Abstract
Background
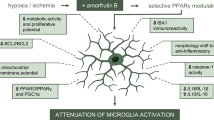
Microglia play a major role in the development of brain inflammation after central nervous system injury. On the other hand, microglia also participate in the repair process. The dualistic role of these cells results from the fact that various states of their activation are associated with specific phenotypes. The M1 phenotype is responsible for the production of proinflammatory mediators, whereas the M2 microglia release anti-inflammatory and trophic factors and take part in immunosuppressive and neuroprotective processes. The histone deacetylase inhibitor sodium butyrate (SB) shows anti-inflammatory and neuroprotective effects in some animal models of brain injury. The aim of this study was to examine the effects of sodium butyrate on the proliferation and M1/M2 polarization of primary microglial cells after oxygen and glucose deprivation (OGD) in vitro.
Methods
Primary microglial cultures were prepared from 1-day-old rats, subjected to the OGD procedure and treated with SB (0.1 mM, 1 mM and 10 mM). The effect of OGD and SB on microglial proliferation was assessed by double immunofluorescence, and microglial phenotypes were evaluated by qPCR.
Results
The OGD procedure stimulated the proliferation of microglia after 24 h of culturing, and SB treatment reduced the division of these cells. This effect was inversely proportional to the SB concentration. The OGD procedure increased proinflammatory CD86 and IL1β gene expression and reduced the expression of the anti-inflammatory M2 markers arginase and CD200 in microglia.
Conclusions
SB can change the polarization of microglia after OGD from an unfavourable M1 to a beneficial M2 phenotype. Our results show that SB is a potential immunosuppressive agent that can modulate microglial activation stimulated by ischaemic-like conditions.



Similar content being viewed by others
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- CNS:
-
Central nervous system
- DIV:
-
Day(s) in vitro
- GDNF:
-
Glial cell line-derived neurotrophic factor
- HATs:
-
Histone acetyl-transferases
- HDACs:
-
Histone deacetylases
- HDACis:
-
Histone deacetylase inhibitors
- HI:
-
Hypoxia–ischaemia
- IGF:
-
Insulin growth factor
- IL-10:
-
Interleukin 10
- IL-1β:
-
Interleukin 1β
- LPS:
-
Lipopolysaccharide
- MMPs:
-
Matrix metalloproteinases
- NO:
-
Nitric oxide,
- NOS:
-
Nitric oxide synthase
- OGD:
-
Oxygen and glucose deprivation
- PBS:
-
Phosphate-buffered saline buffer
- PFA:
-
Paraformaldehyde
- PLL:
-
Poly-L-lysine
- ROS:
-
Reactive oxygen species
- SAHA:
-
Vorinostat
- SB:
-
Sodium butyrate
- TGF β:
-
Transforming growth factor β
- TSA:
-
Trichostatin A
- VPA:
-
Valproic acid
References
Alliot F, Godin I, Pessac B. Microglia derive from progenitors, originating from the yolk sac, and which proliferate in the brain. Brain Res Dev Brain Res. 1999;117:145–52. https://doi.org/10.1016/s0165-3806(99)00113-3.
Winerdal M, Winerdal ME, Kinn J, Urmaliya V, Winqvist O, Adén U. Long lasting local and systemic inflammation after cerebral hypoxic ischemia in newborn mice. PLoS ONE. 2012;7: e36422. https://doi.org/10.1371/journal.pone.0036422.
Algra SO, Groeneveld KM, Schadenberg AW, Haas F, Evens FC, Meerding J, et al. Cerebral ischemia initiates an immediate innate immune response in neonates during cardiac surgery. J Neuroinflamm. 2013;10:796. https://doi.org/10.1186/1742-2094-10-24.
Bona E, Andersson A-L, Blomgren K, Gilland E, Puka-Sundvall M, Gustafson K, et al. Chemokine and inflammatory cell response to hypoxia-ischemia in immature rats. Pediatr Res. 1999;45:500–9. https://doi.org/10.1203/00006450-199904010-00008.
** Y, Silverman AJ, Vannucci SJ. Mast cells are early responders after hypoxia-ischemia in immature rat brain. Stroke. 2009;40:3107–12. https://doi.org/10.1161/STROKEAHA.109.549691.
Hagberg H, Gressens P, Mallard C. Inflammation during fetal and neonatal life: implications for neurologic and neuropsychiatric disease in children and adults. Ann Neurol. 2012;71:444–57. https://doi.org/10.1002/ana.22620.
Hagberg H, Mallard C, Ferriero DM, Vannucci SJ, Levison SW, Vexler ZS, et al. The role of inflammation in perinatal brain injury. Nat Rev Neurol. 2015;11:192–208. https://doi.org/10.1038/nrneurol.2015.13.
Faustino JV, Wang X, Johnson CE, Klibanov A, Derugin N, Wendland MF, et al. Microglial cells contribute to endogenous brain defenses after acute neonatal focal stroke. J Neurosci. 2011;31:12992–3001. https://doi.org/10.1523/JNEUROSCI.2102-11.2011.
