Abstract
Cerebral infarction is the most common type of stroke and often causes long-term disability. To investigate the genetic contribution to cerebral infarction, we conducted a case-control study using 52,608 gene-based tag SNPs selected from the JSNP database. Here we report that a nonsynonymous SNP in a member of protein kinase C (PKC) family, PRKCH, was significantly associated with lacunar infarction in two independent Japanese samples (P = 5.1 × 10−7, crude odds ratio of 1.40). This SNP is likely to affect PKC activity. Furthermore, a 14-year follow-up cohort study in Hisayama (Fukuoka, Japan) supported involvement of this SNP in the development of cerebral infarction (P = 0.03, age- and sex-adjusted hazard ratio of 2.83). We also found that PKCη was expressed mainly in vascular endothelial cells and foamy macrophages in human atherosclerotic lesions, and its expression increased as the lesion type progressed. Our results support a role for PRKCH in the pathogenesis of cerebral infarction.




Similar content being viewed by others
References
Kiyohara, Y. et al. Ten-year prognosis of stroke and risk factors for death in a Japanese community: the Hisayama study. Stroke 34, 2343–2348 (2003).
Kubo, M. et al. Trends in the incidence, mortality, and survival rate of cardiovascular disease in a Japanese community: the Hisayama study. Stroke 34, 2349–2354 (2003).
National Institute of Neurological Disorders and Stroke. Special report from the National Institute of Neurological Disorders and Stroke: classification of cerebrovascular disease III. Stroke 21, 637–676 (1990).
Flossmann, E., Schulz, U.G.R. & Rothwell, P.M. Systematic review of methods and results of studies of the genetic epidemiology of ischemic stroke. Stroke 35, 212–227 (2004).
Hirschhorn, J.N. & Daly, M.J. Genome-wide association studies for common diseases and complex traits. Nat. Rev. Genet. 6, 95–108 (2005).
Ozaki, K. et al. Functional SNPs in the lymphotoxin-α gene that are associated with susceptibility to myocardial infarction. Nat. Genet. 32, 650–654 (2002).
Suzuki, A. et al. Functional haplotypes of PADI4, encoding citrullinating enzyme peptidylarginine deiminase 4, are associated with rheumatoid arthritis. Nat. Genet. 34, 395–402 (2003).
Kochi, Y. et al. A functional variant in FCRL3, encoding Fc receptor-like 3, is associated with rheumatoid arthritis and several autoimmunities. Nat. Genet. 37, 478–485 (2005).
Yamazaki, K. et al. Single nucleotide polymorphisms in TNFSF15 confer susceptibility to Crohn's disease. Hum. Mol. Genet. 14, 3499–3506 (2005).
Gretarsdottir, S. et al. The gene encoding phosphsdiesterase 4D confers risk of ischemic stroke. Nat. Genet. 35, 131–138 (2003).
Tsunoda, T. et al. Variation of gene-based SNPs and linkage disequilibrium patterns in the human genome. Hum. Mol. Genet. 13, 1623–1632 (2004).
Pritchard, J.K. & Rosenberg, N.A. Use of unlinked genetic markers to detect population stratification in association studies. Am. J. Hum. Genet. 65, 220–228 (1999).
Osada, S. et al. A phorbol ester receptor/protein kinase, nPKCη, a new member of the protein kinase C family predominantly expressed in lung and skin. J. Biol. Chem. 265, 22434–22440 (1990).
Osada, S. et al. Predominant expression of nPKCη, a Ca2+-independent isoform of protein kinase C in epithelial tissues, in association with epithelial differentiation. Cell Growth Differ. 4, 167–175 (1993).
Stary, H.C. et al. A definition of advanced types of atherosclerotic lesions and a histological classification of atherosclerosis. A report from the Committee on Vascular Lesions of the Council on Arteriosclerosis, American Heart Association. Circulation 92, 1355–1374 (1995).
Spitaler, M. & Cantrell, D.A. Protein kinase C and beyond. Nat. Immunol. 5, 785–790 (2004).
Poole, A.W., Pula, G., Hers, I., Crosby, D. & Jones, M.L. PKC-interacting proteins: from function to pharmacology. Trends Pharmacol. Sci. 25, 528–535 (2004).
Li, A.C. & Glass, C.K. The macrophage form cell as a target for therapeutic intervention. Nat. Med. 8, 1235–1242 (2002).
Pham, T.N., Brown, B.L., Dobson, P.R. & Richardson, V.J. Protein kinase C-eta (PKCη) is required for the development of inducible nitric oxide synthase (iNOS) positive phenotype in human monocytic cells. Nitric Oxide 9, 123–134 (2003).
Chen, C.C., Wang, J.K., Chen, W.C. & Lin, S.B. Protein kinase C η mediates lipopolysaccharide-induced nitric-oxide synthase expression in primary astrocytes. J. Biol. Chem. 273, 19424–19430 (1998).
Fima, E. et al. PKCη enhances cell cycle progression, the expression of G1 cyclins and p21 in MCF-7 cells. Oncogene 20, 6794–6804 (2001).
Matsumura, M., Tanaka, N., Kuroki, T., Ichihashi, M. & Ohba, M. The η isoform of protein kinase C inhibits UV-induced activation of caspase-3 in normal human keratinocytes. Biochem. Biophys. Res. Commun. 303, 350–356 (2003).
