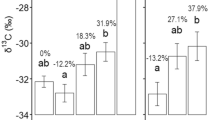
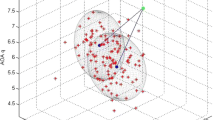
Abstract
-
Refined conversion factors for soil fungal biomarkers are proposed.
-
High interspecific variability is present in all fungal biomarkers.
-
A modeling approach supports the validity of biomarker estimates in diverse soils.
-
ITS1 copies vary strongly, but are fungal-specific with least phylogenetic bias.
-
A combination of fungal biomarkers will reveal soil fungal physiology and activity.
The abundances of fungi and bacteria in soil are used as simple predictors for carbon dynamics, and represent widely available microbial traits. Soil biomarkers serve as quantitative estimates of these microbial groups, though not quantifying microbial biomass per se. The accurate conversion to microbial carbon pools, and an understanding of its comparability among soils is therefore needed. We refined conversion factors for classical fungal biomarkers, and evaluated the application of quantitative PCR (qPCR, rDNA copies) as a biomarker for soil fungi. Based on biomarker contents in pure fungal cultures of 30 isolates tested here, combined with comparable published datasets, we propose average conversion factors of 95.3 g fungal C g−1 ergosterol, 32.0 mg fungal C µmol−1 PLFA 18:2ω6,9 and 0.264 pg fungal C ITS1 DNA copy−1. As expected, interspecific variability was most pronounced in rDNA copies, though qPCR results showed the least phylogenetic bias. A modeling approach based on exemplary agricultural soils further supported the hypothesis that high diversity in soil buffers against biomarker variability, whereas also phylogenetic biases impact the accuracy of comparisons in biomarker estimates. Our analyses suggest that qPCR results cover the fungal community in soil best, though with a variability only partly offset in highly diverse soils. PLFA 18:2ω6,9 and ergosterol represent accurate biomarkers to quantify Ascomycota and Basidiomycota. To conclude, the ecological interpretation and coverage of biomarker data prior to their application in global models is important, where the combination of different biomarkers may be most insightful.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Andrade-Linares, D.R., Veresoglou, S.D., Rillig, M.C., 2016. Temperature priming and memory in soil filamentous fungi. Fungal Ecology 21, 10–15.
Anthony, M.A., Bender, S.F., van der Heijden, M.G.A., 2023. Enumerating soil biodiversity. Proceedings of the National Academy of Sciences of the United States of America 120, e2304663120.
Antibus, R.K., Sinsabaugh, R.L., 1993. The extraction and quantification of ergosterol from ectomycorrhizal fungi and roots. Mycorrhiza 3, 137–144.
Baldrian, P., Větrovský, T., Cajthaml, T., Dobiášová, P., Petránková, M., Šnajdr, J., Eichlerová, I., 2013. Estimation of fungal biomass in forest litter and soil. Fungal Ecology 6, 1–11.
Bar-On, Y.M., Phillips, R., Milo, R., 2018. The biomass distribution on Earth. Proceedings of the National Academy of Sciences of the United States of America 115, 6506–6511.
Barajas-Aceves, M., Hassan, M., Tinoco, R., Vazquez-Duhalt, R., 2002. Effect of pollutants on the ergosterol content as indicator of fungal biomass. Journal of Microbiological Methods 50, 227–236.
Brondz, I., Høiland, K., Ekeberg, D., 2004. Multivariate analysis of fatty acids in spores of higher basidiomycetes: a new method for chemotaxonomical classification of fungi. Journal of Chromatography B 800, 303–307.
Camenzind, T., Mason-Jones, K., Mansour, I., Rillig, M.C., Lehmann, J., 2023. Formation of necromass-derived soil organic carbon determined by microbial death pathways. Nature Geoscience 16, 115–122.
Camenzind, T., Philipp Grenz, K., Lehmann, J., Rillig, M.C., 2021. Soil fungal mycelia have unexpectedly flexible stoichiometric C:N and C:P ratios. Ecology Letters 24, 208–218.
