high CVAD users.
Results
CVAD use decreased from 6.3% to 3.8% of hospitalizations over 10 years. From 2017–2021, 67,830 encounters with CVAD were identified. Median age was 7 (IQR 2–13) years; 46% were female. Significant variation in CVAD utilization exists (range 1.4–16.9%). Rates of CLABSI and VTE were 4.0% and 3.4%, respectively. Survey responses from 33/41 (80%) hospitals showed 91% had vascular access teams, 30% used vascular access selection guides, and 70% used midline/long peripheral catheters. Low CVAD users were more likely to have a team guiding device selection (100% vs 43%, p = 0.026).
Conclusions
CVAD utilization decreased over time. Significant variation in CVAD use remains and may be associated with hospital resources.
Impact
-
Central venous access device (CVAD) use outside of the ICU is trending down; however, significant variation exists between institutions.
-
Children with CVADs hospitalized on the acute care units had a CLABSI rate of 4% and VTE rate of 3.4%.
-
91% of surveyed institutions have a vascular access team; however, the services provided vary between institutions.
-
Even though 70% of the surveyed institutions have the ability to place midline/long peripheral catheters, the majority use these catheters less than a few times per month.
-
Institutions with low CVAD use are more likely to have a vascular access team that guides device selection.

Similar content being viewed by others
Data availability
The dataset analyzed during this study are available from the Pediatric Health Information System (PHIS), but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission from PHIS.
References
Ullman, A. J. et al. The Michigan appropriateness guide for intravenous catheters in pediatrics: miniMAGIC. Pediatrics 145, S269–S284 (2020).
Lyren, A. et al. Children’s hospitals’ solutions for patient safety collaborative impact on hospital-acquired harm. Pediatrics 140, e20163494 (2017).
Goudie, A., Dynan, L., Brady, P. W. & Rettiganti, M. Attributable cost and length of stay for central line-associated bloodstream infections. Pediatrics 133, e1525–e1532 (2014).
Goudie, A. et al. Costs of venous thromboembolism, catheter-associated urinary tract infection, and pressure ulcer. Pediatrics 136, 432–439 (2015).
Paterson, R. S. et al. Selection and insertion of vascular access devices in pediatrics: a systematic review. Pediatrics 145, S243–S268 (2020).
Burek, A. G. et al. The development of a long peripheral catheter program at a large pediatric academic center: a pilot study. Hosp. Pediatr. 10, 897–901 (2020).
Gibson, C. et al. Peripherally inserted central catheters: use at a tertiary care pediatric center. J. Vasc. Inter. Radio. 24, 1323–1331 (2013).
House, S. A. et al. Development and use of a calculator to measure pediatric low-value care delivered in US children’s hospitals. JAMA Netw. Open 4, 1–13 (2021).
Pernar, L. I. M., Wolf, L. L., Seshadri, A. & Patel, V. Impact of a surgeon-led peripherally inserted central venous catheter team on peripherally inserted central venous catheter-related complications and costs. Surg. Infect. (Larchmt.) 17, 352–356 (2016).
Swaminathan, L. et al. Improving PICC use and outcomes in hospitalised patients: an interrupted time series study using MAGIC criteria. BMJ Qual. Saf. 27, 271–278 (2018).
DeVries, M., Lee, J. & Hoffman, L. Infection free midline catheter implementation at a community hospital (2 years). Am. J. Infect. Control 47, 1118–1121 (2019).
Moureau, N., Sigl, G. & Hill, M. How to establish an effective midline program: a case study of 2 hospitals. J. Assoc. Vasc. Access. 20, 179–188 (2015).
**ong, Z. & Chen, H. Interventions to reduce unnecessary central venous catheter use to prevent central-line-associated bloodstream infections in adults: a systematic review. Infect. Control Hosp. Epidemiol. 39, 1442–1448 (2018).
von Elm, E. et al. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: Guidelines for reporting observational studies. Int. J. Surg. 12 https://doi.org/10.1016/j.ijsu.2014.07.013 (2014).
Willer, B. L., Tobias, J. D., Suttle, M. L., Nafiu, O. O. & Mpody, C. Trends of racial/ethnic disparities in pediatric central line-associated bloodstream infections. Pediatrics 150, e2021054955 (2022).
Dillman, D. A., Smyth, J. D. & Christian, L. M. Internet, Phone, Mail and Mixed-Mode Survey: The Tailored Design Survey. 4th edn (Wiley, 2014).
