Abstract
Background:
This study investigated the clinical relevance and prognostic impact of the overall expression of programmed cell death protein ligand-1 (PD-L1) and programmed cell death protein ligand-2 (PD-L2), in patients with Epstein–Barr virus-associated gastric cancer (EBVaGC).
Methods:
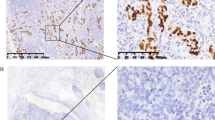
After reviewing 1318 consecutive cases of surgically resected or endoscopic submucosal dissected gastric cancers, the expression status of PD-L1 and PD-L2 in 120 patients with EBVaGC identified by EBV-encoded RNA in situ hybridisation was retrospectively analysed using immunohistochemistry (IHC). For each IHC marker, positivity was separately in intraepithelial tumour cells (iTu-) and immune cells in the tumour stroma area (str-).
Results:
Among 116 eligible patients, 57 (49.1%) and 66 patients (56.9%) were determined as iTu-PD-L1-positive and str-PD-L1-positive, respectively, whereas 23 (21.6%) and 45 patients (38.8%) were determined as iTu-PD-L2 positive and str-PD-L2 positive, respectively. Intraepithelial tumour cell PD-L1 positivity was found to be significantly associated with lymph node (LN) metastasis (P=0.012) and a poor disease-free survival (DFS) (P=0.032), yet not overall survival (P=0.482). In a multivariate analysis, iTu-PD-L1 positivity was independently associated with a poor DFS (P=0.006, hazard ratio=12.085). In contrast, str-PD-L2-positivity was related to a lower T category (P=0.003), absence of LN metastasis (P=0.032) and perineural invasion (P=0.028). Intraepithelial tumour cell and str-PD-L2 positivity showed a trend towards an improved DFS, although not significant (P=0.060 and P=0.073, respectively).
Conclusions:
Intraepithelial tumour cells PD-L1 expression can be used to predict a poor outcome in patients with EBVaGC and can represent a rational approach for PD-1/PD-L pathway-targeted immunotherapy.
Similar content being viewed by others
Main
Immunotherapy has begun to revolutionise cancer treatment, by introducing therapies such as checkpoint inhibitors that target the host immune system instead of the tumour (Chen and Mellman, 2013). For an anticancer immune response, the programmed cell death protein-1 (PD-1) pathway is considered an important inhibitory mechanism regulating T-cell exhaustion (** and Yoon, 2016). PD-1, which belongs to the CD28 family of proteins, is a receptor expressed on a number of immune cells, including T cells, B cells, monocytes, NK cells, and dendritic cells (Park et al, 2016). PD-1 has two ligands, the programmed cell death protein ligand-1 (PD-L1) and programmed cell death protein-ligand-2 (PD-L2). Programmed cell death protein ligand-1 is expressed on T cells, B cells, dendritic cells, macrophages, mesenchymal stem cells, bone marrow-derived mast cells, and some non-haematopoietic cells, whereas PD-L2 is mainly expressed on antigen-presenting cells, including macrophages, dendritic cells, and non-haematopoietic tissues (** and Yoon, 2016). Several studies have already demonstrated that PD-L1 or PD-1 is highly expressed on tumour cells in gastric cancers (GCs) (Wu et al, 2006; Takaya et al, 2015; Zhang et al, 2015; Böger et al, 2016; Derks et al, 2016; Dong et al, 2016a; Eto et al, 2016; Kim et al, 2016; Takano et al, 2016; Cho et al, 2017). Interestingly, Wang et al (2016) suggested that GC patients with positive PD-L1 expression had a significantly shorter survival than PD-L1-negative patients, although the prognostic impact remained inconsistent.
