Abstract
Background
The relationship between infant colic and breast milk beta-endorphin (BE) and relaxin-2 (RLX-2) has not been studied before.
Methods
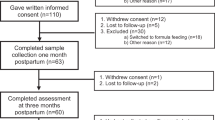
Thirty colic infants and their mothers constituted the study group, and the same sex, similar age and healthy infants and their mothers formed the control group. Maternal predisposing factors were analysed with questionnaires.
Results
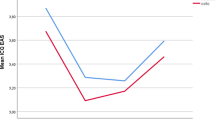
The frequency of headache and myalgia in the mothers was significantly higher in the study group compared to the control group. Sleep quality of mothers in the study group was worse than in the control group (p = 0.028). While breast milk RLX-2 level in the study group was not different from the control group, breast milk BE level in the study group was significantly higher than the control group (p = 0.039). A positive correlation was found between breast milk BE levels and crying times, and between sleep quality scores and crying times. Headache, myalgia, sleep quality and breast milk BE levels were found to have a significant effect on infant colic.
Conclusions
Breast milk RLX-2 has no role on infant colic. Breast milk BE may act as a biological mediator in transmitting of maternal predisposing factors such as poor sleep quality, headache and myalgia from mother to infant.
Impact
-
The relationship between infant colic and breast milk beta-endorphin (BE) and elaxin-2 (RLX-2) has not been studied before.
-
Maternal sleep quality, headache, and myalgia are predisposing factors associated with infant colic.
-
Breast milk RLX-2 has no effect on infant colic.
-
Breast milk BE may play a role as a biological mediator in transmitting the effects of predisposing factors from mother to infant.
-
Breast milk BE may be a mediator in biological communication between mother and infant.


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Zeevenhooven, J., Koppen, I. J. & Benninga, M. A. The new Rome IV criteria for functional gastrointestinal disorders in infants and toddlers. Pediatr. Gastroenterol. Hepatol. Nutr. 20, 1–13 (2017).
Lam, T. M. L., Chan, P. C. & Goh, L. H. Approach to infantile colic in primary care. Singap. Med J. 60, 12–16 (2019).
Zeevenhooven, J. et al. Infant colic: mechanisms and management. Nat. Rev. Gastroenterol. Hepatol. 15, 479–496 (2018).
Doneray, H., Orbak, Z. & Yildiz, L. The relationship between breast milk leptin and neonatal weight gain. Acta Paediatr. 98, 643–647 (2009).
Doneray, H., Olcaysu, E., Yildirim, A. & Ozden, A. The effect of the zinc concentration in breast milk on neonatal weight gain. J. Trace Elem. Med Biol. 41, 32–35 (2017).
Pilozzi, A., Carro, C. & Huang, X. Roles of β-endorphin in stress, behavior, neuroinflammation, and brain energy metabolism. Int J. Mol. Sci. 22, 338 (2020).
Huidobro-Toro, J. P. & Way, E. L. Contractile effect of morphine and related opioid alkaloids, beta-endorphin and methionine enkephalin on the isolated colon from Long Evans rats. Br. J. Pharm. 74, 681–694 (1981).
Nijkamp, F. P. & Van Ree, J. M. Effects of endorphins on different parts of the gastrointestinal tract of rat and guinea-pig in vitro. Br. J. Pharm. 68, 599–606 (1980).
Ohkawa, H. Effects of enkephalin and endorphin on the inhibitory junction potentials in the duodenal smooth muscle cells of the guinea-pig. Jpn J. Physiol. 34, 89–102 (1984).
Moulin, D. E. et al. Randomised trial of oral morphine for chronic non-cancer pain. Lancet 347, 143–147 (1996).
Borland, M. L. et al. Intranasal fentanyl is an equivalent analgesic to oral morphine in paediatric burns patients for dressing changes: a randomised double blind crossover study. Burns 31, 831–837 (2005).
Poonai, N. et al. Oral administration of morphine versus ibuprofen to manage postfracture pain in children: a randomized trial. CMAJ 186, 1358–1363 (2014).