Camenzind, T., Weimershaus, P., Lehmann, A., Aguilar-Trigueros, C., Rillig, M.C., 2022. Soil fungi invest into asexual sporulation under resource scarcity, but trait spaces of individual isolates are unique. Environmental Microbiology 24, 2962–2978.
Canarini, A., Fuchslueger, L., Schnecker, J., Metze, D., Nelson, D. B., Kahmen, A., Watzka, M., Pötsch, E.M., Schaumberger, A., Bahn, M., Richter, A., 2023. Soil fungi remain active and invest in storage compounds during drought independent of future climate conditions. bioRxiv, DOI: https://doi.org/10.1101/2023.10.23.563577.
Charcosset, J.Y., Chauvet, E., 2001. Effect of culture conditions on ergosterol as an indicator of biomass in the aquatic hyphomycetes. Applied and Environmental Microbiology 67, 2051–2055.
Chen, C., Chen, X.L., Chen, H.Y.H., 2023. Map** N deposition impacts on soil microbial biomass across global terrestrial ecosystems. Geoderma 433, 116429.
Crowther, T.W., van den Hoogen, J., Wan, J., Mayes, M.A., Keiser, A.D., Mo, L., Averill, C., Maynard, D.S., 2019. The global soil community and its influence on biogeochemistry. Science 365, eaav0550.
Delmont, T.O., Prestat, E., Keegan, K.P., Faubladier, M., Robe, P., Clark, I.M., Pelletier, E., Hirsch, P.R., Meyer, F., Gilbert, J.A., Le Paslier, D., Simonet, P., Vogel, T.M., 2012. Structure, fluctuation and magnitude of a natural grassland soil metagenome. The ISME Journal 6, 1677–1687.
Djajakirana, G., Joergensen, R.G., Meyer, B., 1996. Ergosterol and microbial biomass relationship in soil. Biology and Fertility of Soils 22, 299–304.
Domsch, K.H., Gams, W., Anderson, T.H., 2007. Compendium of Soil Fungi. 2nd ed. Eching: IHW-Verlag.
Ekblad, A., Mikusinska, A., Agren, G.I., Menichetti, L., Wallander, H., Vilgalys, R., Bahr, A., Eriksson, U., 2016. Production and turnover of ectomycorrhizal extramatrical mycelial biomass and necromass under elevated CO2 and nitrogen fertilization. New Phytologist 211, 874–885.
Federle, T.W., 1986. Microbial Distribution in Soil - New Techniques. In: Megusar, F., Gantar, M., eds. Perspectives in Microbial Ecology. Ljulbljana: Slovene Society for Microbiology, 493–498.
Fierer, N., Jackson, J.A., Vilgalys, R., Jackson, R.B., 2005. Assessment of soil microbial community structure by use of taxon-specific quantitative PCR assays. Applied and Environmental Microbiology 71, 4117–4120.
Frostegård, Å., Bååth, E., 1996. The use of phospholipid fatty acid analysis to estimate bacterial and fungal biomass in soil. Biology and Fertility of Soils 22, 59–65.
Frostegård, Å., Tunlid, A., Bååth, E., 1991. Microbial biomass measured as total lipid phosphate in soils of different organic content. Journal of Microbiological Methods 14, 151–163.
Frostegård, A., Tunlid, A., Bååth, E., 1993. Phospholipid fatty-acid composition, biomass and activity of microbial communities from two soil types experimentally exposed to different heavy-metals. Applied and Environmental Microbiology 59, 3605–3617.
Frostegård, A., Tunlid, A., Bååth, E., 2011. Use and misuse of PLFA measurements in soils. Soil Biology & Biochemistry 43, 1621–1625.
Gorka, S., Darcy, S., Horak, J., Imai, B., Mohrlok, M., Salas, E., Richter, A., Schmidt, H., Wanek, W., Kaiser, C., Canarini, A., 2023. Beyond PLFA: concurrent extraction of neutral and glycolipid fatty acids provides new insights into soil microbial communities. Soil Biology and Biochemistry 187, 109205.