The Cancer Genome Atlas data revealed that Epstein–Barr virus (EBV)-associated GC (EBVaGC) closely associated with a PIK3CA mutation, PD-L1/2 overexpression, EBV-CIMP, CDKN2A silencing, and immune cell signalling activation among GC subtypes. As previous studies have suggested that high PD-L1 expression is related to a good responsiveness to anti-PD-1/PD-L1 therapy in human cancers, the EBVaGC subtype, in particular, may be a potential candidate for anti-PD-1/PD-L1 therapy among GC subtypes (Wang et al, 2003; ** and Yoon, 2016). In the present study, str-PD-L2 expression was positively correlated with dense iTu- and/or str-TILs, yet not iTu-PD-L2 expression. Ohigashi et al. suggested that PD-L2 expression inversely correlated with tumour-infiltrating CD8+ T cells in 41 patients with oesophageal cancer. Thus, whether PD-L1 and PD-L2 are influenced by specific markers of TILs, such as CD4, CD8, and FOXP3, and interferon-γ would be an interesting focus for further studies of the tumour immunity of EBVaGC. Meanwhile, for the correlation between PD-L1 and PD-L2 expression, PD-L1 was found to be expressed independently of PD-L2 expression. This finding supports previous report, where iTu-PD-L2 expression was induced in the absence of PD-L1 co-expression tumours, in which case PD-L2 expression was less likely to possess PD-L1+ immune cells (Derks et al, 2015). These observations can also help to understand the exact role of PD-L2 on the natural outcomes of EBVaGC.
Tumour-infiltrating lymphocytes could be used to identify a subgroup of patients with excellent outcomes (Chang et al, 2014) and our previous study also showed an independent association between a high density of TILs and a favourable recurrence-free survival or DFS in 120 patients with EBVaGC (Kang et al, 2016). This finding supports that TILs exhibit a host cellular immune response against tumours, indicating that immunotherapy may have a potential role in patients with EBVaGC (Kang et al, 2015). Several studies have already demonstrated a correlation between TILs and PD-L1 expression in EBVaGC (Kawazoe et al, 2016; Ma et al, 2016; Saito et al, 2016). Interestingly, when combining the iTu-PD-L1 expression and TIL score, the patient group with iTu-PD-L1 negativity and str-TIL positivity was associated with a better DFS than the other EBVaGC patients. This result creates a very meaningful subgroup that can provide more information for tailored therapy for each group of patients. In particular, this finding can provide a novel strategy for subgroups with decreased immunoediting via the PD-1 pathway and a strengthened immune response by TILs, and vice versa, considering that the state of the immune equilibrium is a critical factor for therapeutic success (Chen and Mellman, 2013; Palucka and Coussens, 2016). In fact, alterations of TILs have already been implicated in immune homeostasis, as well as immune response in the PD-1 pathway (Menon et al, 2016).
Although the present data identified a significant prognostic role of PD-L1 expression in operable EBVaGC, these results should be cautiously interpreted due to potential limitations. The current study is a retrospective evaluation, and the evaluation and interpretation of PD-L1 and PD-L2 within tumours and/or a tumour microenvironment have not yet been standardised in GCs (Manson et al, 2016). Moreover, a standardised cutoff value for PD-L1- and PD-L2 positivity has not yet been clearly established (Festino et al, 2016), plus PD-L1 and PD-L2 expression cannot be used to predict treatment response to anti-PD-1/PD-L1 therapy in patients with EBVaGC. Nevertheless, the present study has several important differences with previous studies as follows: (1) a relatively large-scale cohort of EBVaGC patients with a Korean homogeneous ethnic identity (** and Yoon, 2016, 2) equivalent treatment application, (3) minimal loss to follow-up, (4) assessment of TILs, and (5) comprehensive interpretation of expression in cancer cells and immune cells as separate microenvironments of cancer cells.
Conclusion
In conclusion, str-PD-L1- and str-PD-L2 positivity was shown to be associated with dense iTu- and str-TILs in patients with EBVaGC. The current study also revealed significant prognostic impact of iTu-PD-L1 positivity by demonstrating an independent association with a poor DFS. Therefore, these findings support the concept that PD-L1 can be a prognostic indicator for predicting patient outcomes and a rationale for therapeutic targeting in EBVaGC.
Change history
05 December 2017
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Böger C, Behrens H-M, Mathiak M, Krüger S, Kalthoff H, Röcken C (2016) PD-L1 is an independent prognostic predictor in gastric cancer of Western patients. Oncotarget 7 (17): 24269–24283.
Chang W-J, Du Y, Zhao X, Ma L-Y, Cao G-W (2014) Inflammation-related factors predicting prognosis of gastric cancer. World J Gastroenterol 20 (16): 4586–4596.
