Abstract
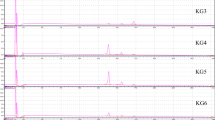
The objective of this study was to perform preliminary screening of phytochemical compounds and quantification of major phenolics and flavonoid markers in Italian ryegrass extract using HPLC-DAD. Previously, LC-MS analysis has identified different phenolic acids, including caffeic acid, ferulic acid, p-coumaric acid, chlorogenic acid, dihydroxy benzoic acid, propyl gallate, catechin, and six flavonoids including rutin hydroxide, luteolin, kaemferol, vitexin, narcissoside, and myricetin from Italian ryegrass extract. In the present study, Italian ryegrass silage powder was extracted with ethanol: water for 20 min at 90°C. The extract targeted optimum yield of phenolic acids and flavonoids. Crude phenolic acid and flavonoids were then purified by solid phase extraction method. Purified fractions were then injected into HPLC with a diode-array detector. Quantified concentrations of isolated phenolic acids and flavonoids ranged from 125 to 220 μg/g dry weight. Limits of detection and limits of quantification for all standards (unknown compounds) ranged from 0.38 to 1.71 and 0.48 to 5.19 μg/g dry weight, respectively. Obtained values were compared with previous literatures, indicating that our HPLC-DAD quantification method showed more sensitivity. This method showed better speed, accuracy, and effectiveness compared to previous reports. Furthermore, this study could be very useful for develo** phenolic acids and flavonoids from compositions in Italian ryegrass silage feed for pharmaceutical applications and ruminant animals in livestock industries.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Badalica-Petrescu, M., Dragan, S., Ranga, F., Fetea, F., Socaciu, C., 2014. Comparative HPLC-DAD-ESI(+) ms fingerprint and quantification of phenolic and flavonoid composition of aqueous leaf extracts of Cornusmas and Crataegus monogyna, in relation to their cardiotonic potential. Not. Bot. Horti. Agrobo. 42, 9–18.
Baldinger, L., Zollitsch, W., Knaus, W.F., 2012. The use of Italian ryegrass silage in a low input dairy cow feeding system in Austrian organic agriculture. Org. Agric. 2, 43–53.
Buono, D.D., Ioli, G., Nasini, L., Proietti, P., 2011. A comparative study on the interference of two herbicides in wheat and Italian ryegrass and on their antioxidant activities and detoxification rates. J. Agric. Food Chem. 59, 12109–12115.
Burin, V.M., Arcari, S.G., Costa, L.L.F., Bordignon-Luiz, M.T., 2011. Determination of some phenolic compounds in red wine by RP-HPLC: method development and validation. J. Chromatogr. Sci. 49, 647–651.
Campagnoli, A., Dell’Orto, V., 2013. Potential application of electronic olfaction systems in feedstuffs analysis and animal nutrition. Sensors (Basel) 13, 14611–14632.
Chavan, J.J., Gaikwad, N.B., Kshirsagar, P., 2013. Total phenolics, flavonoids and antioxidant properties of three Ceropegia species from Western Ghats of India. S. Afr. J. Bot. 88, 273–277.
Chen, H.J., Inbaraj, B.S., Chen, B.H., 2012. Determination of phenolic acids and flavonoids in Taraxacum formosanum Kitam by liquid chromatography-tandem mass spectrometry coupled with a post-column derivatization technique. Int. J. Mol. Sci. 13, 260–285.
Choi, G.J., Ji, H.C., Kim, K.Y., Park, H.S., Seo, S., Lee, K.W., Lee, S.H., 2011. Growth characteristics and productivity of cold-tolerant “Kowinearly” Italian ryegrass in the northern part of South Korea. Afr. J. Biotechnol. 10, 2676–2682.
Choi, K.C., Son, Y.O., Hwang, J.M., Kim, B.T., Chae, M., Lee, J.C., 2017. Antioxidant, antiinflammatory and anti-septic potential of phenolic acids and flavonoid fractions isolated from Lolium multiflorum. Pharm. Biol. 55, 611–619.