Hu X, Li P, Guo Y, Wang H, Leak RK, Chen S, et al. Microglia/macrophage polarization dynamics reveal novel mechanism of injury expansion after focal cerebral ischemia. Stroke. 2012;43:3063–70. https://doi.org/10.1161/STROKEAHA.112.659656.
Hu X, Leak RK, Shi Y, Suenaga J, Gao Y, Zheng P, et al. Microglial and macrophage polarization—new prospects for brain repair. Nat Rev Neurol. 2015;11:56–64. https://doi.org/10.1038/nrneurol.2014.207.
Lalancette-Hébert M, Gowing G, Simard A, Weng YC, Kriz J. Selective ablation of proliferating microglial cells exacerbates ischemic injury in the brain. J Neurosci. 2007;27:2596–605. https://doi.org/10.1523/JNEUROSCI.5360-06.2007.
Capone C, Frigerio S, Fumagalli S, Gelati M, Principato M-C, Storini C, et al. Neurosphere-derived cells exert a neuroprotective action by changing the ischemic microenvironment. PLoS ONE. 2007;2: e373. https://doi.org/10.1371/journal.pone.0000373.
Colton CA. Heterogeneity of microglial activation in the innate immune response in the brain. J Neuroimmune Pharmacol. 2009;4:399–418. https://doi.org/10.1007/s11481-009-9164-4.
Lehrmann H, Pritchard LL, Harel-Bellan A. Histone acetyltransferases and deacetylases in the control of cell proliferation and differentiation. Adv Cancer Res. 2002;86:41–65. https://doi.org/10.1016/S0065-230X(02)86002-X.
Mai A, Massa S, Rotili D, Cerbara I, Valente S, Pezzi R, et al. Histone deacetylation in epigenetics: an attractive target for anticancer therapy. Med Res Rev. 2005;25:261–309. https://doi.org/10.1002/med.20024.
Glozak MA, Sengupta N, Zhang X, Seto E. Acetylation and deacetylation of non-histone proteins. Gene. 2005;363:15–23. https://doi.org/10.1016/j.gene.2005.09.010.
Kim HJ, Rowe M, Ren M, Hong J-S, Chen P-S, Chuang D-M. Histone deacetylase inhibitors exhibit anti-inflammatory and neuroprotective effects in a rat permanent ischemic model of stroke: multiple mechanisms of action. J Pharmacol Exp Ther. 2007;321:892–901. https://doi.org/10.1124/jpet.107.120188.
Jaworska J, Ziemka-Nalecz M, Sypecka J, Zalewska T. The potential neuroprotective role of a histone deacetylase inhibitor, sodium butyrate, after neonatal hypoxia-ischemia. J Neuroinflamm. 2017;14:34. https://doi.org/10.1186/s12974-017-0807-8.
Jaworska J, Zalewska T, Sypecka J, Ziemka-Nalecz M. Effect of the HDAC inhibitor, sodium butyrate, on neurogenesis in a rat model of neonatal hypoxia-ischemia: potential mechanism of action. Mol Neurobiol. 2019;56:6341–70. https://doi.org/10.1007/s12035-019-1518-1.
Liu F, Mccullough LD. Inflammatory responses in hypoxic ischemic encephalopathy. Acta Pharmacol Sin. 2013;34:1121–30. https://doi.org/10.1038/aps.2013.89.
Ramlackhansingh AF, Brooks DJ, Greenwood RJ, Bose SK, Turkheimer FE, Kinnunen KM, et al. Inflammation after trauma: microglial activation and traumatic brain injury. Ann Neurol. 2011;70:374–83. https://doi.org/10.1002/ana.22455.
Li T, Pang S, Yu Y, Wu X, Guo J, Zhang S. Proliferation of parenchymal microglia is the main source of microgliosis after ischaemic stroke. Brain. 2013;136:3578–88. https://doi.org/10.1093/brain/awt287.
Li H, Zhang Q, Li N, Wang F, **ang H, Zhang Z, et al. Plasma levels of Th17-related cytokines and complement C3 correlated with aggressive behavior in patients with schizophrenia. Psychiatry Res. 2016;246:700–6. https://doi.org/10.1016/j.psychres.2016.10.061.
Huang Y, Xu Z, **ong S, Sun F, Qin G, Hu G, et al. Repopulated microglia are solely derived from the proliferation of residual microglia after acute depletion. Nat Neurosci. 2018;21:530–40. https://doi.org/10.1038/s41593-018-0090-8.
Zhang G, Li T, Chang X, **ng J. Long noncoding RNA SNHG14 promotes ischemic brain injury via regulating miR-199b/AQP4 Axis. Neurochem Res. 2021;46:1280–90. https://doi.org/10.1007/s11064-021-03265-6.
Yao Z, Liu N, Zhu X, Wang L, Zhao Y, Liu Q, et al. Subanesthetic isoflurane abates ROS-activated MAPK/NF-κB signaling to repress ischemia-induced microglia inflammation and brain injury. Aging (Albany NY). 2020;12:26121–39. https://doi.org/10.18632/aging.202349.
