Abstract
Background
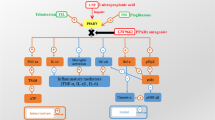
Peroxisome proliferator-activated receptor γ (PPAR-γ) agonists have received much attention in research because of their neuroprotective and anti-inflammatory effects that reduce cell death and halt the progression of neurodegeneration. Thus, this study observed the pioglitazone effects on the main inflammatory markers after 6-hydroxydopamine (6-OHDA) lesion.
Methods
The effects of a 5-day administration of the PPAR-γ agonist pioglitazone (30 mg/kg) in male Wistar rats that received bilateral intranigral infusions of 6-OHDA. After surgery, the rats were evaluated in the open-field test on days 1,7,14, and 21. Immediately after the behavioral tests on day 21, the rats were euthanized, and the substantia nigra was removed to analyze the expression of nuclear factor κB (NF-κB) and IκB by western blot. To immunohistochemical, animals were intracardially perfused, with brain removal that was frozen and sectioned, being selected slices of the SNc region to detect tyrosine hydroxylase (TH) immunoreactivity, microglia activation (Iba-1) and NF-κB translocation in the nucleus.
Results
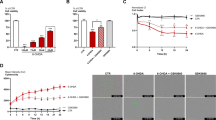
Pioglitazone protected rats against hypolocomotion and 6-OHDA-induced dopaminergic neurodegeneration on day 7. Decreases in the microglial activation and the NF-κB expression were observed, and the p65 activation was inhibited.
Conclusions
These results suggest that pioglitazone may be a potential adjuvant for the treatment of Parkinson`s disease because of its effects on pathological markers of the progression of neurodegeneration.
Similar content being viewed by others
References
Schapira AH. Neurobiology and treatment of Parkinson’s disease. Trends Pharmacol Sci 2009;30:41–7.
Connolly BS, Lang AE. Pharmacological treatment of Parkinson’s disease? A review. JAMA 2014;311:1670–83.
Dauer W, Przedborski S. Parkinson’s disease: mechanisms and models. Neuron 2003;39:889–09.
Kalia LV, Lang AE, Shulman G. Parkinson’s disease. Lancet 2015;386:896–12.
Salat D, Noyce AJ, Schrag A, Tolosa E. Challenges of modifying disease progression in prediagnostic Parkinson’s disease. Lancet Neurol 2016;15:637–48.
Gao HM, Jiang J, Wilson B, Zhang W, Hong JS, Liu B. Microglial activation-mediated delayed and progressive degeneration of rat nigral dopaminergic neurons: relevance to Parkinson’s disease. J Neurochem 2002;81:1285–97.
Hirsch EC, Vyas S, Hunot S. Neuroinflammation in Parkinson’s disease. Parkinsonism Relat Disord 2012;18(Suppl. 1):S210–2.
Hong H, Kim BS, Im HI. Pathophysiological role of neuroinflammation in neurodegenerative diseases and psychiatric disorders. Int Neurourol J 2016;20(Suppl. 1):S2–7.
Liu Y, Nakahara T, Miyakoshi J, Hu DL, Nakane A, Abe Y. Nuclear accumulation and activation of nuclear factor κB after split-dose irradiation in LS174T cells. J Radiat Res 2007;48:13–20.
Hirsch EC, Jenner P, Przedborski S. Pathogenesis of Parkinson’s disease. Mov Disord 2013;28:24–30.
Blandini F. Neural and immune mechanisms in the pathogenesis of Parkinson’s disease. J Neuroimmune Pharmacol 2013;8:189–201.
Zhang J, Zhang Y, **ao F, Liu Y, Wang J, Gao H, et al. The peroxisome proliferator-activated receptor γ agonist pioglitazone prevents NF-κB activation in cisplatin nephrotoxicity through the reduction of p65 acetylation via the AMPK-SIRT1/p300 pathway. Biochem Pharmacol 2016;101:100–11.
De Filippis B, Linciano P, Ammazzalorso A, Di Giovanni C, Fantacuzzi M, Giampietro L, et al. Structural development studies of PPARs ligands based on tyrosine scaffold. Eur J Med Chem 2015;89:817–25.