Willis, G. B. Analysis of the cognitive interview in questionnaire design: understanding qualitative research. Oxford Univ. Press. Published online 2015).
Burek, A. G. et al. Long peripheral catheters in children: a sco** review. J. Hosp. Med. 1–10 https://doi.org/10.1002/jhm.12968 (2022).
Sankaran, R. et al. Changes in hospital safety following penalties in the US Hospital acquired condition reduction program: retrospective cohort study. BMJ 366, 4109 (2019).
Solutions for Patient Safety. 2022. https://www.solutionsforpatientsafety.org/our-network-hospitals. Accessed May 16, 2024.
Tripathi, S. et al. Compliance with central line maintenance bundle and infection rates. Pediatrics 152, e2022059688 (2023).
Woods, C. R. et al. Clinical Practice Guideline by the Pediatric Infectious Diseases Society and the Infectious Diseases Society of America: 2021 Guideline on Diagnosis and Management of Acute Hematogenous Osteomyelitis in Pediatrics. J. Pediatr. Infect. Dis. Soc. 10, 801–844 (2021).
van den Bosch, C. H. et al. Central venous catheter–associated complications in pediatric patients diagnosed with Hodgkin lymphoma: implications for catheter choice. Support Care Cancer 30, 8069–8079 (2022).
Zhang, H. et al. Comparison of peripherally inserted central catheters (PICCs) versus totally implantable venous-access ports in pediatric oncology patients, a single center study. Sci. Rep. 12, 3510 (2022).
Barrier, A., Williams, D. J., Connelly, M. & Buddy Creech, C. Frequency of peripherally inserted central catheter complications in children. Pediatr. Infect. Dis. J. 31, 519–521 (2012).
Ullman, A. J., Marsh, N., Mihala, G., Cooke, M. & Rickard, C. M. Complications of central venous access devices: a systematic review. Pediatrics 136, e1331–e1344 (2015).
McGrath, C. L. et al. Identifying and mitigating disparities in central line-associated bloodstream infections in minoritized racial, ethnic, and language groups. JAMA Pediatr. 177, 700–709 (2023).
Parikh, K. et al. Disparities in racial, ethnic, and payer groups for pediatric safety events in US hospitals. Pediatrics 153, e2023063714 (2024).
Lyren, A. et al. Racial and ethnic disparities in common inpatient safety outcomes in a children’s hospital cohort. BMJ Qual. Saf. 33, 86–97 (2023).
Kleidon, T. M. et al. Comparison of midline catheters and peripherally inserted central catheters to reduce the need for general anesthesia in children with respiratory disease: a feasibility randomized controlled trial. Pediatr. Anesth. 31, 985–995 (2021).
Coon, E. R. et al. Shortened IV antibiotic course for uncomplicated, late-onset group B streptococcal bacteremia. Pediatrics 142, e20180345 (2018).
Funding
A.G.B. work on this study was in part completed while participating in a 2-year Clinical Research Scholar Program funded by a training grant (UL1 TR001436).
Author information
Authors and Affiliations
Contributions
A.G.B. conceptualized and designed the study, led data summary, analysis and interpretation; drafted and revised the manuscript. C.B. contributed to the design of the study, performed PHIS data retrieval and harmonization, participated in data analysis and interpretation, critically reviewed and revised the manuscript. T.L. and E.R.C., K.P. contributed to the design of the study, participated in data analysis and interpretation, critically reviewed and revised the manuscript. A.Y.P. and M.L. contributed to study design, performed the quantitative statistical analysis, reviewed and revised the manuscript. K.E.F., A.J.U. and D.C.B. contributed to the conceptualization and the study design, reviewed the data analysis plan and data output, and critically reviewed and revised the manuscript. All authors approved the final manuscript as submitted.
Corresponding author
Ethics declarations
Competing interests
Kathryn Flynn has received consultancy funding from Inhibikase, Pfizer, and Novartis unrelated to this work. Amanda Ullman’s employer (UQ) has received investigator-initiated research grants from vascular access product manufacturers (3 M, Becton Dickinson, Medline) on behalf of her research, unrelated to the submitted project. David Brousseau receives consultancy funding from CSL Behring unrelated to this work. The remaining authors have no conflicts of interest to disclose.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Burek, A.G., Bumgardner, C., Liljestrom, T. et al. Use of central venous access devices outside of the pediatric intensive care units. Pediatr Res (2024). https://doi.org/10.1038/s41390-024-03337-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41390-024-03337-7
- Springer Nature America, Inc.