Green, C.T., Scow, K.M., 2000. Analysis of phospholipid fatty acids (PLFA) to characterize microbial communities in aquifers. Hydrogeology Journal 8, 126–141.
Grimmett, I.J., Shipp, K.N., Macneil, A., Bärlocher, F., 2013. Does the growth rate hypothesis apply to aquatic hyphomycetes? Fungal Ecology 6, 493–500.
He, L.Y., Lipson, D.A., Mazza Rodrigues, J.L., Mayes, M., Björk, R. G., Glaser, B., Thornton, P., Xu, X.F., 2021. Dynamics of fungal and bacterial biomass carbon in natural ecosystems: site-level applications of the CLM-microbe model. Journal of Advances in Modeling Earth Systems 13, e2020MS002283.
Heaton, L.L.M., Jones, N.S., Fricker, M.D., 2016. Energetic constraints on fungal growth. The American Naturalist 187, E27–E40.
Hsieh, C.W.C., Cannella, D., Jørgensen, H., Felby, C., Thygesen, L. G., 2014. Cellulase inhibition by high concentrations of monosaccharides. Journal of Agricultural and Food Chemistry 62, 3800–3805.
Hungate, B.A., Mau, R.L., Schwartz, E., Caporaso, J.G., Dijkstra, P., van Gestel, N., Koch, B.J., Liu, C.M., McHugh, T.A., Marks, J.C., Morrissey, E.M., Price, L.B., 2015. Quantitative microbial ecology through stable isotope probing. Applied and Environmental Microbiology 81, 7570–7581.
Joergensen, R.G., 2018. Amino sugars as specific indices for fungal and bacterial residues in soil. Biology and Fertility of Soils 54, 559–568.
Joergensen, R.G., 2022. Phospholipid fatty acids in soil—drawbacks and future prospects. Biology and Fertility of Soils 58, 1–6.
Joergensen, R.G., Emmerling, C., 2006. Methods for evaluating human impact on soil microorganisms based on their activity, biomass, and diversity in agricultural soils. Journal of Plant Nutrition and Soil Science 169, 295–309.
Joergensen, R.G., Wichern, F., 2008. Quantitative assessment of the fungal contribution to microbial tissue in soil. Soil Biology and Biochemistry 40, 2977–2991.
Junicke, H., Abbas, B., Oentoro, J., van Loosdrecht, M., Kleerebezem, R., 2014. Absolute quantification of individual biomass concentrations in a methanogenic coculture. AMB Express 4, 35.
Keck, F., Rimet, F., Bouchez, A., Franc, A., 2016. phylosignal: an R package to measure, test, and explore the phylogenetic signal. Ecology and Evolution 6, 2774–2780.
Klamer, M., Bååth, E., 2004. Estimation of conversion factors for fungal biomass determination in compost using ergosterol and PLFA 18:2ω6,9. Soil Biology and Biochemistry 36, 57–65.
Klein, D.A., Paschke, M.W., 2004. Filamentous fungi: the indeterminate lifestyle and microbial ecology. Microbial Ecology 47, 224–235.
Kramer, S., Dibbern, D., Moll, J., Huenninghaus, M., Koller, R., Krueger, D., Marhan, S., Urich, T., Wubet, T., Bonkowski, M., Buscot, F., Lueders, T., Kandeler, E., 2016. Resource partitioning between bacteria, fungi, and protists in the detritusphere of an agricultural soil. Frontiers in Microbiology 7, 1524.
Lavrinienko, A., Jernfors, T., Koskimäki, J.J., Pirttilä, A.M., Watts, P. C., 2021. Does intraspecific variation in rDNA copy number affect analysis of microbial communities? Trends in Microbiology 29, 19–27.
Leckie, S.E., Prescott, C.E., Grayston, S.J., Neufeld, J.D., Mohn, W. W., 2004. Comparison of chloroform fumigation-extraction, phospholipid fatty acid, and DNA methods to determine microbial biomass in forest humus. Soil Biology and Biochemistry 36, 529–532.