Aeder, S.E., Martin, P.M., Soh, J.W. & Hussaini, I.M. PKC-η mediated glioblastoma cell proliferation through the Akt and mTOR signaling pathways. Oncogene 23, 9062–9069 (2004).
Kubo, M. et al. Decreasing incidence of lacunar vs. other types of cerebral infarction in a Japanese population. Neurology 66, 1539–1544 (2006).
Cerebral Embolism Task Force. Cardiogenic brain embolism. Arch. Neurol. 43, 71–84 (1986).
Adams, H.P., Jr. et al. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org10172 in acute stroke treatment. Stroke 24, 35–41 (1993).
Ohnishi, Y. et al. A high-throughout SNP ty** system for genome-wide association studies. J. Hum. Genet. 46, 471–477 (2001).
Ikuta, T. et al. Cholesterol sulfate, a novel activator for the η isoform of protein kinase C. Cell Growth Differ. 5, 943–947 (1994).
Nakano, T. et al. Angiogenesis and lymphangiogenesis and expression of lymphangiogenic factors in the atherosclerotic intima of human coronary arteries. Hum. Pathol. 36, 330–340 (2005).
Masso-Welch, P.A. et al. Altered expression and localization of PKC eta in human breast tumors. Breast Cancer Res. Treat. 68, 211–223 (2001).
Acknowledgements
We thank the residents of Hisayama and the patients with cerebral infarction for their participation; T. Omae and the staff of the Division of Health and Welfare of Hisayama for their cooperation and many members of the Hisayama study for assistance. For collecting clinical samples, we thank T. Ago, H. Ooboshi, M. Kamouchi, H. Sugimori, J. Kuroda, Y. Kumai, N. Hagiwara and S. Yoshimura (Kyushu University Hospital); K. Tamaki and Y. Wakugawa (Hakujyuji Hospital); K. Fujii (Fukuoka Red Cross Hospital); Y. Okada and K. Toyoda (National Hospital Organization, Kyushu Medical Center); T. Nagao (Imazu Red Cross Hospital); H. Nakane (National Hospital Organization, Fukuoka Higashi Medical Center) and Y. Yamashita and K. Kusuda (Seiai Rehabilitation Hospital). We thank K. Chida (Department of Animal Resource Sciences, Graduate School of Agricultural and Life Sciences, The University of Tokyo) for technical advice. This study was supported in part by a Special Coordination Fund for Promoting Science and a Fund for Technology and Innovative Development Project in Life Sciences from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Contributions
M.K., J.H., T.M. and K. Yamazaki performed SNP genoty**; T. Ninomiya and K. Yonemoto provided Hisayama samples and carried out statistical analyses; T. Nakano and K.S. performed immunohistochemical experiments; M.K., K. Yamazaki and K.M. performed protein blotting experiments; Y.O. and S.S. performed genoty** of genome-wide screening samples; T.K. and S.I. provided clinical information and samples of individuals with cerebral infarction; Y.N. provided BioBank Japan samples; M.K. performed all other experiments and wrote the manuscript with contributions from K.S., Y.N. and Y.K.; T. Ninomiya, K. Yonemoto, Y.K. helped with revisions and M.I., Y.N. and Y.K. jointly directed the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Stratification analysis of screening samples. (PDF 628 kb)
Supplementary Fig. 2
Kaplan-Meier estimate of the incidence of lacunar infarction by 1425G→A genotype in the Hisayama study. (PDF 574 kb)
Supplementary Fig. 3
Age- and sex-adjusted cumulative incidence of coronary heart disease by 1425G→A genotype in the Hisayama study. (PDF 571 kb)
Supplementary Fig. 4
Relative mRNA expression of PKCη in various human tissues. (PDF 620 kb)
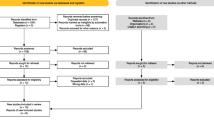
Supplementary Fig. 5
Flow diagram of the study. (PDF 609 kb)
Supplementary Table 1
Case-control association results of 45 SNPs in PRKCH for lacunar infarction. (PDF 126 kb)
Supplementary Table 2
Multivariate logistic analysis of nonsynonymous SNPs in PRKCH among cases with lacunar infarction and age- and sex-matched controls. (PDF 118 kb)
Supplementary Table 3
Clinical characteristics of the study population in the case-control study. (PDF 114 kb)
Rights and permissions
About this article
Cite this article
Kubo, M., Hata, J., Ninomiya, T. et al. A nonsynonymous SNP in PRKCH (protein kinase C η) increases the risk of cerebral infarction. Nat Genet 39, 212–217 (2007). https://doi.org/10.1038/ng1945
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1945
- Springer Nature America, Inc.
This article is cited by
-
Exploring the prognostic significance of PKCε variants in cervical cancer
BMC Cancer (2023)
-
Analyzing PKC Gamma (+ 19,506 A/G) polymorphism as a promising genetic marker for HCV-induced hepatocellular carcinoma
Biomarker Research (2022)
-
Single nucleotide polymorphisms in the ANGPTL4 gene and the SNP-SNP interactions on the risk of atherosclerotic Ischaemic stroke
BMC Neurology (2021)
-
Association of genetic variants of RNF213 with ischemic stroke risk in Koreans
Genes & Genomics (2021)
-
Monogenic, Polygenic, and MicroRNA Markers for Ischemic Stroke
Molecular Neurobiology (2019)