Costa, G.M., Ortmann, C.F., Schenkel, E.P., Reginatto, F.H., 2011. An HPLC-DAD method to quantification of main phenolic compounds from leaves of Cecropia species. J. Braz. Chem. Soc. 22, 1096–1102.
De la Torre-Carbot, K., Jauregui, O., Gimeno, E., Castellote, A.I., Lamuela-Raventós, R.M., López-Sabater, M.C., 2005. Characterization and quantification of phenolic compounds in olive oils by solid-phase extraction, HPLC-DAD, and HPLC-MS/MS. J. Agric. Food Chem. 53, 4331–4340.
De Rijke, E., Out, P., Niessen, W.M.A., Ariese, F., Gooijer, C., Brinkman, U.A., 2006. Analytical separation and detection methods for flavonoids. J. Chromatogr. A 1112, 31–63.
Dohi, H., Yamada, A., Fukukawa, T., 1997. Intake stimulants in perennial ryegrass (Lolium perenne L.) fed to sheep. J. Diary Sci. 80, 2083–2086.
Fan, X.H., Cheng, Y.Y., Ye, Z.L., Lin, R.C., Qian, Z.Z., 2006. Multiple chromatographic fingerprinting and its application to the quality control of herbal medicines. Anal. Chim. Acta 555, 217–224.
Francisco, M.L.d.L., Resurreccion, A.V.A., 2009. Development of reversed phase high performance liquid chromatography (RP-HPLC) procedure forthe simultaneous determination of phenolic compounds in peanut skin extracts. Food Chem. 117, 356–363.
Ghasemzadeh, A., Ghasemzadeh, N., 2011. Flavonoids and phenolic acids: role and biochemical activity in plants and human. J. Med. Plant Res. 531, 6697–6703.
Gomez-Alonso, S., Garcıa-Romero, E., Hermosın-Gutierrez, I., 2007. HPLC analysis of diverse grape and wine phenolics using direct injection and multi-detection by DAD and fluorescence. J. Food Compos. Anal. 20, 618–626.
Inbaraj, S., Lu, H., Kao, T.H., Chen, B.H., 2010. Simultaneous determination of phenolic acids and flavonoids in Lycium barbarum Linnaeus by HPLC-DAD-ESI-MS. J. Pharm. Biomed. Anal. 51, 549–556.
**, Y., **ao, Y.S., Zhang, F.F., Xue, X.Y., Xu, Q., Liang, X.M., 2008. Systematic screening and characterization of flavonoid glycosides in Carthamus tinctorius L. by liquid chromatography/UV diode-array detection/electrospray ionization tandem mass spectrometry. J. Pharm. Biomed. Anal. 46, 418–430.
Kao, T.H., Huang, S.C., Inbaraj, B.S., Chen, B.H., 2008. Determination of flavonoids and saponins in Gynostemma pentaphyllum (Thunb.) Makino by liquid chromatography-mass spectrometry. Anal. Chim. Acta 26, 200–211.
Khoddami, A., Wilkes, M.A., Roberts, T.H., 2013. Techniques forthe analysis of plant phenolic compounds. Molecules 18, 2328–2375.
Magwaza, L.S., Opara, U.L., Cronje, P.J.R., Landahl, S., Ortiz, J.O., Terry, L.A., 2016. Rapid methods for extracting and quantifying phenolic compounds in citrus rinds. Food Sci. Nutr. 4, 4–10.
Mocan, A., Vlase, L., Vodnar, D.C., Bischin, C., Hanganu, D., Gheldiu, A.M., Oprean, R., Silaghi-Dumitrescu, R., Crişan, G., 2014. Polyphenolic content, antioxidant and antimicrobial activities of Lycium barbarum L. and Lycium chinense Mill. leaves. Molecules 19, 10056–10073.
Naczk, M., Shahidi, F., 2004. Extraction and analysis of phenolics in food. J. Chromatogr. A 1054, 95–111.
NRC, 1996. Nutrient Requirements of Beef Cattle, 7th revised ed. National Research Council, National Academy Press, Washington, DC
Numonov, S.R., Qureshi, M.N., Aisa, H.A., 2015. Development of HPLC protocol and simultaneous quantification of four free flavonoids from Dracocephalum heterophyllum Benth. Int. J. Anal. Chem. 503139, 1–5.
