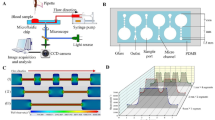
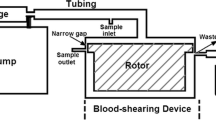
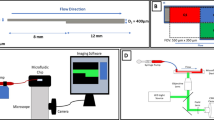
Abstract
Cordycepin can effectively inhibit adenosine diphosphate-induced platelet aggregation; however, the inhibitory effect on shear-induced platelet aggregation is not known. Pathological high shear stress is an important factor in inducing platelet aggregation in the human body. In order to further study the antithrombotic effect and mechanism of cordycepin, in this study, a microfluidic chip model was developed that not only can mimic stenotic vessels but also provide high shear stress needed in the experiment. The inhibitory effects of cordycepin on platelet aggregation, activation of P-selectin and glycoprotein IIb/IIIa, phosphatidyl serine exposure, glycoprotein Ib/von Willebrand factor binding, and Akt protein phosphorylation, induced by a pathological high shear rate (10,000 s−1), were studied. In addition, we also investigated the effect of cordycepin on adenosine diphosphate and arachinodic acid-induced platelet aggregation by turbidimetry. The results showed that cordycepin can inhibit platelet aggregation, P-selectin expression, glycoprotein IIb/IIIa activation, phosphatidylserine exposure, and Akt protein phosphorylation induced by a pathological high shear rate. It did not affect the combination of GPIb and vWF because the combination of CD14-staind platelets and CD41-staind platelets by von Willebrand factor was not inhibited by cordycepin. Under static conditions, cordycepin can inhibit adenosine diphosphate and arachinodic acid-induced platelet aggregation.
Graphical Abstract







Similar content being viewed by others
Data Availability
The data available on the request to the corresponding author.
References
Bark DL, Ku DN (2010) Wall shear over high degree stenoses pertinent to atherothrombosis. J Biomech 43:2970–2977. https://doi.org/10.1016/j.jbiomech.2010.07.011
Casa LDC, Ku DN (2017) Thrombus formation at high shear rates. Annu Rev Biomed Eng 19:415–433. https://doi.org/10.1146/annurev-bioeng-071516-044539
Chan CH, Simmonds MJ, Fraser KH, Igarashi K, Ki KK, Murashige T, Joseph MT, Fraser JF, Tansley GD, Watanabe N (2022) Discrete responses of erythrocytes, platelets, and von Willebrand factor to shear. J Biomech 130:110898. https://doi.org/10.1016/j.jbiomech.2021.110898
Chen Z, Mondal NK, Ding J, Koenig SC, Slaughter MS, Wu ZJ (2016) Paradoxical effect of nonphysiological shear stress on platelets and von Willebrand factor. Artif Organs 40:659–668. https://doi.org/10.1111/aor.12606
Chen Z, Koenig SC, Slaughter MS, Griffith BP, Wu ZJ (2018) Quantitative characterization of shear-induced platelet receptor shedding: glycoprotein iba, glycoprotein VI, and glycoprotein IIb/IIIa. Asaio 64:773–778. https://doi.org/10.1097/MAT.0000000000000722
Chen Z, Li T, Kareem K, Tran D, Griffith BP, Wu ZJ (2019) The role of PI3K/Akt signaling pathway in non-physiological shear stress-induced platelet activation. Artif Organs 43:897–908. https://doi.org/10.1111/aor.13465
Estevez B, Shen B, Du X (2015) Targeting integrin and integrin signaling in treating thrombosis. Arterioscler Thromb Vasc Biol 35:24–29. https://doi.org/10.1161/ATVBAHA.114.303411
Fan HY, Fu FH, Yang MY, Xu H, Zhang AH, Liu K (2010) Antiplatelet and antithrombotic activities of salvianolic acid A. Thromb Res 126:e17-22. https://doi.org/10.1016/j.thromres.2010.04.006
Fuentes E, Wehinger S, Trostchansky A (2021) Regulation of key antiplatelet pathways by bioactive compounds with minimal bleeding risk. Int J Mol Sci 22:12380. https://doi.org/10.3390/ijms222212380
Goncalves I, Nesbitt WS, Yuan Y, Jackson SP (2005) Importance of temporal flow gradients and integrin alphaIIbbeta3 mechanotransduction for shear activation of platelets. J Biol Chem 280:15430–15437. https://doi.org/10.1074/jbc.M410235200
Grande GN, Shankar KN, Sinno T, Diamond SL (2021) Thrombosis and hemodynamics: external and intrathrombus gradients. Curr Opin Biomed Eng 19:100316. https://doi.org/10.1016/j.cobme.2021.100316
He C, Yu L, Dan W, Deng SH, Ma H, Liu BZ, Li Y (2021) Application of a simple microfluidic chip analysis technology to evaluate the inhibitory role of protocatechuic acid on shear-induced platelet aggregation. Evid-Based Compl Alt Med 2021:5574413. https://doi.org/10.1155/2021/5574413
Jurk K, Kehrel BE (2010) Pathophysiology and biochemistry of platelets. Der Internist 51:1086–1094. https://doi.org/10.1007/s00108-010-2595-4
Khodadi E (2020) Platelet function in cardiovascular disease: activation of molecules and activation by molecules. Cardiovasc Toxicol 20:1–10. https://doi.org/10.1007/s12012-019-09555-4