Chen DS, Mellman I (2013) Oncology meets immunology: the cancer-immunity cycle. Immunity 39 (1): 1–10.
Cho J, Lee J, Bang H, Tae Kim S, Hoon Park S, Yeong An J, Gew Choi M, Ho Lee J, Sung Sohn T, Moon Bae J, Ki Kang W, Kim S, Kim K-M (2017) Programmed cell death-ligand 1 expression predicts survival in patients with gastric carcinoma with microsatellite instability. Oncotarget 8 (8): 13320–13328.
Derks S, Liao X, Chiaravalli AM, Xu X, Camargo MC, Solcia E, Sessa F, Fleitas T, Freeman GJ, Rodig SJ, Rabkin CS, Bass AJ (2016) Abundant PD-L1 expression in Epstein-Barr Virus-infected gastric cancers. Oncotarget 7 (22): 32925–32932.
Derks S, Nason KS, Liao X, Stachler MD, Liu KX, Liu JB, Sicinska E, Goldberg MS, Freeman GJ, Rodig SJ, Davison JM, Bass AJ (2015) Epithelial PD-L2 expression marks Barrett’s esophagus and oesophageal adenocarcinoma. Cancer Immunol Res 3 (10): 1123–1129.
Dieci MV, Criscitiello C, Goubar A, Viale G, Conte P, Guarneri V, Ficarra G, Mathieu MC, Delaloge S, Curigliano G, Andre F (2014) Prognostic value of tumour-infiltrating lymphocytes on residual disease after primary chemotherapy for triple-negative breast cancer: a retrospective multicenter study. Ann Oncol 25 (3): 611–618.
Dong M, Wang H-Y, Zhao X-X, Chen J-N, Zhang Y-W, Huang Y, Xue L, H-G Li, Du H, Wu X-Y, Shao C-K (2016a) Expression and prognostic roles of PIK3CA, JAK2, PD-L1, and PD-L2 in Epstein-Barr virus–associated gastric carcinoma. Hum Pathol 53: 25–34.
Dong Y, Sun Q, Zhang X (2016b) PD-1 and its ligands are important immune checkpoints in cancer. Oncotarget 8 (2): 2171–2186.
Edge SB, Compton CC (2010) The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol 17 (6): 1471–1474.
Eto S, Yoshikawa K, Nishi M, Higashijima J, Tokunaga T, Nakao T, Kashihara H, Takasu C, Iwata T, Shimada M (2016) Programmed cell death protein 1 expression is an independent prognostic factor in gastric cancer after curative resection. Gastric Cancer 19 (2): 466–471.
Fang W, Zhang J, Hong S, Zhan J, Chen N, Qin T, Tang Y, Zhang Y, Kang S, Zhou T, Wu X, Liang W, Hu Z, Ma Y, Zhao Y, Tian Y, Yang Y, Xue C, Yan Y, Hou X, Huang P, Huang Y, Zhao H, Zhang L (2014) EBV-driven LMP1 and IFN-γ up-regulate PD-L1 in nasopharyngeal carcinoma: implications for oncotargeted therapy. Oncotarget 5 (23): 12189–12202.
Festino L, Botti G, Lorigan P, Masucci GV, Hipp JD, Horak CE, Melero I, Ascierto PA (2016) Cancer treatment with anti-PD-1/PD-L1 agents: is PD-L1 expression a biomarker for patient selection? Drugs 76 (9): 925–945.
Herbst RS, Soria JC, Kowanetz M, Fine GD, Hamid O, Gordon MS, Sosman JA, McDermott DF, Powderly JD, Gettinger SN, Kohrt HE, Horn L, Lawrence DP, Rost S, Leabman M, **ao Y, Mokatrin A, Koeppen H, Hegde PS, Mellman I, Chen DS, Hodi FS (2014) Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 515 (7528): 563–567.
** Z, Yoon HH (2016) The promise of PD-1 inhibitors in gastro-oesophageal cancers: microsatellite instability vs. PD-L1. J Gastrointest Oncol 7 (5): 771–788.
Kang BW, Seo AN, Yoon S, Bae HI, Jeon SW, Kwon OK, Chung HY, Yu W, Kang H, Kim JG (2015) Prognostic value of tumour-infiltrating lymphocytes in Epstein–Barr virus-associated gastric cancer. Ann Oncol 27 (3): 494–501.
