Abstract
Objectives
Fibroid tumor growth in the myometrium appears to be regulated by estrogens but the role of estrogen receptor (ER) coregulators, such as the steroid receptor coactivator (SRC) family members, in fibroid growth is currently unknown. The aims of this study were to compare the expression of the SRC-1, SRC-2, and SRC-3 coactivators between fibroids and normal myometrium in pure populations of cultured smooth muscle cells (SMC) and microvascular endothelial cells (MEC), and also between both cell types, and to identify any relationship between the SRC expression profiles and the known ER status of the SMC and MEC samples examined in this study.
Methods
Reverse transcriptase—polymerase chain reaction (RT-PCR) coupled with Southern blot analysis was used to derive a semiquantitative estimate of the relative levels of SRC-1, SRC-2, and SRC-3 expression in pure populations of SMC (>98% α-smooth muscle actin [SMA]+) and MEC (>99% CD31+) isolated and cultured from eight samples of paired human myometrial and fibroid tissue.
Results
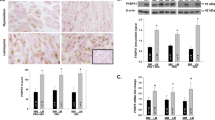
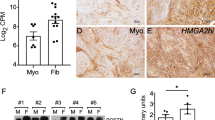
The mean levels of SRC-1, SRC-2, and SRC-3 were each similar in normal myometrium compared to fibroids for SMC and also for MEC. However, SRC-1, SRC-2, and SRC-3 levels were each significantly higher in SMC compared to MEC from both myometrial and fibroid samples, although for SRC-3 there was a trend for higher levels in myometrial samples that did not reach significance. While all SMC samples expressed ERa and high coactivator levels, there does not appear to be a relationship between coactivator expression levels and the presence or absence of ERa in MEC samples.
Conclusion
Coactivators may be more important in ERa-mediated growth of SMC than for MEC. Although the SRC family members are likely to play a role in the response of fibroid SMC to estrogen, via ERa, changes in their levels do not appear to contribute to the increased sensitivity of fibroid SMC to estrogen.
Similar content being viewed by others
References
Vollenhoven B. Introduction: The epidemiology of uterine leiomyomas. Baillieres Clin Obstet Gynaecol 1998;12:169–76.
Scully R. Pathology of leiomyomas. Semin Reprod Endocrinol 1992;10:325–31.
Casey R, Rogers PA, Vollenhoven BJ. An immunohistochemical analysis of fibroid vasculature. Hum Reprod 2000;15:1469–75.
Flake GP, Andersen J, Dixon D. Etiology and pathogenesis of uterine leiomyomas: A review. Environ Health Perspect 2003;111:1037–54.
Vollenhoven BJ, Pearce P, Herington AC, Healy DL. Steroid receptor binding and messenger RNA expression in fibroids from untreated and gonadotrophin-releasing hormone agonist pretreated women. Clin Endocrinol (Oxf) 1994;40:537–44.
Andersen J, Barbieri RJL. Abnormal gene expression in uterine leiomyomas. J Soc Gynecol Investig 1995;2:663–72.
Brandon DD, Erickson TE, Keenan EJ, et al. Estrogen receptor gene expression in human uterine leiomyomata. J Clin Endocrinol Metab 1995;80:1876–81.
Lessl M, Klotzbuecher M, Schoen S, Reles A, Stockemann K, Fuhrmann U. Comparative messenger ribonucleic acid analysis of immediate early genes and sex steroid receptors in human leiomyoma and healthy myometrium. J Clin Endocrinol Metab 1997;82:2596–600.
Englund K, Blanck A, Gustavsson I, et al. Sex steroid receptors in human myometrium and fibroids: Changes during the menstrual cycle and gonadotropin-releasing hormone treatment. J Clin Endocrinol Metab 1998;83:4092–6.
Andersen J, DyReyes VM, Barbieri RL, Coachman DM, Miksicek RJ. Leiomyoma primary cultures have elevated transcriptional response to estrogen compared with autologous myometrial cultures. J Soc Gynecol Investig 1995;2:542–51.