Kwon HW, Shin JH, Lim DH, Ok WJ, Nam GS, Kim MJ, Kwon HK, Noh JH, Lee JK, Kim HH, Kim JL, Park HJ (2016) Antiplatelet and antithrombotic effects of cordycepin-enriched WIB-801CE from Cordyceps militaris ex vivo, in vivo, and in vitro. BMC Complement Altern Med 16:508. https://doi.org/10.1186/s12906-016-1463-8
Larsson A, Eriksson M, Lindahl TL (2002) Studies of fibrinogen binding to porcine platelets by flow cytometry: a method for studies of porcine platelet activation. Platelets 13:153–157. https://doi.org/10.1080/0953371027588
Lee DH, Kim HH, Lim DH, Kim JL, Park HJ (2015) Effect of cordycepin-enriched WIB801C from Cordyceps militaris suppressing fibrinogen binding to glycoprotein IIb/IIIa. Biomol Ther 23:60–70. https://doi.org/10.4062/biomolther.2014.086
Lee DH, Kim HH, Cho HJ, Yu YB, Kang HC, Kim JL, Lee JJ, Park HJ (2014) Cordycepin-enriched WIB801C from Cordyceps militaris inhibits collagen-induced [Ca(2+)]i mobilization via cAMP-dependent phosphorylation of inositol 1,4,5-trisphosphate receptor in human platelets. Biomol Ther 22:223–231. https://www.ncbi.nlm.nih.gov/pubmed/25009703
Leung R, Gwozdz AM, Wang H, Bang KWA, Packham MA, Freedman J, Rand ML (2007) Persistence of procoagulant surface expression on activated human platelets: involvement of apoptosis and aminophospholipid translocase activity. J Thromb Haemost 5:560–570. https://doi.org/10.1111/j.1538-7836.2007.02354.x
McFadyen JD, Schaff M, Peter K (2018) Current and future antiplatelet therapies: emphasis on preserving haemostasis. Nat Rev Cardiol 15:181–191. https://doi.org/10.1038/nrcardio.2017.206
Nesbitt WS, Kulkarni S, Giuliano S, Goncalves I, Dopheide SM, Yap CL, Harper IS, Salem HH, Jackson SP (2002) Distinct glycoprotein Ib/V/IX and integrin alpha IIbbeta 3-dependent calcium signals cooperatively regulate platelet adhesion under flow. J Biol Chem 277:2965–2972. https://doi.org/10.1074/jbc.M110070200
Ngo T, Kim K, Bian Y, Noh H, Lim KM, Chung JH, Bae ON (2019) Antithrombotic effects of paeoniflorin from Paeonia suffruticosa by selective inhibition on shear stress-induced platelet aggregation. Int J Mol Sci 20:5040. https://doi.org/10.3390/ijms20205040
Pang A, Cui Y, Chen Y, Cheng N, Delaney MK, Gu M, Stojanovic-Terpo A, Zhu C, Du X (2018) Shear-induced integrin signaling in platelet phosphatidylserine exposure, microvesicle release, and coagulation. Blood 132:533–543. https://doi.org/10.1182/blood-2017-05-785253
Rahman SM, Hlady V (2019) Downstream platelet adhesion and activation under highly elevated upstream shear forces. Acta Biomater 91:135–143. https://doi.org/10.1016/j.actbio.2019.04.028
Rana A, Westein E, Niego B, Hagemeyer CE (2019) Shear-dependent platelet aggregation: mechanisms and therapeutic opportunities. Front Cardiovasc Med 6:141. https://doi.org/10.3389/fcvm.2019.00141
Sang Y, Roest M, De LB, De GP, Huskens D (2021) Interplay between platelets and coagulation. Blood Rev 46:100733. https://doi.org/10.1016/j.blre.2020.100733
Tovar LF, Rosengarten G, Nasabi M, Sivan V, Khoshmanesh K, Jackson SP, Mitchell A, Nesbitt WS (2013) An investigation on platelet transport during thrombus formation at micro-scale stenosis. PLoS One 8:e74123. https://www.ncbi.nlm.nih.gov/pubmed/24194822
Vander MD, Heemskerk JWM (2019) Platelet biology and functions: new concepts and clinical perspectives. Nat Rev Cardiol 16:166–179. https://doi.org/10.1371/journal.pone.0074123
Zhang TC, Liu L, Huang XJ, Gao XM, Chen D, Huan XR, Li Y (2022) Application of microfluidic chip technology to study the inhibitory effect of tetramethylpyrazine on platelet aggregation, activation, and phosphatidylserine exposure mediated by pathological high shear rate. Blood Coagul Fibrin 34:47–60. https://doi.org/10.1097/MBC.0000000000001179
Funding
This work was supported by Graduate Innovation Fund of Yongchuan Hospital affiliated to Chongqing Medical University (YJSCX202204), and Joint Project of Chongqing Health Commission and Science and Technology Bureau (2023GDRC008).
Author information
Authors and Affiliations
Contributions
XG contributed to the study design, data collection, statistical analysis, data interpretation, and manuscript preparation. TZ contributed to data collection. XH contributed to statistical analysis. CH and XH contributed to data collection and statistical analysis. YL contributed to the manuscript preparation. All the authors read and approved the final manuscript.
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gao, X., Zhang, T., Huang, X. et al. Cordycepin Inhibits Pathological High Shear-Induced Platelet Aggregation, Activation, and Phosphatidylserine Exposure by Regulating the Phosphorylation of Akt Protein. Rev. Bras. Farmacogn. 33, 768–777 (2023). https://doi.org/10.1007/s43450-023-00408-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43450-023-00408-4