Abstract
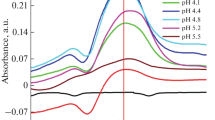
A highly sensitive and simple solid-phase colorimetry for Cr(VI) was proposed. It was based on the ion-pair solid-phase extraction of Cr-diphenylcarbazide (DPC) complex with sedimentable dispersed particulates. The concentration of Cr(VI) was measured from the color tones obtained by image analysis of the photo of sediment. Various conditions, e.g., material and amounts of adsorbent particulates, chemical properties and concentration of counter ions, and pH, were optimized for the formation and quantitative extraction of the complex. In the recommended procedure, 1 mL of sample was put into a 1.5 mL microtube where powder form adsorbent and reagents, i.e., XAD-7HP particles, DPC, sodium dodecyl sulfate, amido sulfuric acid, and sodium chloride had been packed. The analytical operation was completed within 5 min by gently shaking the microtube and allowing it to stand until enough amounts of particulates were deposited to take a picture. Chromium (VI) up to 2.0 ppm was determined, and the detection limit was 0.0034 ppm. The sensitivity was enough to determine Cr(VI) at lower concentrations than the water quality of standard (0.02 ppm). This method was successfully applied to the analysis of simulated industrial wastewater samples. The stoichiometry of the extracted chemical species was also investigated by applying the same equilibrium model as the ion-pair solvent extraction.
Graphical abstract







Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
B. Dhal, H.N. Thatoi, N.N. Das, B.D. Pandey, Hazard. Mater. (2013). https://doi.org/10.1016/j.jhazmat.2013.01.048
M.-H. Ho, C.-M. Chan, I. Bakar, Int. J. Sustain. Constr. Eng. (2011) https://publisher.uthm.edu.my/ojs/index.php/IJSCET/article/view/66
M. Chrysochoou, S.C. Fakra, M.A. Marcus, H.M. Deok, D. Dermatas, Environ. Sci. Technol. (2009). https://doi.org/10.1021/ES9005338
X. Liuyang, H. Yang, S. Huang, Y. Zhang, S. **a, J. Environ. Chem. Eng. (2020). https://doi.org/10.1016/j.jece.2020.104474
M.A. Schneegurt, J.C. Jain, J.A. Menicucci, S.A. Brown, K.M. Kemner, D.F. Garofalo, M.R. Quallick, C.R. Neal, J.F. Kulpa, Environ. Sci. Technol. (2001). https://doi.org/10.1021/es010766e
C. Földi, R. Dohrmann, K. Matern, T. Mansfeldt, J. Soils Sediments. (2013). https://doi.org/10.1007/S11368-013-0714-2
A.M. Zayed, N. Terry, Plant Soil. (2003). https://doi.org/10.1023/A:1022504826342
Chromium in drinking-water Background document for development of WHO Guidelines for drinking-water quality, 2020. http://apps.who.int/bookorders Accessed 7 October 2021
Chromium in Drinking Water | US EPA, https://www.epa.gov/sdwa/chromium-drinking-water Accessed 7 October 2021
Chromium in Water - Theory, http://www.acornusers.org/education/HNC-Web/Theory.html#Top Accessed 8 October 2021
N. Unceta, F. Séby, J. Malherbe, O.F.X. Donard, Anal. Bioanal. Chem. (2010). https://doi.org/10.1007/s00216-009-3417-1
S. Gumfawar, B. Godboley, A Review on removal of heavy metal (Cr and Cd) using plant seeds for purification of water. Engineering 6, 934–937 (2017)
Diphenylcarbazide - an overview | ScienceDirect Topics, https://www.sciencedirect.com/topics/pharmacology-toxicology-and-pharmaceutical-science/diphenylcarbazide Accessed 8 October 2021
SW-846 Test Method 7196A: Chromium, Hexavalent (Colorimetric) | US EPA, https://www.epa.gov/hw-sw846/sw-846-test-method-7196a-chromium-hexavalent-colorimetric Accessed 30 August 2022
R. Soares, M.C. Carneiro, M.I.C. Monteiroa, S. S. de Henriquae, F.V.M. Pontes, L.I. Dias da Silva, A.A. Neto, R.E. Santelli, Chem. Speciat. Bioavailab. (2009) https://doi.org/10.3184/095422909X466095
A. Sanchez-Hachair, A. Hofmann, Compt. Rendus Chim. (2018). https://doi.org/10.1016/J.CRCI.2018.05.002
M. Gardner, S. Comber, Analyst. (2002). https://doi.org/10.1039/B109374F
K.K. Onchoke, S.A. Sasu, Adv. Environ. Chem. (2016). https://doi.org/10.1155/2016/3468635
K. Shigeta, A. Fujita, T. Nakazato, H. TAO, Anal. Sci. (2018) https://doi.org/10.2116/ANALSCI.18P012
Z. Li, Y. Shi, P. Gao, X. Gu, T. Zhou, Fresenius’ J. Anal. Chem. (1997). https://doi.org/10.1007/S002160050458
S. Matsuoka, Y. Nakatsu, K. Takehara, S. Saputro, K. Yoshimura, Anal. Sci. (2006). https://doi.org/10.2116/analsci.22.1519