McKenna NJ, O’Malley BW. Combinatorial control of gene expression by nuclear receptors and coregulators. Cell 2002;108:465–74.
Xu J, Li Q. Review of the in vivo functions of the p160 steroid receptor coactivator family. Mol Endocrinol 2003;17:1681–92.
List HJ, Lauritsen KJ, Reiter R, Powers C, Wellstein A, Riegel AT. Ribozyme targeting demonstrates that the nuclear receptor coactivator AIB1 is a rate-limiting factor for estrogen-dependent growth of human MCF-7 breast cancer cells. J Biol Chem 2001;276:23763–8.
Cavarretta IT, Mukopadhyay R, Lonard DM, et al. Reduction of coactivator expression by antisense oligodeoxynucleotides inhibits ERalpha transcriptional activity and MCF-7 proliferation. Mol Endocrinol 2002;16:253–70.
Tai H, Kubota N, Kato S. Involvement of nuclear receptor coactivator SRC-1 in estrogen-dependent cell growth of MCF-7 cells. Biochem Biophys Res Commun 2000;267:311–6.
Xu J, Qiu Y, DeMayo FJ, Tsai SY, Tsai MJ, O’Malley BW. Partial hormone resistance in mice with disruption of the steroid receptor coactivator-1 (SRC-1) gene. Science 1998;279:1922–5.
Smith CL, O’Malley BW. Coregulator function: A key to understanding tissue specificity of selective receptor modulators. Endocr Rev 2004;25:45–71.
Liao L, Kuang SQ, Yuan Y, Gonzalez SM, O’Malley BW, Xu J. Molecular structure and biological function of the cancer-amplified nuclear receptor coactivator SRC-3/AIB1. J Steroid Biochem Mol Biol 2003;83:3–14.
Kershah SM, Desouki MM, Koterba KL, Rowan BG. Expression of estrogen receptor coregulators in normal and malignant human endometrium. Gynecol Oncol 2004;92:304–13.
Suen CS, Berrodin TJ, Mastroeni R, Cheskis BJ, Lyttle CR, Frail DE. A transcriptional coactivator, steroid receptor coactivator-3, selectively augments steroid receptor transcriptional activity. J Biol Chem 1998;273:27645–53.
Wong CW, Komm B, Cheskis BJ. Structure-function evaluation of ER alpha and beta interplay with SRC family coactivators. ER selective ligands. Biochemistry 2001;40:6756–65.
Gargett CE, Bucak K, Zaitseva M, et al. Estrogen receptor-alpha and -beta expression in microvascular endothelial cells and smooth muscle cells of myometrium and leiomyoma. Mol Hum Reprod 2002;8:770–5.
Noyes RW, Hertig AT, Rock J. Dating the endometrial biopsy. Am J Obstet Gynecol 1975;122:262–3.
Gargett CE, Bucak K, Rogers PA. Isolation, characterization and long-term culture of human myometrial microvascular endothelial cells. Hum Reprod 2000;15:293–301.
Hussein-Fikret S, Fuller PJ. Expression of nuclear receptor co-regulators in ovarian stromal and epithelial tumours. Mol Cell Endocrinol 2005;229:149–60.
Vienonen A, Miettinen S, Blauer M, et al. Expression of nuclear receptors and cofactors in human endometrium and myometrium. J Soc Gynecol Investig 2004;11:104–12.
Wei J, Chiriboga L, Mizuguchi M, Yee H, Mittal K. Expression profile of tuberin and some potential tumorigenic factors in 60 patients with uterine leiomyomata. Mod Pathol 2005;18:179–88.
Gregory CW, Wilson EM, Apparao KB, et al. Steroid receptor coactivator expression throughout the menstrual cycle in normal and abnormal endometrium. J Clin Endocrinol Metab 2002;87:2960–6.