Lehmann, A., Zheng, W.S., Soutschek, K., Roy, J., Yurkov, A.M., Rillig, M.C., 2019. Tradeoffs in hyphal traits determine mycelium architecture in saprobic fungi. Scientific Reports 9, 14152.
Lewe, N., Hermans, S., Lear, G., Kelly, L.T., Thomson-Laing, G., Weisbrod, B., Wood, S.A., Keyzers, R.A., Deslippe, J.R., 2021. Phospholipid fatty acid (PLFA) analysis as a tool to estimate absolute abundances from compositional 16S rRNA bacterial metabarcoding data. Journal of Microbiological Methods 188, 106271.
Li, J., Wang, X., Wu, J.H., Sun, Y.X., Zhang, Y.Y., Zhao, Y.F., Huang, Z., Duan, W.H., 2023. Climate and geochemistry at different altitudes influence soil fungal community aggregation patterns in alpine grasslands. Science of the Total Environment 881, 163375.
Liang, C., Amelung, W., Lehmann, J., Kästner, M., 2019. Quantitative assessment of microbial necromass contribution to soil organic matter. Global Change Biology 25, 3578–3590.
Lofgren, L.A., Uehling, J.K., Branco, S., Bruns, T.D., Martin, F., Kennedy, P.G., 2019. Genome-based estimates of fungal rDNA copy number variation across phylogenetic scales and ecological lifestyles. Molecular Ecology 28, 721–730.
Malik, A.A., Chowdhury, S., Schlager, V., Oliver, A., Puissant, J., Vazquez, P.G.M., Jehmlich, N., von Bergen, M., Griffiths, R.I., Gleixner, G., 2016. Soil fungal: bacterial ratios are linked to altered carbon cycling. Frontiers in Microbiology 7, 1247.
Manerkar, M.A., Seena, S., Bärlocher, F., 2008. Q-RT-PCR for assessing archaea, bacteria, and fungi during leaf decomposition in a stream. Microbial Ecology 56, 467–473.
Mason-Jones, K., Breidenbach, A., Dyckmans, J., Banfield, C.C., Dippold, M.A., 2023. Intracellular carbon storage by microorganisms is an overlooked pathway of biomass growth. Nature Communications 14, 2240.
Miltner, A., Bombach, P., Schmidt-Brücken, B., Kästner, M., 2012. SOM genesis: microbial biomass as a significant source. Biogeochemistry 111, 41–55.
Moore, D., Robson, G., Trinci, A., 2021. 21st Century Guidebook to Fungi. 2nd ed. Cambridge: Cambridge University Press.
Mouginot, C., Kawamura, R., Matulich, K.L., Berlemont, R., Allison, S.D., Amend, A.S., Martiny, A.C., 2014. Elemental stoichiometry of Fungi and Bacteria strains from grassland leaf litter. Soil Biology and Biochemistry 76, 278–285.
Ngosong, C., Gabriel, E., Ruess, L., 2012. Use of the signature Fatty Acid 16:1ω5 as a tool to determine the distribution of arbuscular mycorrhizal fungi in soil. Journal of Lipids 2012, 236807.
Niemenmaa, O., Galkin, S., Hatakka, A., 2008. Ergosterol contents of some wood-rotting basidiomycete fungi grown in liquid and solid culture conditions. International Biodeterioration & Biodegradation 62, 125–134.
Nisha, A., Rastogi, N.K., Venkateswaran, G., 2011. Optimization of media components for enhanced arachidonic acid production by Mortierella alpina under submerged cultivation. Biotechnology and Bioprocess Engineering 16, 229–237.
Nuccio, E.E., Blazewicz, S.J., Lafler, M., Campbell, A.N., Kakouridis, A., Kimbrel, J.A., Wollard, J., Vyshenska, D., Riley, R., Tomatsu, A., Hestrin, R., Malmstrom, R.R., Firestone, M., Pett-Ridge, J., 2022. HT-SIP: a semi-automated stable isotope probing pipeline identifies cross-kingdom interactions in the hyphosphere of arbuscular mycorrhizal fungi. Microbiome 10, 199.