Kang BW, Seo AN, Yoon S, Bae HI, Jeon SW, Kwon OK, Chung HY, Yu W, Kang H, Kim JG (2016) Prognostic value of tumour-infiltrating lymphocytes in Epstein-Barr virus-associated gastric cancer. Ann Oncol 27 (3): 494–501.
Kawazoe A, Kuwata T, Kuboki Y, Shitara K, Nagatsuma AK, Aizawa M, Yoshino T, Doi T, Ohtsu A, Ochiai A (2016) Clinicopathological features of programmed death ligand 1 expression with tumour-infiltrating lymphocyte, mismatch repair, and Epstein–Barr virus status in a large cohort of gastric cancer patients. Gastric Cancer 20 (3): 407–415.
Kim JW, Nam KH, Ahn S-H, Park DJ, Kim H-H, Kim SH, Chang H, Lee J-O, Kim YJ, Lee HS, Kim JH, Bang S-M, Lee JS, Lee K-W (2016) Prognostic implications of immunosuppressive protein expression in tumours as well as immune cell infiltration within the tumour microenvironment in gastric cancer. Gastric Cancer 19 (1): 42–52.
Koh J, Ock C-Y, Kim JW, Nam SK, Kwak Y, Yun S, Ahn S-H, Park DJ, Kim H-H, Kim WH, Lee HS (2017) Clinicopathologic implications of immune classification by PD-L1 expression and CD8-positive tumour-infiltrating lymphocytes in stage II and III gastric cancer patients. Oncotarget 8 (16): 26356–26367.
Latchman Y, Wood CR, Chernova T, Chaudhary D, Borde M, Chernova I, Iwai Y, Long AJ, Brown JA, Nunes R, Greenfield EA, Bourque K, Boussiotis VA, Carter LL, Carreno BM, Malenkovich N, Nishimura H, Okazaki T, Honjo T, Sharpe AH, Freeman GJ (2001) PD-L2 is a second ligand for PD-1 and inhibits T cell activation. Nat Immunol 2 (3): 261–268.
Loke Pn, Allison JP (2003) PD-L1 and PD-L2 are differentially regulated by Th1 and Th2 cells. Proc Natl Acad Sci USA 100 (9): 5336–5341.
Ma C, Patel K, Singhi AD, Ren B, Zhu B, Shaikh F, Sun W (2016) Programmed death-ligand 1 expression is common in gastric cancer associated with Epstein-Barr virus or microsatellite instability. Am J Surg Pathol 40 (11): 1496–1506.
Manson G, Norwood J, Marabelle A, Kohrt H, Houot R (2016) Biomarkers associated with checkpoint inhibitors. Ann Oncol 27 (7): 1199–1206.
McShane LM, Altman DG, Sauerbrei W, Taube SE, Gion M, Clark GM (2005) REporting recommendations for tumour MARKer prognostic studies (REMARK). Nat Clin Pract Oncol 2 (8): 416–422.
Menon S, Shin S, Dy G (2016) Advances in cancer immunotherapy in solid tumours. Cancers 8 (12): 106.
Ohigashi Y, Sho M, Yamada Y, Tsurui Y, Hamada K, Ikeda N, Mizuno T, Yoriki R, Kashizuka H, Yane K, Tsushima F, Otsuki N, Yagita H, Azuma M, Nakajima Y (2005) Clinical significance of programmed death-1 ligand-1 and programmed death-1 ligand-2 expression in human oesophageal cancer. Clin Cancer Res 11 (8): 2947–2953.
Palucka AK, Coussens LM (2016) The basis of oncoimmunology. Cell 164 (6): 1233–1247.
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12 (4): 252–264.
Park J, Kwon M, Shin E-C (2016) Immune checkpoint inhibitors for cancer treatment. Arch Pharm Res 39 (11): 1577–1587.
Saito R, Abe H, Kunita A, Yamashita H, Seto Y, Fukayama M (2016) Overexpression and gene amplification of PD-L1 in cancer cells and PD-L1+ immune cells in Epstein-Barr virus-associated gastric cancer: the prognostic implications. Mod Pathol 30 (3): 427–439.