Wieser F, Schneeberger C, Hudelist G, et al. Endometrial nuclear receptor co-factors SRC-1 and N-CoR are increased in human endometrium during menstruation. Mol Hum Reprod 2002;8:644–50.
Shiozawa T, Shih HC, Miyamoto T, et al. Cyclic changes in the expression of steroid receptor coactivators and corepressors in the normal human endometrium. J Clin Endocrinol Metab 2003;88:871–8.
Uchikawa J, Shiozawa T, Shih HC, et al. Expression of steroid receptor coactivators and corepressors in human endometrial hyperplasia and carcinoma with relevance to steroid receptors and Ki-67 expression. Cancer 2003;98:2207–13.
Glaeser M, Floetotto T, Hanstein B, Beckmann MW, Niederacher D. Gene amplification and expression of the steroid receptor coactivator SRC3 (AIB1) in sporadic breast and endometrial carcinomas. Horm Metab Res 2001;33:121–6.
Anzick SL, Kononen J, Walker RL, et al. AIB1, a steroid receptor coactivator amplified in breast and ovarian cancer. Science 1997;277:965–8.
Havrilesky LJ, McMahon CP, Lobenhofer EK, Whitaker R, Marks JR, Berchuck A. Relationship between expression of coactivators and corepressors of hormone receptors and resistance of ovarian cancers to growth regulation by steroid hormones. J Soc Gynecol Investig 2001;8:104–13.
Folkman J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat Med 1995;1:27–31.
Morales DE, McGowan KA, Grant DS, et al. Estrogen promotes angiogenic activity in human umbilical vein endothelial cells in vitro and in a murine model. Circulation 1995;91:755–63.
Johns A, Freay AD, Fraser W, Korach KS, Rubanyi GM. Disruption of estrogen receptor gene prevents 17 beta estradiol-induced angiogenesis in transgenic mice. Endocrinology 1996;137:4511–3.
Suzuma I, Mandai M, Takagi H, et al. 17 Beta-estradiol increases VEGF receptor-2 and promotes DNA synthesis in retinal microvascular endothelial cells. Invest Ophthalmol Vis Sei 1999;40:2122–9.
Gargett CE, Zaitseva M, Bucak K, Chu S, Fuller PJ, Rogers PA. 17Beta-estradiol up-regulates vascular endothelial growth factor receptor-2 expression in human myometrial microvascular endothelial cells: Role of estrogen receptor-alpha and -beta. J Clin Endocrinol Metab 2002;87:4341–9.
Zaitseva M, Yue DS, Katzenellenbogen JA, Rogers PA, Gargett CE. Estrogen receptor-alpha agonists promote angiogenesis in human myometrial microvascular endothelial cells. J Soc Gynecol Investig 2004;11:529–35.
Weston G, Trajstman AC, Gargett CE, Manuelpillai U, Vollenhoven BJ, Rogers PA. Fibroids display an anti-angiogenic gene expression profile when compared with adjacent myometrium. Mol Hum Reprod 2003;9:541–9.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Australia Bank Ovarian Cancer Research Foundation PhD Scholarship Grant (S.H-F.), the National Health and Medical Research Council of Australia Senior Principal Research Fellowship (P.J.F.), and Grants No. 284344 and 124331, and the Monash University Early Career Researcher Grant (C.E.G.).
The authors thank Marina Zaitseva and Kris Bucak for technical assistance; Drs Mark Lawrence, Peter Lutjen, and Beverley Vollenhoven at Monash Medical Centre for provision of hysterectomy tissue; Nancy Taylor for collection of tissue; and Mai Forsyth and the Australian Red Cross Service, Melbourne, for provision of male human serum.
Rights and permissions
About this article
Cite this article
Hussein-Fikret, S., Fuller, P.J. & Gargett, C.E. Expression of Steroid Receptor Coactivators in Cultured Cells From Paired Myometrial and Fibroid Tissues. Reprod. Sci. 12, 445–451 (2005). https://doi.org/10.1016/j.jsgi.2005.04.008
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jsgi.2005.04.008