Abstract
Background
Tetrabromobisphenol A (TBBPA) is a brominated flame retardant widely used in a variety of commercial and household products. TBBPA can become bioaccumulated in human body fluids, and also in different brain regions. The aim of the present study was to determine the viability and apoptosis of cultured mouse hippocampal neurons in vitro after exposure to TBBPA. Additionally, we examined the involvement of ROS generation in the effect of TBBPA.
Methods
Primary hippocampal neuron cultures were prepared from Swiss mouse embryos on day 17/18 of gestation. The cultures were treated with TBBPA at concentrations ranging from 1 nM to 100 μM for 30 min or 3, 6 or 24 h. To study apoptosis, the activity of caspase-3 was measured, and apoptotic body formation was evaluated. To investigate the cytotoxic effect of TBBPA, the level of lactate dehydrogenase (LDH) was measured in the culture medium.
Results
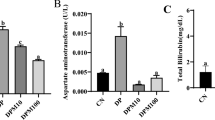
Our results demonstrated that TBBPA concentrations ranging from 100 nM to 100 μM caused caspase-3 activation and apoptotic body formation. The cytotoxic effects of TBBPA were observed at concentrations ranging from 50 nM to 100 μM. To detect intracellular ROS, the fluorogenic dye H2DCFDA was used. We did not observe any significant increase in the level of cellular ROS in cultured cells after TBBPA treatment. However, in a cell-free model, TBBPA at concentrations ranging from 10 to 100 μM interacted with H2DCFDA and enhanced the fluorescence signal.
Conclusion
We suggest that the H2DCFDA assay cannot be used to measure TBBPA-stimulated cell-mediated ROS production.
Similar content being viewed by others
References
De Wit CA, Herzke D, Vorkamp K. Brominated flame retardants in the Arctic environment — trends and new candidates. Sci Total Environ 2010;408:2885–918.
Sellström U, Jansson B. Analysis of tetrabrombisphenol A in a product and environmental samples. Chemosphere 1995;31:3085–92.
Hakk H, Letcher RJ. Metabolism in the toxicokinetics and fate of brominated flame retardants — a review. Environ Int 2003;29:801–28.
Thomsen C, Lundanes E, Becher G. Brominated flame retardants in archived serum samples from Norway: a study on temporal trends and the role of age. Environ Sci Technol 2002;36:1414–8.
Fujii Y, Nishimura E, Kato Y, Harada KH, Koizumi A, Haraguchi K. Dietary exposure to phenolic and methoxylated organohalogen contaminants in relation to their concentrations in breast milk and serum in Japan. Environ Int 2014;63:19–25.
Shi Z-X, Wu Y-N, Li J-G, Zhao Y-F, Feng J-F. Dietary exposure assessment of Chinese adults and nursing infants to tetrabromobisphenol-A and hexabromocyclododecanes: occurrence measurements in foods and human milk. Environ Sci Technol 2009;43:4314–9.
Thomsen C, Lundanes E, Becher G. Brominated flame retardants in plasma samples from three different occupational groups in Norway. J Environ Monit 2001;3:366–70.
Nakajima A, Saigusa D, Tetsu N, Yamakuni T, Tomioka Y, Hishinuma T. Neurobehavioral effects of tetrabromobisphenol A, a brominated flame retardant, in mice. Toxicol Lett 2009;189:78–83.
Viberg H, Eriksson P. Differences in neonatal neurotoxicity of brominated flame retardants, PBDE 99 and TBBPA, in mice. Toxicology 2011;289:59–65.
Wojtowicz AK, Szychowski KA, Kajta M. PPAR-γ agonist GW1929 but not antagonist GW9662 reduces TBBPA-induced neurotoxicity in primary neocortical cells. Neurotox Res 2014;25:311–22.
Reistad T, Mariussen E, Ring A, Fonnum F. In vitro toxicity of tetrabromobisphenol-A on cerebellar granule cells: cell death, free radical formation, calcium influx and extracellular glutamate. Toxicol Sci 2007;96:268–78.
Ziemińska E, Stafiej A, Toczyłowska B, Lazarewicz JW. Synergistic neurotoxicity of oxygen-glucose deprivation and tetrabromobisphenol A in vitro: role of oxidative stress. Pharmacol Rep 2012;64:1166–78.
Hendriks HS, Van Kleef RGDM, van den Berg M, Westerink RHS. Multiple novel modes of action involved in the in vitro neurotoxic effects of tetrabromobisphenol-A. Toxicol Sci 2012;128:235–46.
Halliwell B. Antioxidant defence mechanisms: from the beginning to the end (of the beginning). Free Radic Res 1999;31:261–72.
Le Bras M, Clément M-V, Pervaiz S, Brenner C. Reactive oxygen species and the mitochondrial signaling pathway of cell death. Histol Histopathol 2005;20:205–19.
Kajta M, Wójtowicz AK, Maćkowiak M, Lasoń W. Aryl hydrocarbon receptor-mediated apoptosis of neuronal cells: a possible interaction with estrogen receptor signaling. Neuroscience 2009;158:811–22.
Kajta M, Makarewicz D, Ziemińska E, Jantas D, Domin H, Lasoń W. Neuroprotection by co-treatment and post-treating with calcitriol following the ischemic and excitotoxic insult in vivo and in vitro. Neurochem Int 2009;55:265–74.
