Abstract
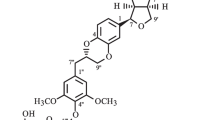
A new lignan (3a → 9′:6a → 9-[(7′S,8′R )-4,4′-dihydroxy-3,3′,5,5′-tetramethoxy-2,7′-cycloligna-7-en-9,9′dicarbonyl]-6b-(p-coumaroyl) sucrose) was isolated from Heliotropium angiospermum Murray, Boraginaceae, along with four known substances: trigonotin A, rosmarinic acid, narcissin, and ethyl lithospermate. Their structures were deduced based on spectroscopic data analysis (MS and NMR of 1H and 13C) and a comparison with the published data. The absolute configuration for the undescribed lignan was determined using electronic circular dichroism analysis. In addition, the anti-inflammatory activity of this aryldihydronaphthalene was investigated by measuring its ability to inhibit the production of cytokines and nitric oxide by stimulated J774 macrophages. The glycosylated lignan, in a range of non-cytotoxic concentrations, reduced the production/release of pro-inflammatory mediators by stimulated macrophages, showing efficacy like that of dexamethasone, a glucocorticoid medication used as a control drug.

Graphical Abstract



Similar content being viewed by others
References
Ai C, Li L (1992) Salvianolic acids D and E: two new depsides from Salvia miltiorrhiza. Planta Med 58:197–199. https://doi.org/10.1055/s-2006-961428
Cinelli MA, Do HT, Miley GP, Silverman RB (2019) Inducible nitric oxide synthase: regulation, structure, and inhibition. Med Res Rev 40:158–159. https://doi.org/10.1002/med.21599
Dinarello CA (2017) Overview of the IL-1 family in innate inflammation and acquired immunity. Immunol Rev 281:8–27. https://doi.org/10.1111/imr.12621
El-Rokh AR, Negm A, El-Shamy M, El-Gindy M, Abdel-Mogib M (2018) Sucrose diester of aryldihydronaphthalene-type lignans from Echium angustifolium Mill. and their antitumor activity. Phytochemistry 149:155–160. https://doi.org/10.1016/j.phytochem.2018.02.014
Erosa-Rejon G, Pena-Rodriguez LM, Sterner O (2009) Secondary metabolites from Heliotropium angiospermum. J Mex Chem Soc 53:44–47. https://doi.org/10.29356/jmcs.v53i2.1004
Espírito-Santo RF, Meira CS, Costa RS, Souza Filho OP, Evangelista AF, Trossini GHG, Ferreira GM, Veelozo ES, Villarreal CF, Soares MBP (2017) The anti-inflammatory and immunomodulatory potential of braylin: pharmacological properties and mechanisms by in silico, in vitro and in vivo approaches. Plos One 12:e0179174. https://doi.org/10.1371/journal.pone.0179174.g003
Fayed MAA (2021) Heliotropium; a genus rich in pyrrolizidine alkaloids: a systematic review following its phytochemistry and pharmacology. Phytomedicine Plus 1:100036. https://doi.org/10.1016/j.phyplu.2021.100036
Fujiwara N, Kobayashi K (2005) vMacrophages in inflammation. Curr. Drug Target-Inflamm Allergy 4:281–286. https://doi.org/10.2174/1568010054022024
Giulietti AM, Pirani JR, Harley RM (1997) Espinhaço range region, eastern Brazil. In: Davis SD, Heywood VH, Herrera MacBryde O, Villa-Lobos J, Hamilton AC (eds) Centres of plant diversity: a guide and strategy for their conservation. Information Press, Oxford
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Anal Biochem 126:131–138. https://doi.org/10.1016/0003-2697(82)90118-X
Hernández-Bolio GI, Ruiz-Vargas JA, Peña-Rodríguez LM (2019) Natural products from the Yucatecan Flora: structural diversity and biological activity. J Nat Prod 82:647–656. https://doi.org/10.1021/acs.jnatprod.8b00959
Hosseini N, Abolhassani M (2011) Immunomodulatory properties of borage (Echium amoenum) on BALB/c mice infected with Leishmania major. J Clin Immunol 31:465–471. https://doi.org/10.1007/s10875-010-9502-6
Kobayashi W, Miyase T, Suzuki S, Noguchi H, Chen XM (2000) Oligosaccharide esters from the roots of Polygala arillata. J Nat Prod 63:1066–1069. https://doi.org/10.1021/np0000567