Nurika, I., Eastwood, D.C., Barker, G.C., 2018. A comparison of ergosterol and PLFA methods for monitoring the growth of ligninolytic fungi during wheat straw solid state cultivation. Journal of Microbiological Methods 148, 49–54.
Olsson, P.A., Johansen, A., 2000. Lipid and fatty acid composition of hyphae and spores of arbuscular mycorrhizal fungi at different growth stages. Mycological Research 104, 429–434.
Osburn, E.D., McBride, S.G., Kupper, J.V., Nelson, J.A., McNear, D. H., McCulley, R.L., Barrett, J.E., 2022. Accurate detection of soil microbial community responses to environmental change requires the use of multiple methods. Soil Biology and Biochemistry 169, 108685.
Parikh, S.J., James, B.R., 2012. Soil: the foundation of agriculture. Nature Education Knowledge 3, 2.
Pasanen, A.L., Yli-Pietila, K., Pasanen, P., Kalliokoski, P., Tarhanen, J., 1999. Ergosterol content in various fungal species and biocontaminated building materials. Applied and Environmental Microbiology 65, 138–142.
Pawłowska, J., Okrasińska, A., Kisło, K., Aleksandrzak-Piekarczyk, T., Szatraj, K., Dolatabadi, S., Muszewska, A., 2019. Carbon assimilation profiles of mucoralean fungi show their metabolic versatility. Scientific Reports 9, 11864.
Pérez-Guzmán, L., Phillips, L.A., Acevedo, M.A., Acosta-Martínez, V., 2021. Comparing biological methods for soil health assessments: EL-FAME, enzyme activities, and qPCR. Soil Science Society of America Journal 85, 636–653.
Pinheiro, J., Bates, D., DebRoy, S., Sarkar, D., R Core Team, 2021. nlme: linear and nonlinear mixed effects models. R package version 3.1–152, available at the website CRAN. R-project.
Pusztahelyi, T., Molnár, Z., Emri, T., Klement, É., Miskei, M., Kerékgyártó, J., Balla, J., Pócsi, I., 2006. Comparative studies of differential expression of chitinolytic enzymes encoded by chiA, chiB, chiC and nagA genes in Aspergillus nidulans. Folia Microbiologica 51, 547–554.
R Core Team, 2021. R: a language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing.
Ruess, L., Chamberlain, P.M., 2010. The fat that matters: Soil food web analysis using fatty acids and their carbon stable isotope signature. Soil Biology and Biochemistry 42, 1898–1910.
Ruess, L., Häggblom, M.M., García Zapata, E.J., Dighton, J., 2002. Fatty acids of fungi and nematodes—possible biomarkers in the soil food chain? Soil Biology and Biochemistry 34, 745–756.
Ruess, L., Schütz, K., Migge-Kleian, S., Häggblom, M.M., Kandeler, E., Scheu, S., 2007. Lipid composition of Collembola and their food resources in deciduous forest stands—Implications for feeding strategies. Soil Biology and Biochemistry 39, 1990–2000.
Sae-Tun, O., Maftukhah, R., Noller, C., Remlinger, V.I., Meyer-Laker, V., Sørensen, A.C.T., Sustic, D., Socianu, S.I., Bernardini, L.G., Mentler, A., Keiblinger, K.M., 2020. Comparison of commonly used extraction methods for ergosterol in soil samples. International Agrophysics 34, 425–432.
Schliep, K.P., 2011. phangorn: phylogenetic analysis in R. Bioinformatics 27, 592–593.
Song, Z.W., Vail, A., Sadowsky, M.J., Schilling, J.S., 2014. Quantitative PCR for measuring biomass of decomposer fungi in planta. Fungal Ecology 7, 39–46.