Ivell, R. & Anand-Ivell, R. Neohormones in milk. Best. Pr. Res Clin. Endocrinol. Metab. 31, 419–425 (2017).
Nair, V. B. et al. Human relaxin-2: historical perspectives and role in cancer biology. Amino Acids 43, 1131–1140 (2012).
Ganella, D. E., Ma, S. & Gundlach, A. L. Relaxin-3/RXFP3 Signaling and neuroendocrine function - A perspective on extrinsic hypothalamic control. Front Endocrinol. (Lausanne) 4, 128 (2012).
Bongers-Binder, S. et al. Distribution of immunoreactive relaxin in the genital tract and in the mammary gland of non-pregnant women. Clin. Exp. Obstet. Gynecol. 18, 161–164 (1991).
Steinetz, B. G., Horton, L. & Lasano, S. The source and secretion of immunoactive relaxin in rat milk. Exp. Biol. Med. (Maywood) 234, 562–565 (2009).
Squecco, R. et al. Relaxin affects smooth muscle biophysical properties and mechanical activity of the female mouse colon. Endocrinology 156, 4398–4410 (2015).
Beck, A. T., Steer, R. A. & Carbin, M. G. Psychometric properties of the Beck Depression Inventory: twenty-five years of evaluation. Clin. Psychol. Rev. 8, 77–100 (1988).
Beck, A. T., Epstein, N., Brown, G. & Steer, R. A. An inventory for measuring clinical anxiety: psychometric properties. J. Consult Clin. Psychol. 56, 893–897 (1988).
Leyfer, O. T., Ruberg, J. L. & Woodruff-Borden, J. Examination of the utility of the Beck Anxiety Inventory and its factors as a screener for anxiety disorders. J. Anxiety Disord. 20, 444–458 (2006).
Mollayeva, T. et al. The Pittsburgh sleep quality index as a screening tool for sleep dysfunction in clinical and non-clinical samples: a systematic review and meta-analysis. Sleep. Med. Rev. 25, 52–73 (2016).
Cole, J. C. et al. Validation of a 3-factor scoring model for the Pittsburgh sleep quality index in older adults. Sleep 29, 112–116 (2006).
Misra, U. K., Kalita, J., Tripathi, G. M. & Bhoi, S. K. Is β endorphin related to migraine headache and its relief? Cephalalgia 33, 316–322 (2013).
Anselmi, B. et al. Serum betaendorphin increase after intravenous histamine treatment of chronic daily headache. Recent. Prog. Med 88, 321–324 (1997).
Bach, F. W. Beta-endorphin in migraine. Cephalalgia 12, 390–391 (1992).
Genazzani, A. R. et al. Progressive impairment of CSF beta-EP levels in migraine sufferers. Pain 18, 127–133 (1984).
Bach, F. W., Langemark, M., Secher, N. H. & Olesen, J. Plasma and cerebrospinal fluid beta-endorphin in chronic tension-type headache. Pain 51, 163–168 (1992).
Leone, M. et al. Beta-endorphin concentrations in the peripheral blood mononuclear cells of migraine and tension-type headache patients. Cephalalgia 12, 154–157 (1992).
Bach, F. W. et al. Effect of sulpiride or paroxetine on cerebrospinal fluid neuropeptide concentrations in patients with chronic tension-type headache. Neuropeptides 27, 129–136 (1994).
Franceschini, R. et al. Evaluation of beta-endorphin secretion in patients suffering from episodic cluster headache. Headache 36, 603–607 (1996).
Bidari, A. et al. The acute effect of maximal exercise on plasma beta-endorphin levels in fibromyalgia patients. Korean J. Pain. 29, 249–254 (2016).
Panerai, A. E. et al. Peripheral blood mononuclear cell beta-endorphin concentration is decreased in chronic fatigue syndrome and fibromyalgia but not in depression: preliminary report. Clin. J. Pain. 18, 270–273 (2002).
