Abstract
Background
The nature in which health care insurance companies have interacted with drug manufacturers has evolved over the last 50 years, demanding shifts in how the pharmaceutical industry responds. The purpose of this article is to highlight how medical groups within Pharma Collaboration for Transparent Medical Information (phactMI™) member companies respond to requests from health care decision makers (HCDMs) and to understand the similarities and differences among drug manufacturers (DMs) in this evolving climate of information exchange.
Methods
As health care insurance companies transition towards a managed care model and began assessments of both the clinical and economical aspects of drug products, DMs began to respond to requests from HCDMs. An anonymous 16-question survey was conducted to evaluate how 27 Medical Information departments (MIDs) respond to payer requests for information, and to identify payer needs in order to provide a better overall customer experience.
Results
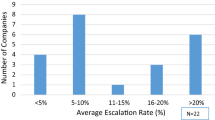
The results from this survey provided insight into the overall management of payer unsolicited medical requests (PUMRs) and focused on a few different areas, including how requests are received, what materials are requested and which are sent, online availability, and availability of materials for products in the pipeline.
Conclusion
Further discussion is recommended to ensure a focused approach in develo** tools and identifying appropriate resources are available to address inquiries from HCDMs.


Similar content being viewed by others
References
D’Ascoli A, Gazo A, Yueh S. A phactMITM Benchmarking Survey: 27 Medical Information Departments on Healthcare Decision Making Materials for Payer Inquiries [poster]. Drug Information Association Medical Affairs and Scientific Communications Forum; May 18–20, 2019, Orlando FL.
Gazo A. PhactMI Benchmarking Survey Highlights: How are the Twenty Seven Member Companies Executing on Medical Information Initiatives [oral presentation]. Drug Information Association Annual Meeting; June 23, 2019; San Diego CA.
Carson CB. The Effect of Managed Care on the Pharmaceutical Industry. Digital Access to Scholarship at Harvard. https://nrs.harvard.edu/urn-3:HUL.InstRepos:896LenciD. 5565. Published1997. Accessed 8 Sept 2019.
Perfetto EM, Burke L, Oehrlein EM, et al. FDAMA Section 114: why the renewed interest? J Manag Care Spec Pharm. 2015;21(5):368–74. https://doi.org/10.18553/jmcp.2015.21.5.368.
Solga C, Smith D. Pre-Approval Communication of Health Care Economic Information to U.S. Payers. The Evidence Forum. 2018. https://www.evidera.com/wp-content/uploads/2018/05/Pre-Approval-Communication-of-Health-Care.pdf. Accessed 8 Sept 2019.
Neumann PJ. Evidence-based and value-based formulary guidelines. Health Aff (Millwood). 2004;23(1):124–34. https://doi.org/10.1377/hlthaff.23.1.124.
Academy of Managed Care Pharmacy. The AMCP Format for Formulary Submissions. Version 4.1; Alexandria, VA: Academy of Managed Care Pharmacy; 2019.
Niyazov A, Lenci D. Communicating healthcare economic and pre-approval information with healthcare decision- makers: opportunities following the 21st century cures act and FDA guidance. Front Pub Health. 2018;6(304):1–5.
Food and Drug Administration. Drug and Device Manufacturer Communications With Payors, Formulary Committees, and Similar Entities—Questions and Answers Guidance for Industry and Review Staff. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/drug-and-device-manufacturer-communications-payors-formulary-committees-and-similar-entities; 2018. Accessed 8 Sept 2019.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Gazo reports other from Sanofi, outside the submitted work. The other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
D’Ascoli, A., Gazo, A., Yueh, S. et al. phactMI™ Benchmarking Survey: Analysis of 27 Medical Information Departments Responses to Inquiries from Health Care Decision Makers. Ther Innov Regul Sci 54, 1291–1295 (2020). https://doi.org/10.1007/s43441-020-00148-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43441-020-00148-w