Stahl, P.D., Klug, M.J., 1996. Characterization and differentiation of filamentous fungi based on fatty acid composition. Applied and Environmental Microbiology 62, 4136–4146.
Stahl, P.D., Parkin, T.B., Eash, N.S., 1995. Sources of error in direct microscopic methods for estimation of fungal biomass in soil. Soil Biology and Biochemistry 27, 1091–1097.
Sterner, R.W., Elser, J.J., 2002. Ecological Stoichiometry: the Biology of Elements from Molecules to the Biosphere. Princeton: Princeton University Press.
Stigler, S.M., 1997. Regression towards the mean, historically considered. Statistical Methods in Medical Research 6, 103–114.
Strickland, M.S., Rousk, J., 2010. Considering fungal: bacterial dominance in soils - Methods, controls, and ecosystem implications. Soil Biology and Biochemistry 42, 1385–1395.
Taube, R., Fabian, J., Van den Wyngaert, S., Agha, R., Baschien, C., Gerphagnon, M., Kagami, M., Krüger, A., Premke, K., 2019. Potentials and limitations of quantification of fungi in freshwater environments based on PLFA profiles. Fungal Ecology 41, 256–268.
Tedersoo, L., Anslan, S., Bahram, M., Drenkhan, R., Pritsch, K., Buegger, F., Padari, A., Hagh-Doust, N., Mikryukov, V., Gohar, D., Amiri, R., Hiiesalu, I., Lutter, R., Rosenvald, R., Rähn, E., Adamson, K., Drenkhan, T., Tullus, H., Jürimaa, K., Sibul, I., Otsing, E., Põlme, S., Metslaid, M., Loit, K., Agan, A., Puusepp, R., Varik, I., Kõljalg, U., Abarenkov, K., 2020. Regional-scale in-depth analysis of soil fungal diversity reveals strong pH and plant species effects in northern Europe. Frontiers in Microbiology 11, 1953.
Tedersoo, L., Mikryukov, V., Zizka, A., Bahram, M., Hagh-Doust, N., Anslan, S., Prylutskyi, O., Delgado-Baquerizo, M., Maestre, F.T., Pärn, J., Öpik, M., Moora, M., Zobel, M., Espenberg, M., Mander, Ü., Khalid, A.N., Corrales, A., Agan, A., Vasco-Palacios, A.M., Saitta, A., Rinaldi, A.C., Verbeken, A., Sulistyo, B.P., Tamgnoue, B., Furneaux, B., Ritter, C.D., Nyamukondiwa, C., Sharp, C., Marín, C., Gohar, D., Klavina, D., Sharmah, D., Dai, D.Q., Nouhra, E., Biersma, E.M., Rähn, E., Cameron, E.K., De Crop, E., Otsing, E., Davydov, E.A., Albornoz, F.E., Brearley, F.Q., Buegger, F., Zahn, G., Bonito, G., Hiiesalu, I., Barrio, I.C., Heilmann-Clausen, J., Ankuda, J., Kupagme, J.Y., Maciá-Vicente, J. G., Fovo, J.D., Geml, J., Alatalo, J.M., Alvarez-Manjarrez, J., Põldmaa, K., Runnel, K., Adamson, K., Bråthen, K.A., Pritsch, K., Tchan, K.I., Armolaitis, K., Hyde, K.D., Newsham, K.K., Panksep, K., Lateef, A.A., Tiirmann, L., Hansson, L., Lamit, L.J., Saba, M., Tuomi, M., Gryzenhout, M., Bauters, M., Piepenbring, M., Wijayawardene, N., Yorou, N.S., Kurina, O., Mortimer, P.E., Meidl, P., Kohout, P., Nilsson, R.H., Puusepp, R., Drenkhan, R., Garibay-Orijel, R., Godoy, R., Alkahtani, S., Rahimlou, S., Dudov, S.V., Põlme, S., Ghosh, S., Mundra, S., Ahmed, T., Netherway, T., Henkel, T.W., Roslin, T., Nteziryayo, V., Fedosov, V.E., Onipchenko, V.G., Yasanthika, W.A.E., Lim, Y.W., Soudzilovskaia, N.A., Antonelli, A., Kõljalg, U., Abarenkov, K., 2022. Global patterns in endemicity and vulnerability of soil fungi. Global Change Biology 28, 6696–6710.