Vaeroy, H. et al. Cerebrospinal fluid levels of beta-endorphin in patients with fibromyalgia (fibrositis syndrome). J. Rheumatol. 15, 1804–1806 (1988).
Yunus, M. B., Denko, C. W. & Masi, A. T. Serum beta-endorphin in primary fibromyalgia syndrome: a controlled study. J. Rheumatol. 13, 183–186 (1986).
Hamaty, D. et al. The plasma endorphin, prostaglandin and catecholamine profile of patients with fibrositis treated with cyclobenzaprine and placebo: a 5-month study. J. Rheumatol. Suppl. 19, 164–168 (1989).
Younger, J. W., Zautra, A. J. & Cummins, E. T. Effects of naltrexone on pain sensitivity and mood in fibromyalgia: no evidence for endogenous opioid pathophysiology. PLoS One 4, e5180 (2009).
King, C., Masserano, J. M., Codd, E. & Byrne, W. L. Effects of beta-endorphin and morphine on the sleep-wakefulness behavior of cats. Sleep 4, 259–262 (1981).
Song, J. et al. Sleep and anesthesia. Sleep. Med. Res. 9, 11–19 (2018).
Dent, R. R. et al. Diurnal rhythm of plasma immunoreactive beta-endorphin and its relationship to sleep stages and plasma rhythms of cortisol and prolactin. J. Clin. Endocrinol. Metab. 52, 942–947 (1981).
Gessa, G. L., Pani, L., Fadda, P. & Fratta, W. Sleep deprivation in the rat: an animal model of mania. Eur. Neuropsychopharmacol. 5, 89–93 (1995).
Fratta, W. et al. Stress-induced insomnia: opioid-dopamine interactions. Eur. J. Pharm. 142, 437–440 (1987).
Przewłocka, B. et al. Deprivation of REM sleep in the rat and the opioid peptides beta-endorphin and dynorphin. Neurosci. Lett. 70, 138–142 (1986).
Covelli, V. et al. Interleukin-1 beta and beta-endorphin circadian rhythms are inversely related in normal and stress-altered sleep. Int J. Neurosci. 63, 299–305 (1992).
Zanardo, V. et al. Labor pain effects on colostral milk beta-endorphin concentrations of lactating mothers. Biol. Neonate 79, 87–90 (2001).
Ferrando, T. et al. Beta-endorphin-like and alpha-MSH-like immunoreactivities in human milk. Life Sci. 47, 633–635 (1990).
Banks, W. A., Kastin, A. J. & Coy, D. H. Delta sleep-inducing peptide (DSIP)-like material is absorbed by the gastrointestinal tract of the neonatal rat. Life Sci. 33, 1587–1597 (1983).
Dick, P., Grandjean, M. E. & Tissot, R. Successful treatment of withdrawal symptoms with delta sleep-inducing peptide, a neuropeptide with potential agonistic activity on opiate receptors. Neuropsychobiology 10, 205–208 (1983).
Murphy, P. B., Bechmann, S. & Barrett, M. J. Morphine. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan. 2022 May 9
Benyamin, R. et al. Opioid complications and side effects. Pain. Physician 11, S105–S120 (2008).
Leache, L., Aquerreta, I., Moraza, L. & Ortega, A. Morphine-induced hearing loss. Am. J. Health Syst. Pharm. 73, 1840–1843 (2016).
Funding
This study was supported by a grant coded TAB-2021-9686 from Atatürk University Scientific Research Project Office.
Author information
Authors and Affiliations
Contributions
H.D., G.T., A.O., N.O.: substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; drafting the article or revising it critically for important intellectual content; and final approval of the version to be published. H.D.: drafting the article or revising it critically for important intellectual content; and final approval of the version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Atatürk University Faculty of Medicine Ethics Committee approved the study, and the infants’ parents gave written informed consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Doneray, H., Tavlas, G., Ozden, A. et al. The role of breast milk beta-endorphin and relaxin-2 on infant colic. Pediatr Res 94, 1416–1421 (2023). https://doi.org/10.1038/s41390-023-02617-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-023-02617-y
- Springer Nature America, Inc.