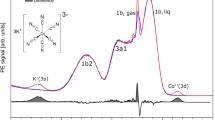
Abstract
INFRA-RED spectroscopy has shown that only a small proportion of the external OH groups of kaolinite contribute to the bonding mechanism between layers and also that these interlayer hydrogen bonds are very weak (absorption bands above 3,600 cm−1) (ref. 1).
Article PDF
Similar content being viewed by others
References
Serratosa, J. M., Hidalgo, A., and Viñas, J. M., Nature, 195, 486 (1962).
Serratosa, J. M., and Bradley, W. F., J. Phys. Chem., 62, 1164 (1958).
Lippincott, E. R., and Schroeder, R., J. Chem. Phys., 23, 1099 (1955).
Steinfink, H., Acta Cryst., 11, 191 (1958).
Brown, B. E., and Bailey, S. W., Amer. Mineral., 48, 42 (1963).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
SERRATOSA, J., VIÑAS, J. Infra-red Investigation of the OH Bonds in Chlorites. Nature 202, 999 (1964). https://doi.org/10.1038/202999a0
Issue Date:
DOI: https://doi.org/10.1038/202999a0
- Springer Nature Limited