D.E. Kimbrough, Y. Cohen, A.M. Winer, L. Creelman, C. Mabuni, Crit. Rev. Environ. Sci. Technol. (2010). https://doi.org/10.1080/10643389991259164
F.J. Alguacil, A.G. Coedo, M.T. Dorado, A.M. Sastre, Hydrometallurgy (2001). https://doi.org/10.1016/S0304-386X(01)00147-5
V. Camel, Acta Part B At. Spectrosc. (2003). https://doi.org/10.1016/S0584-8547(03)00072-7
I.M.M. Rahman, Z.A. Begum, H. Hasegawa, Microchem. J. (2013). https://doi.org/10.1016/J.MICROC.2013.06.006
B. Galán, D. Castañeda, I. Ortiz, Water Res. (2005). https://doi.org/10.1016/J.WATRES.2005.08.015
N. Rajesh, B.G. Mishra, P.K. Pareek, Spectrochim. Biomol. Spectrosc. Acta - Part A Mol. (2008). https://doi.org/10.1016/j.saa.2007.05.011
N. Rajesh, R.K. Jalan, P. Hotwany, J. Hazard. Mater. (2008). https://doi.org/10.1016/j.jhazmat.2007.05.025
R. Verma, S. Sarkar, Ind. Eng. Chem. Res. (2020). https://doi.org/10.1021/ACS.IECR.0C04347
T. Kamidate, T. Segawa, H. Watanabe, K. Yamaguchi, Anal. Sci. (1989). https://doi.org/10.2116/ANALSCI.5.429
T. Inui, K. Fujita, M. Kitano, T. Nakamura, Anal. Sci. (2010). https://doi.org/10.2116/ANALSCI.26.1093
S.I. Hasegawa, ISIJ Int. (2014). https://doi.org/10.2355/ISIJINTERNATIONAL.54.131
M. Sarenqiqige, K. Ashitomi, Yoshimura Anal. Sci. (2013). https://doi.org/10.2116/ANALSCI.29.823
Y. Yokota, A. Manaka, M. Tafu, T. Kato, K. Tomita, T. Akazawa, Bull. Chem. Soc. Jpn. (2019). https://doi.org/10.1246/bcsj.20180351
T. Kato, Y. Nagashima, A. Manaka, C. Nakamura, S. Oshite, S. Igarashi, Anal. Sci. (2019). https://doi.org/10.2116/ANALSCI.19N001
A. Manaka, Y. Ueno, M. Tafu, T. Kato, Anal. Sci. (2022). https://doi.org/10.2116/ANALSCI.21N029
K. Yoshimura, S. Ohashi, Talanta (1978). https://doi.org/10.1016/0039-9140(78)80041-1
S. Saputro, K. Yoshimura, S. Matsuika, K. Takehara, Anal. Sci. (2009). https://doi.org/10.2116/analsci.25.1445
S. Sarenqiqige, S. Saputro, M. Kai, S. Satoda, K. Matsuoka, Yoshimura Anal. Sci. (2013). https://doi.org/10.2116/ANALSCI.29.677
T. Takahashi, E. Kaneko, T. Yotsuyanagi, Anal. Sci. (2006). https://doi.org/10.2116/analsci.22.1585
H. Asano, Y. Shiraishi, Anal. Sci. (2018). https://doi.org/10.2116/analsci.34.71
H.M. Zhai, T. Zhou, F. Fang, Z.Y. Wu, Talanta (2020). https://doi.org/10.1016/j.talanta.2019.120635
A. Muhammed, A. Hussen, M. Redi, T. Kaneta, Anal. Sci. (2021). https://doi.org/10.2116/analsci.20P325
W. Tan, Aruna, Z. Xu, L. Zhang, W. Shen (2020) Talanta. https://doi.org/10.1016/j.talanta.2020.121116
W. Alahmad, P. Varanusupakul, T. Kaneta, P. Varanusupakul, Anal. Chim. Acta. (2019). https://doi.org/10.1016/J.ACA.2019.08.002
W. Alahmad, N. Tungkijanansin, T. Kaneta, P. Varanusupakul, Talanta (2018). https://doi.org/10.1016/j.talanta.2018.07.056
N. Kohama, K. Matsuhira, T. Okazaki, K. Sazawa, N. Hata, H. Kuramitz, S. Taguchi, Anal Chem Bioanal (2022). https://doi.org/10.1007/s00216-022-04375-y
D.K. Sharma, R. Sharma, Indian J. Sci. Technol. (2015) https://doi.org/10.17485/IJST/2015/V8I11/71773
L. Liu, Y. Leng, H. Lin, Microchim. Acta. (2016). https://doi.org/10.1007/S00604-016-1777-8
T. Watanabe, H. Mizuguchi, J. Shida, Bunseki Kagaku (2011). https://doi.org/10.2116/bunsekikagaku.60.339
S. Taguchi, K. Nakamura, T. Hiraide, K. Goto, Bunseki Kagaku (1982). https://doi.org/10.2116/bunsekikagaku.31.9_548
I. Kasahara, Y. Ohgaki, K. Matsui, K. Kano, S. Taguchi, K. Goto, Nippon Kagaku Kaishi (1986). https://doi.org/10.1246/nikkashi.1986.894
K. Miyabe, S. Taguchi, I. Kasahara, K. Goto, J. Phys. Chem. B. (2000). https://doi.org/10.1021/jp000918c
Acknowledgements
This work was supported by JST SPRING, Grant Number JPMJSP2145and the Japan Society for the Promotion of Science (JSPS) via a Grant-in-Aid for Scientific Research (Grant JP 22H01624).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kohama, N., Okazaki, T., Sazawa, K. et al. Simple solid-phase colorimetry for trace Cr(VI) by combination of complexation with diphenylcarbazide and ion-pair solid-phase extraction with sedimentable dispersed particulates. ANAL. SCI. 39, 857–865 (2023). https://doi.org/10.1007/s44211-023-00286-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s44211-023-00286-9