Abstract
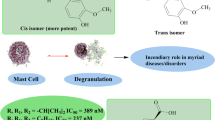
Purpose. Mast cell and neutrophil degradations are the important players in inflammatory disorders. Combined with potent inhibition of chemical mediators released from mast cells and neutrophil degranulations, it could be a promising anti-inflammatory agent. 2′,5′-Dihydroxychalcone has been reported as a potent chemical mediator and cyclooxygenase inhibitor. In an effort to continually develop potent anti-inflammatory agents, a novel series of chalcone, 2′- and 3′-hydroxychalcones, 2′,5′-dihydroxychalcones and flavanones were continually synthesized to evaluate their inhibitory effects on the activation of mast cells and neutrophils and the inhibitory effect on phlogist-induced hind-paw edema in mice.
Methods. A series of chalcones and related compounds were prepared by Claisen-Schmidt condensation of appropriate acetophenones with appropriate aromatic aldehyde and the anti-inflammatory activities of these synthetic compounds were studied on inhibitory effects on the activation of mast cells and neutrophils.
Results. Some chalcones showed strong inhibitory effects on the release of β-glucuronidase and histamine from rat peritoneal mast cells stimulated with compound 48/80. Almost all chalcones and 4′-hydroxyflavanone exhibited potent inhibitory effects on the release of β-glucuronidase and lysozyme from rat neutrophils stimulated with formyl-Met-Leu-Phe (fMLP). Some chalcones showed potent inhibitory effects on superoxide formation of rat neutrophils stimulated with fMLP/cytochalasin B (CB) or phorbol myristate acetate (PMA). 2′,3-Dihydroxy-, 2′,5′-dihydroxy-4-chloro-, and 2′,5′-dihydroxychalcone showed remarkable inhibitory effects on hind-paw edema induced by polymyxin B in normal as well as in adrenalectomized mice.
Conclusions. These results indicated that the anti-inflammatory effects of these compounds were mediated, at least partly, through the suppression of chemical mediators released from mast cells and neutrophils.
Similar content being viewed by others
REFERENCES
Jr. Elliott Middleton, and C. Kandaswami. Biochem. Pharmacol. 43:1167–1179 (1992).
B. M. Babior. New Engl. J. Med. 298:659–668 (1978).
A. I. Tauber, and B. M. Babior. Free Radic. Biol. Med. 1:265–307 (1985).
S. Sogawa, Y. Nihro, H. Ueda, A. Izumi, T. Miki, H. Matsumoto, and T. Satoh. J. Med. Chem. 36:3904–3909 (1993).
C. N. Lin, T. H. Lee, M. F. Hsu, J. P. Wang, F. N. Ko, and C. M. Teng. J. Pharm. Pharmacol., 49:530–536 (1997).
J. P. Wang, S. L. Raung, C. N. Lin, and C. M. Teng. Eur. J. Pharmacol. 251:35–42 (1994).
A. Boyum. Scand. J. Clin. Invest. 97(Suppl.):77–89 (1968).
R. J. Smith and S. S. Iden. Biochem. Biophys. Res. Commun. 91:263–271 (1979).
M. Markert, P. C. Andrews, and B. M. Babior. Methods Enzymol. 105:358–365 (1984).
J. P. Wang, M. F. Hsu, S. L. Raung, C. C. Chen, J. S. Kuo, and C. M Teng. Naunyn Schmiedebergs Arch. Pharmacol. 346:707–712 (1992).
H. B. Waynforth. Academic Press, London, 1980, pp. 124–208.
L. C. Mcphail, and R. Snyderman. J. Clin. Invest. 72:192–200 (1983).
F. Morel, J. Doussiere, and P. V. Vignais. Eur. J. Biochem. 210:523–546 (1991).
Bertelli, and G. Soldani. Arzneim. Forsch. 29:777–778 (1979).
C. N. Lin, M. I. Chung, S. Y. Liou, T. H. Lee, and J. P. Wang. J. Pharm. Pharmacol. 48:532–538 (1996).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hsieh, HK., Lee, TH., Wang, JP. et al. Synthesis and Anti-inflammatory Effect of Chalcones and Related Compounds. Pharm Res 15, 39–46 (1998). https://doi.org/10.1023/A:1011940401754
Issue Date:
DOI: https://doi.org/10.1023/A:1011940401754