Liu J, Ma W, Zang C-H, Wang G-D, Zhang S-J, Wu H-J, et al. Salidroside inhibits NLRP3 inflammasome activation and apoptosis in microglia induced by cerebral ischemia/reperfusion injury by inhibiting the TLR4/NF-κB signaling pathway. Ann Transl Med. 2021;9:1694–1694. https://doi.org/10.21037/atm-21-5752.
Zhang Q, Chen C, Lü J, **e M, Pan D, Luo X, et al. Cell cycle inhibition attenuates microglial proliferation and production of IL-1β, MIP-1α, and NO after focal cerebral ischemia in the rat. Glia. 2009;57:908–20. https://doi.org/10.1002/glia.20816.
Chen PS, Wang C-C, Bortner CD, Peng G-S, Wu X, Pang H, et al. Valproic acid and other histone deacetylase inhibitors induce microglial apoptosis and attenuate lipopolysaccharide-induced dopaminergic neurotoxicity. Neuroscience. 2007;149:203–12. https://doi.org/10.1016/j.neuroscience.2007.06.053.
He X-B, Wu Y, Huang H, Guo F. A novel histone deacetylase inhibitor-based approach to eliminate microglia and retain astrocyte properties in glial cell culture. J Neurochem. 2022;161:405–16. https://doi.org/10.1111/jnc.15581.
Zhang H, Zhao W. Resveratrol alleviates ischemic brain injury by inhibiting the activation of pro-inflammatory microglia via the CD147/MMP-9 pathway. J Stroke Cerebrovasc Dis. 2022;31: 106307. https://doi.org/10.1016/j.jstrokecerebrovasdis.2022.106307.
Kim HJ, Leeds P, Chuang D-M. The HDAC inhibitor, sodium butyrate, stimulates neurogenesis in the ischemic brain. J Neurochem. 2009;110:1226–40. https://doi.org/10.1111/j.1471-4159.2009.06212.x.
Hsing C-H, Hung S-K, Chen Y-C, Wei T-S, Sun D-P, Wang J-J, et al. Histone deacetylase inhibitor trichostatin A ameliorated endotoxin-induced neuroinflammation and cognitive dysfunction. Mediat Inflamm. 2015;2015: 163140. https://doi.org/10.1155/2015/163140.
Durham BS, Grigg R, Wood IC. Inhibition of histone deacetylase 1 or 2 reduces induced cytokine expression in microglia through a protein synthesis independent mechanism. J Neurochem. 2017;143:214–24. https://doi.org/10.1111/jnc.14144.
Jiao F-Z, Wang Y, Zhang H-Y, Zhang W, Wang L-W, Gong Z-J. Histone deacetylase 2 Inhibitor CAY10683 alleviates lipopolysaccharide induced neuroinflammation through attenuating TLR4/NF-κB signaling pathway. Neurochem Res. 2018;43:1161–70. https://doi.org/10.1007/s11064-018-2532-9.
Mota M, Porrini V, Parrella E, Benarese M, Bellucci A, Rhein S, et al. Neuroprotective epi-drugs quench the inflammatory response and microglial/macrophage activation in a mouse model of permanent brain ischemia. J Neuroinflamm. 2020;17:361. https://doi.org/10.1186/s12974-020-02028-4.
Meng Q, Yang G, Yang Y, Ding F, Hu F. Protective effects of histone deacetylase inhibition by Scriptaid on brain injury in neonatal rat models of cerebral ischemia and hypoxia. Int J Clin Exp Pathol. 2020;13:179–91.
Chen S, Ye J, Chen X, Shi J, Wu W, Lin W, et al. Valproic acid attenuates traumatic spinal cord injury-induced inflammation via STAT1 and NF-κB pathway dependent of HDAC3. J Neuroinflamm. 2018;15:150. https://doi.org/10.1186/s12974-018-1193-6.
Wang G, Shi Y, Jiang X, Leak RK, Hu X, Wu Y, et al. HDAC inhibition prevents white matter injury by modulating microglia/macrophage polarization through the GSK3β/PTEN/Akt axis. Proc Natl Acad Sci USA. 2015;112:2853–8. https://doi.org/10.1073/pnas.1501441112.
Acknowledgements
The confocal analysis of immunocytochemical staining was performed in the Laboratory of Advanced Microscopy Techniques, Mossakowski Medical Research Institute, Polish Academy of Sciences.
Funding
This work was supported by National Science Centre, Poland grant no 2017/27/B/NZ3/00582. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
KZ performed and analysed the data, prepared figures, and contributed to writing the manuscript, JG performed some in vitro experiments, JS contributed to writing the manuscript, MZ-N designed experiments, analysed and interpreted the data, and was a major contributor in writing the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ziabska, K., Gargas, J., Sypecka, J. et al. The impact of the histone deacetylase inhibitor sodium butyrate on microglial polarization after oxygen and glucose deprivation. Pharmacol. Rep 74, 909–919 (2022). https://doi.org/10.1007/s43440-022-00384-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43440-022-00384-x