Brewer GJ. Serum-free B27/neurobasal medium supports differentiated growth of neurons from the striatum, substantia nigra, septum, cerebral cortex, cerebellum, and dentate gyrus. J Neurosci Res 1995;42:674–83.
Kajta M, Trotter A, Lasoń W, Beyer C. Effect of NMDA on staurosporine-induced activation of caspase-3 and LDH release in mouse neocortical and hippocampal cells. Brain Res Dev Brain Res 2005;160:40–52.
Kaech S, Banker G. Culturing hippocampal neurons. Nat Protoc 2006;1:2406–15.
Kajta M, Lasoń W, Kupiec T. Effects of estrone on N-methyl-d-aspartic acid- and staurosporine-induced changes in caspase-3-like protease activity and lactate dehydrogenase-release: time- and tissue-dependent effects in neuronal primary cultures. Neuroscience 2004;123:515–26.
Koh JY, Choi DW. Quantitative determination of glutamate mediated cortical neuronal injury in cell culture by lactate dehydrogenase efflux assay. J Neurosci Methods 1987;20:83–90.
Nicholson DW, Ali A, Thornberry NA, Vaillancourt JP, Ding CK, Gallant M. Identification and inhibition of the ICE/CED-3 protease necessary for mammalian apoptosis. Nature 1995;376:37–43.
Gomes A, Fernandes E, Lima JLFC. Fluorescence probes used for detection of reactive oxygen species. J Biochem Biophys Methods 2005;65:45–80.
Vatsyayan R, Kothari H, Pendurthi UR, Rao LVM. 4-Hydroxy-2-nonenal enhances tissue factor activity in human monocytic cells via p38 mitogen-activated protein kinase activation-dependent phosphatidylserine exposure. Arterioscler Thromb Vasc Biol 2013;33:1601–11.
Lobner D, Piana PMT, Salous AK, Peoples RW. Beta-N-methylamino-l-alanine enhances neurotoxicity through multiple mechanisms. Neurobiol Dis 2007;25:360–6.
Ling L-U, Tan K-B, Lin H, Chiu GNC. The role of reactive oxygen species and autophagy in safingol-induced cell death. Cell Death Dis 2011;2:e129.
Tetz LM, Kamau PW, Cheng AA, Meeker JD, Loch-Caruso R. Troubleshooting the dichlorofluorescein assay to avoid artifacts in measurement of toxicant-stimulated cellular production of reactive oxidant species. J Pharmacol Toxicol Methods 2013;67:56–60.
Honkisz E, Wójtowicz AK. Modulation of estradiol synthesis and aromatase activity in human choriocarcinoma JEG-3 cells exposed to tetrabromobisphenol A. Toxicol In vitro 2015;29:44–50.
Kling P, Förlin L. Proteomic studies in zebrafish liver cells exposed to the brominated flame retardants HBCD and TBBPA. Ecotoxicol Environ Saf 2009;72:1985–93.
Al-Mousa F, Michelangeli F. Some commonly used brominated flame retardants cause Ca2+-ATPase inhibition, beta-amyloid peptide release and apoptosis in SH-SY5Y neuronal cells. PLoS ONE 2012;7:e33059.
Nicholson DW. Caspase structure, proteolytic substrates, and function during apoptotic cell death. Cell Death Differ 1999;6:1028–42.
Nikoletopoulou V, Markaki M, Palikaras K, Tavernarakis N. Crosstalk between apoptosis, necrosis and autophagy. Biochim Biophys Acta 2013;1833:3448–59.
Bazzoni F, Beutler B. The tumor necrosis factor ligand and receptor families. N Engl J Med 1996;334:1717–25.
Elmore S. Apoptosis: a review of programmed cell death. Toxicol Pathol 2007;35:495–516.
Ashkenazi A, Holland P, Eckhardt SG. Ligand-based targeting of apoptosis in cancer: the potential of recombinant human apoptosis ligand 2/Tumor necrosis factor-related apoptosis-inducing ligand (rhApo2L/TRAIL). J Clin Oncol 2008;26:3621–30.
Reistad T, Mariussen E, Fonnum F. The effect of a brominated flame retardant, tetrabromobisphenol-A, on free radical formation in human neutrophil granulocytes: the involvement of the MAP kinase pathway and protein kinase C. Toxicol Sci 2005;83:89–100.
Hendriks HS, Meijer M, Muilwijk M, Van Den Berg M, Westerink RHS. A comparison of the in vitro cyto- and neurotoxicity of brominated and halogen-free flame retardants: prioritization in search for safe(r) alternatives. Arch Toxicol 2014;88:857–69.
Tollefson KE, Kroczynski J, Cutaia MV. Time-dependent interactions of oxidant-sensitive fluoroprobes with inhibitors of cellular metabolism. Lab Invest 2003;83:367–75.
Chen X, Zhong Z, Xu Z, Chen L, Wang Y. 2′,7′-Dichlorodihydrofluorescein as a fluorescent probe for reactive oxygen species measurement: forty years of application and controversy. Free Radic Res 2010;44:587–604.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Szychowski, K.A., Wójtowicz, A.K. TBBPA causes neurotoxic and the apoptotic responses in cultured mouse hippocampal neurons in vitro. Pharmacol. Rep 68, 20–26 (2016). https://doi.org/10.1016/j.pharep.2015.06.005
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2015.06.005