Liu M, Bachstetter AD, Cass WA, Lifshitz J, Bing G. Pioglitazone attenuates neuroinflammation and promotes dopaminergic neuronal survival in the nigrostriatal system of rats after diffuse brain injury. J Neurotrauma 2017;34(2):414–22.
O’Sullivan SE. An update on PPAR activation by cannabinoids. Br J Pharmacol 2016;173:1899–910.
Dehmer T, Heneka MT, Sastre M, Dichgans J, Schulz JB. Protection by pioglitazone in the MPTP model of Parkinson’s disease correlates with IκBα induction and block of NF-κB and iNOS activation. J Neurochem 2004;88(2):494–501.
Sadaghiani MS, Javadi-Paydar M, Gharedaghi MH, Fard YY, Dehpour AR. Antidepressant-like effect of pioglitazone in the forced swimming test in mice: the role of PPAR-gamma receptor and nitric oxide pathway. Behav Brain Res 2011;224:336–43.
Barbiero JK, Santiago RM, Lima MMS, Ariza D, Morais LH, Andreatini R, et al. Acute but not chronic administration of pioglitazone promoted behavioral and neurochemical protective effects in the MPTP model of Parkinson’s disease. Behav Brain Res 2011;216:186–92.
Paxinos G, Watson C. The rat brain in stereotaxic coordinates — the new coronal set. 5th ed San Diego: Academic Press; 2005.
Gomes FV, Llorente R, Del Bel EA, Viveros MP, López-Gallardo M, Guimarães FS. Decreased glial reactivity could be involved in the antipsychotic-like effect of cannabidiol. Schizophr Res 2015;164:155–63.
Laloux C, Petrault M, Lecointe C, Devos D, Bordet R. Differential susceptibility to the PPAR-γ agonist pioglitazone in 1-methyl-4phenyl-1,2,3,6-tetrahydropyridine and 6-hydroxydopamine rodent models of Parkinson’s disease. Pharmacol Res 2012;65:514–22.
Sadeghian M, Marinova-Mutafchieva L, Broom L, Davis JB, Virley D, Medhurst AD, et al. Full and partial peroxisome proliferation-activated receptor-γagonists, but not d agonist, rescue of dopaminergic neurons in the 6-OHDA parkinsonian model is associated with inhibition of microglial activation and MMP expression. J Neuroimmunol 2012;246:69–77.
Lima MMS, Reksidler AB, Zanata SM, Machado HB, Tufik S, MABF Vital. Different parkinsonism models produce a time-dependent induction of COX-2 in the substantia nigra of rats. Brain Res 2006;1101:117–25.
Gradowski RW, Santiago RM, Zaminelli T, Bassani TB, Barbiero JK, Boschen SL, et al. Antidepressant-like effect of curcumin in 6-hydroxydopamine model of Parkinson’s disease. Curr Trends Neurol 2013;7:69–80.
Santiago RM, Barbieiro J, Lima MMS, Dombrowski PA, Andreatini R, MABF Vital. Depressive-like behaviors alterations induced by intranigral MPTP, 6-OHDA, LPS and rotenone models of Parkinson’s disease are predominantly associated with serotonin and dopamine. Prog Neuropsychopharmacol Biol Psychiatry 2010;34:1104–14.
Kuter K, Kolasiewicz W, Gołembiowska K, Dziubina A, Schulze G, Berghauzen K, et al. Partial lesion of the dopaminergic innervation of the ventral striatum induces “depressive-like” behavior of rats. Pharmacol Rep 2011;63:1383–92.
Blesa J, Phani S, Jackson-Lewis V, Przedborski S. Classic and new animal models of Parkinson’s disease. J Biomed Biotechnol 2012;2012(84618).
Barbiero JK, Santiago RM, Persike DS, da Silva Fernandes MJ, Tonin FS, da Cunha C, et al. Neuroprotective effects of peroxisome proliferator-activated receptor alpha and gamma agonists in model of parkinsonism induced by intranigral 1-methyl-4-phenyl-1,2,3,6-tetrahyropyridine. Behav Brain Res 2014;274:390–9.
