Abstract
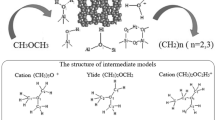
The alkylation reaction of aniline with methanol on zeolites HY and CsOH/CsNaY was studied by in situ 13C NMR spectroscopy under flow and batch conditions. Attention was focused on the identification of intermediates and on the determination of the formation mechanisms of N-methylaniline, N,N-dimethylaniline, and toluidines. To refine the main steps of the reaction, the transformations of the following individual compounds and intermediates, which were detected in the course of alkylation, were studied: dimethyl ether, surface methoxy groups, methylanilinium ions, formaldehyde, and N-methyleneaniline. It was found that N-methylaniline and N,N-dimethylaniline were formed as a result of aniline methylation by methanol dehydration products (methoxy groups or dimethyl ether) on acidic zeolites or as a result of alkylation by formaldehyde or methoxy groups on basic zeolites. Toluidines were formed by the isomerization ofN-methylanilinium ions, which were produced only on acidic zeolites, rather than by the direct alkylation of aniline.
Similar content being viewed by others
REFERENCES
Narayanan, S. and Deshpande, K., Appl. Catal., A, 2000, vol. 199, p. 1.
Narayanan, S., Durga Kumari, V., and Sudhakar Rao, A., Appl. Catal., A, 1994, vol. 111, p. 133.
Park, Y.K., Park, K.Y., and Woo, S.I., Catal. Lett., 1994, vol. 26, p. 169.
Hari Prasad Rao, P.R., Massiani, P., and Barthomeuf, D., Catal. Lett., 1995, vol. 31, p. 115.
Klinowski, J., Thomas, J.M., Audier, M., Vasudevan, S., Fyfe, C.A., and Hartman, J.S., J. Chem. Soc., Chem. Commun., 1981, p. 570.
Anderson, M.V. and Klinowski, J., Nature, 1989, vol. 339, p. 200.
Haw, J.F., NMR Techiques in Catalysis, New York: Marcel Dekker, 1994, p. 139.
Hunger, M. and Horvath, T., J. Chem. Soc., Chem. Commun., 1995, p. 1423.
Ivanova, I.I., Ross. Khim. Zh., 1998, vol. 42, p. 67.
Stepanov, A.G., Usp. Khim., 1999, vol. 68, no.7, p. 619.
Hunger, M., Schenk, U., and Weitkamp, J., Angew. Chem., Int. Ed. Engl., 1997, vol. 63, p. 2504.
Hunger, M., Schenk, U., Seiler, M., and Weitkamp, J., J. Mol. Catal. A: Chem., 2000, vol. 156, p. 153.
Derouane, E.G., Abdul-Hamid, S.B., Ivanova, I.I., Blom, N., and Højlund-Nielsen, P.E., J. Mol. Catal., 1994, vol. 86, p. 371.
Ivanova, I.I., Pomakhina, E.B., Rebrov, A.I., Hunger, M., Kolyagin, Y.G., and Weitkamp, J., J. Catal., 2001, vol. 203, p. 375.
Hunger, M., Seiler, M., and Horvath, T., Catal. Lett., 1999, vol. 57, p. 199.
Ivanova, I.I. and Corma, A., J. Phys. Chem., 1997, vol. 101, p. 547.
Breitmaier, E. and Voelter, W., Carbon-13 NMR Spec-troscopy, Weinheim: VCH, 1987, p. 257.
Wang, W., Seiler, M., Ivanova, I.I., Sternberg, U., Weitkamp, J., and Hunger, M., J. Am. Chem. Soc., 2002, vol. 124, p. 7548.
Wang, W., Seiler, M., Ivanova, I.I., Weitkamp, J., and Hunger, M., J. Chem. Soc., Chem. Cummun., 2001, p. 1362.
Sidorenko, Yu.N. and Galich, P.N., Ukr. Khim. Zh., 1970, vol. 36, p. 1234.
Itoh, H., Miayamoto, A., and Murakami, Y., J. Catal., 1980, vol. 64, p. 284.
Philippou, A. and Anderson, M.W., J. Am. Chem. Soc., 1994, vol. 116, p. 5774.
Hunger, M., Schenk, U., and Weitkamp, J., J. Mol. Catal., A: Chem., 1998, p. 97.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ivanova, I.I., Pomakhina, E.B., Rebrov, A.I. et al. Mechanism of Aniline Methylation on Zeolite Catalysts Investigated by In Situ 13C NMR Spectroscopy. Kinetics and Catalysis 44, 701–709 (2003). https://doi.org/10.1023/A:1026158525990
Issue Date:
DOI: https://doi.org/10.1023/A:1026158525990