Pereira, V., Camara, J.S., Cacho, J., Marques, J.C., 2010. HPLC-DAD methodology for the quantification of organic acids, furans and polyphenols by direct injection of wine samples. J. Sep. Sci. 33, 1204–1215.
Pirisi, F.M., Cabras, P., Cao, C.F., Migliorini, M., Muggelli, M., 2000. Phenolic compounds in virgin olive oil reappraisal of the extraction, HPLC separation, and quantification procedures. J. Agric. Food Chem. 48, 1191–1196.
Plazonić, A., Bucar, F., Maleš, Ž., Mornar, A., Nigović, B., Kujundžić, N., 2009. Identification and quantification of flavonoids and phenolic acids in burr parsley (Caucalis platycarpos L.) using high-performance liquid chromatography with diode array detection and electrospray ionization mass spectrometry. Molecules 14, 2466–2490.
Pulkkinen, M., Gautam, M., Lampi, A.M., Ollilainen, V., Stoddard, F., Sontag-Strohm, T., Salovaara, H., Piironen, V., 2015. Determination of vicine and convicine from faba bean with an optimized high-performance liquid chromatographic method. Food Res. Int. 76, 166–177.
Q2B CH. Validation of analytical procedures: methodology. Presented at International Conference on Harmonization, Geneva, Switzerland, March 1996.
Restivo, A., Degano, I., Ribechini, E., Colombini, M.P., 2014. Development and optimization of an HPLC-DAD-ESI-Q-TOF method for the determination of phenolic acids and derivatives. PLOS ONE 9, https://doi.org/10.1371/journal.pone.0088762.
Robbins, R.J., 2003. Phenolic acids in foods. J. Agric. Food Chem. 51, 2866–2887.
Seal, T., 2016. Quantitative HPLC analysis of phenolic acids, flavonoids and ascorbic acid in four different solvent extracts of two wild edible leaves, Sonchus arvensis and Oenanthe linearis of North-Eastern region in India. J. Appl. Pharm. Sci. 6, 157–166.
Shirazi, O.U., Khattak, M.M.A.K., Shukri, N.A.M., Nasyriq, M.N., 2014. Determination of total phenolic, flavonoid content and free radical scavenging activities of common herbs and spices. J. Pharmacogn. Phytochem. 3, 104–108.
Vagiri, M., Ekholm, A., Andersson, S.C., Johansson, E., Rumpunen, K., 2012. An optimized method for analysis of phenolic compounds in buds, leaves, and fruits of black currant (Ribes nigrum L.). J. Agric. Food Chem. 60, 10501–10510.
Valan Arasu, M., Ilavenil, S., Kim, D.H., Gun Roh, S., Lee, J.C., Choi, K.C., 2014. In vitro and in vivo enhancement of adipogenesis by Italian ryegrass (Lolium multiflorum) in 3T3-L1 cells and mice. PLOS ONE 9, https://doi.org/10.1371/journal.pone.0085297.
Zhang, A., Wan, L., Wu, C., Fang, Y., Han, G., Li, H., Zhang, Z., Wang, H., 2013. Simultaneous determination of 14 phenolic compounds in grape canes by HPLC-DAD-UV using wavelength switching detection. Molecules 18, 14241–14257.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author’s contribution
KCC conceived and designed experiments. SI and SS collected and extracted L. multiflorum plant extract. PK performed separation of phenolic acids and flavonoids from L. multiflorum plant, developed the HPLC-DAD method, and conducted measurements. CES and MVA helped method development and performed data analysis. PK wrote the manuscript. All authors read, commented, and approved the final manuscript.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kuppusamy, P., Lee, K.D., Song, C.E. et al. Quantification of major phenolic and flavonoid markers in forage crop Lolium multiflorum using HPLC-DAD. Rev. Bras. Farmacogn. 28, 282–288 (2018). https://doi.org/10.1016/j.bjp.2018.03.006
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2018.03.006