Kozachok S, Pecio Ł, Kolodziejczyk-Czepas J, Marchyshyn S, Nowak P, Mołdoch J, Oleszek W (2018) γ-Pyrone compounds: flavonoids and maltol glucoside derivatives from Herniaria glabra L. collected in the Ternopil region of the Ukraine. Phytochemistry 152:213–222. https://doi.org/10.1016/j.phytochem.2018.05.009
Krzyżanowska-Kowalczyk J, Pecio Ł, Mołdoch J, Ludwiczuk A, Kowalczyk M (2018) Novel phenolic constituents of Pulmonaria officinalis L. LC-MS/MS comparison of spring and autumn metabolite profiles. Molecules 23:2277. https://doi.org/10.3390/molecules23092277
Melo JIM, Semir J (2008) Taxonomia do gênero Heliotropium L. (Heliotropiaceae) no Brasil. Acta Bot Bras 22:754–770. https://doi.org/10.1590/S0102-33062008000300013
Opretzkaa LCF, Espírito-Santob RF, Nascimento OA, Abreu LS, Alves IM, Döring E, Soares MBP, Velozo ES, Laufer SA, Villarreal CF (2019) Natural chromones as potential anti-inflammatory agents: pharmacological properties and related mechanisms. Int Immunopharmacol 72:31–39. https://doi.org/10.1016/j.intimp.2019.03.044
Otsuka H, Kuwabara H, Hoshiyama H (2008) Identification of sucrose diesters of aryldihydronaphthalene-type lignans from Trigonotis peduncularisand the nature of their fluorescence. J Nat Prod 71:1178–1181. https://doi.org/10.1021/np800071r
Suo MR, Yang JS, Liu QH (2006) Lignan oligosaccharide esters from Eritrichium rupestre. J Nat Prod 69:682–684. https://doi.org/10.1021/np050442u
Vasconcelos MF (2011) O que são campos rupestres e campos de altitude nos topos de montanha do leste do Brasil? Braz J Bot 34:241–246. https://doi.org/10.1590/S0100-84042011000200012
Watanabe S, Alexander M, Misharin AV, Budinger GRS (2019) The role of macrophages in the resolution of inflammation. J Clin Invest 129:2619–2628. https://doi.org/10.1172/JCI124615
Wu ST, Wang YX, Yu BH, Ma CL, Qiu HQ, Wang GS (2022) Phenylpropanoids from Brachybotrys paridiformis Maxim. ex Oliv. and their anti-HBV activities. Phytochemistry 197:113114. https://doi.org/10.1016/j.phytochem.2022.113114
Zhou Y, ** M, ** C, Ye C, Wang J, Sun J, Wei CX, Zhou W, Li G (2019) A new aryldihydronaphthalene-type lignan and other metabolites with potential anti-inflammatory activities from Corispermum mongolicum Il**. Nat Prod Res 34:225–232. https://doi.org/10.1080/14786419.2018.1527835
Funding
This work was supported by Instituto Nacional de Ciência e Tecnologia, Rede Norte Nordeste de Fitoprodutos/Conselho Nacional de Desenvolvimento Científico e Tecnológico (INCT-RENNOFITO/CNPQ).
Author information
Authors and Affiliations
Contributions
GLDS was responsible for collecting the plant material, performing the laboratory work, interpreting the experimental data, and writing the manuscript first draft. JPRS, DIAF, YMN, and TAS assisted in HPLC method development, MS, and NMR analysis. CFV and LCFO carried out biological activity analysis. JIMM collected and identified the plant material. LSA and FMSJ performed ECD analyses and assisted in data interpretation. JFT, IMF, and MSS critically revised the paper and coordinated the project. All authors read the final manuscript and approved the submission.
Corresponding author
Supplementary Information
ESM 1
(PDF 3.31 MB)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
dos Santos, G.L.D., de Araújo, D.I.A.F., Raimundo e Silva, J.P. et al. Sucrose Diester of Aryldihydronaphthalene-Type Lignan with Anti-inflammatory Activity from Heliotropium angiospermum. Rev. Bras. Farmacogn. 32, 734–740 (2022). https://doi.org/10.1007/s43450-022-00295-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43450-022-00295-1