Takano S, Saito H, Ikeguchi M (2016) An increased number of PD-1+ and Tim-3+ CD8+ T cells is involved in immune evasion in gastric cancer. Surg Today 46 (11): 1341–1347.
Takaya S, Saito H, Ikeguchi M (2015) Upregulation of immune checkpoint molecules, PD-1 and LAG-3, on CD4+ and CD8+ T cells after gastric cancer surgery. Yonago Acta Med 58 (1): 39–44.
Taube JM, Klein A, Brahmer JR, Xu H, Pan X, Kim JH, Chen L, Pardoll DM, Topalian SL, Anders RA (2014) Association of PD-1, PD-1 ligands, and other features of the tumour immune microenvironment with response to anti-PD-1 therapy. Clin Cancer Res 20 (19): 5064–5074.
Teng MW, Ngiow SF, Ribas A, Smyth MJ (2015) Classifying cancers based on T-cell infiltration and PD-L1. Cancer Res 75 (11): 2139–2145.
Wang S, Bajorath J, Flies DB, Dong H, Honjo T, Chen L (2003) Molecular modeling and functional map** of B7-H1 and B7-DC uncouple costimulatory function from PD-1 interaction. J Exp Med 197 (9): 1083–1091.
Wang X, Teng F, Kong L, Yu J (2016) PD-L1 expression in human cancers and its association with clinical outcomes. Oncotargets Ther 9: 5023–5039.
Wu C, Zhu Y, Jiang J, Zhao J, Zhang X-G, Xu N (2006) Immunohistochemical localization of programmed death-1 ligand-1 (PD-L1) in gastric carcinoma and its clinical significance. Acta Histochem 108 (1): 19–24.
Zhang L, Qiu M, ** Y, Ji J, Li B, Wang X, Yan S, Xu R, Yang D (2015) Programmed cell death ligand 1 (PD-L1) expression on gastric cancer and its relationship with clinicopathologic factors. Int J Clin Exp Pathol 8 (9): 11084–11091.
Zhang M, Dong Y, Liu H, Wang Y, Zhao S, Xuan Q, Wang Y, Zhang Q (2016) The clinicopathological and prognostic significance of PD-L1 expression in gastric cancer: a meta-analysis of 10 studies with 1,901 patients. Sci Rep 6: 37933.
Zhang Y, Chung Y, Bishop C, Daugherty B, Chute H, Holst P, Kurahara C, Lott F, Sun N, Welcher AA, Dong C (2006) Regulation of T cell activation and tolerance by PDL2. Proc Natl Acad Sci USA 103 (31): 11695–11700.
Acknowledgements
The biospecimens for this study were provided by National Biobank of Korea-Kyungpook National University Hospital (KNUH), which is supported by the Ministry of Health, Welfare, and Affairs. All materials derived from the National Biobank of Korea-KNUH were obtained (with informed consent) under institutional review board (IRB)-approved protocols. This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (2014R1A5A2009242) and Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2017R1D1A3B03032320).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License.
Supplementary Information accompanies this paper on British Journal of Cancer website
Supplementary information
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Seo, A., Kang, B., Kwon, O. et al. Intratumoural PD-L1 expression is associated with worse survival of patients with Epstein–Barr virus-associated gastric cancer. Br J Cancer 117, 1753–1760 (2017). https://doi.org/10.1038/bjc.2017.369
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2017.369
- Springer Nature Limited
Keywords
This article is cited by
-
PD-L1 expression in EBV associated gastric cancer: a systematic review and meta-analysis
Discover Oncology (2022)
-
PD-L1 as a biomarker of response to immune-checkpoint inhibitors
Nature Reviews Clinical Oncology (2021)
-
Global updates in the treatment of gastric cancer: a systematic review. Part 1: staging, classification and surgical treatment
Updates in Surgery (2020)
-
PD-L1 expression and the prognostic significance in gastric cancer: a retrospective comparison of three PD-L1 antibody clones (SP142, 28–8 and E1L3N)
Diagnostic Pathology (2018)
-
Transcriptional analysis of immune genes in Epstein–Barr virus-associated gastric cancer and association with clinical outcomes
Gastric Cancer (2018)