Hunot S, Brugg B, Ricard D, Michel PP, Muriel MP, Ruberg M, et al. Nuclear translocation of NF-κB is increased in dopaminergic neurons of patients with Parkinson’s disease. Proc Natl Acad Sci USA 1997;94:7531–6.
Machado A, Herrera AJ, Venero JL, Santiago M, De Pablos RM, Villarán RF, et al. Peripheral inflammation increases the damage in animal modelsofnigrostriatal dopaminergic neurodegeneration: possible implication in Parkinson’s disease incidence. Parkinsons Dis 2011;2011:393769.
He Y, Appel S, Le W. Minocycline inhibits microglial activation and protects nigral cells after 6-hydroxydopamine injection into mouse striatum. Brain Res 2001;909:187–93.
Barcia C, Sánchez-Bahillo A, Fernández-Villalba E, Bautista V, Poza Y, Poza M, et al. Evidence of active microglia in substantia nigra pars compacta of parkinsonism monkeys 1 year after MPTP exposure. Glia 2014;46:402–9.
Breidert T, Callebert J, Heneka MT, Landreth G, Launay JM, Hirsch EC. Protective action of the peroxisome proliferator-activated receptor-γ agonist pioglitazone in a mouse model of Parkinson’s disease. J Neurochem 2002;82:615–24.
Carta AR, Pisanu A. Modulating microglia activity with PPAR-γ agonists: α promising therapy for Parkinson’s disease? Neurotox Res 2013;23:112–23.
Leonard B, Maes M. Mechanistic explanations how cell-mediated immune activation, inflammation and oxidative and nitrosative stress pathways and their sequels and concomitants play a role in the pathophysiology of unipolar depression. Neurosci Biobehav Rev 2012;36:764–85.
Levites Y, Youdim MBH, Maor G, Mandel S. Attenuation of 6-hydroxydopamine (6-OHDA)-induced nuclear factor-κB (NF-κB) activation and cell death by tea extracts in neuronal cultures. Biochem Pharmacol 2002;63:21–9.
Tarabin V, Schwaninger M. The role of NF-κB in 6-hydroxydopamine- and TNFα-induced apoptosis of PC12 cells. Naunyn Shmiedebergs Arch Pharmacol 2004;369:563–9.
Liang ZQ, Li YL, Zhao XL, Han R, Wang X, Wang Y, et al. NF-κB contributes to 6-hydroxydopamine-induced apoptosis of nigral dopaminergic neurons through p53. Brain Res 2007;1145:190–03.
Cao JP, Wang HJ, Yu JK, Liu HM, Gao DS. The involvement of NF-κB p65/p52 in the effects of GDNF on DA neurons in early PD rats. Brain Res Bull 2008;76:505–11.
Glezer I, Marcourakis T, Christina M, Avellar W, Gorenstein C, Scavone C. The role of the transcription factor NF-κB in the molecular mechanisms of action of psychoactive drugs. Rev Bras Psiquiatr 2000;22:26–30.
Gilmore T. Introduction to NF-κB: players, pathways, perspectives. Oncogene 2006;25:6680–4.
Li X, Wu G, Wu M, Chen W, Liu X. In vitro study of inhibitory millimeter wave treatment effects on the TNF-α-induced NF-κB signal transduction pathway. Int J Mol Med 2011;27:71–8.
Troib A, Azab AN. Effects of psychotropic drugs on nuclear factor kappa B. Eur Rev Med Pharmacol Sci 2015;19:1198–08.
Yin Y, Allen PD, Jia L, Kelsey SM, Newland AC. 8-Cl-adenosine mediated cytotoxicity and sensitization of T-lymphoblastic leukemia cells to TNFα-induced apoptosis is via inactivation of NF-κB. Leuk Res 2001;25:423–31.
Sauer S. Ligands for the nuclear peroxisome proliferator-activated receptor gamma. Trends Pharmacol Sci 2015;36:688–76541.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Machado, M.M.F., Bassani, T.B., Cóppola-Segovia, V. et al. PPAR-γ agonist pioglitazone reduces microglial proliferation and NF-κB activation in the substantia nigra in the 6-hydroxydopamine model of Parkinson’s disease. Pharmacol. Rep 71, 556–564 (2019). https://doi.org/10.1016/j.pharep.2018.11.005
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2018.11.005