Thijs, S., Op De Beeck, M., Beckers, B., Truyens, S., Stevens, V., Van Hamme, J.D., Weyens, N., Vangronsveld, J., 2017. Comparative evaluation of four bacteria-specific primer pairs for 16S rRNA gene surveys. Frontiers in Microbiology 8, 494.
Thorn, R.G., Reddy, C.A., Harris, D., Paul, E.A., 1996. Isolation of saprophytic basidiomycetes from soil. Applied and Environmental Microbiology 62, 4288–4292.
Van der Westhuizen, J.P.J., Kock, J.L.F., Botha, A., Botes, P.J., 1994. The distribution of the õ3- and õ6-series of cellular long-chain fatty acids in fungi. Systematic and Applied Microbiology 17, 327–345.
Venables, W.N., Ripley, B.D., 2002. Modern Applied Statistics with S. 4th ed. New York: Springer.
Vestal, J.R., White, D.C., 1989. Lipid analysis in microbial ecology: quantitative approaches to the study of microbial communities. BioScience 39, 535–541.
Větrovský, T., Baldrian, P., 2013. The variability of the 16S rRNA gene in bacterial genomes and its consequences for bacterial community analyses. PLoS One 8, e57923.
Wallander, H., Ekblad, A., Godbold, D.L., Johnson, D., Bahr, A., Baldrian, P., Björk, R.G., Kieliszewska-Rokicka, B., Kjøller, R., Kraigher, H., Plassard, C., Rudawska, M., 2013. Evaluation of methods to estimate production, biomass and turnover of ectomycorrhizal mycelium in forests soils–A review. Soil Biology and Biochemistry 57, 1034–1047.
Wang, S.N., Cheng, J.K., Li, T., Liao, Y.C., 2020. Response of soil fungal communities to continuous crop** of flue-cured tobacco. Scientific Reports 10, 19911.
Weete, J.D., 1980. Lipid Biochemistry of Fungi and Other Organisms. New York: Springer.
Weete, J.D., Abril, M., Blackwell, M., 2010. Phylogenetic distribution of fungal sterols. PLoS One 5, e10899.
White, T.J., Bruns, T., Lee, S., Taylor, J., 1990. Amplification and direct sequencing of fungal ribosomal rna genes for phylogenetics. In: Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., eds. PCR Protocols: A Guide to Methods and Applications. San Diego: Academic Press, pp. 315–322.
Willers, C., van Rensburg, P.J.J., Claassens, S., 2015. Phospholipid fatty acid profiling of microbial communities-a review of interpretations and recent applications. Journal of Applied Microbiology 119, 1207–1218.
Wright, E.S., 2016. Using DECIPHER v2.0 to Analyze Big Biological Sequence Data in R. The R Journal 8, 352–359.
Yu, K.L., van den Hoogen, J., Wang, Z.Q., Averill, C., Routh, D., Smith, G.R., Drenovsky, R.E., Scow, K.M., Mo, F., Waldrop, M. P., Yang, Y.H., Tang, W.Z., De Vries, F.T., Bardgett, R.D., Manning, P., Bastida, F., Baer, S.G., Bach, E.M., García, C., Wang, Q.K., Ma, L.N., Chen, B.D., He, X.J., Teurlincx, S., Heijboer, A., Bradley, J.A., Crowther, T.W., 2022. The biogeography of relative abundance of soil fungi versus bacteria in surface topsoil. Earth System Science Data 14, 4339–4350.
Zelles, L., 1997. Phospholipid fatty acid profiles in selected members of soil microbial communities. Chemosphere 35, 275–294.
Zhang, Z.J., Qu, Y.Y., Li, S.Z., Feng, K., Wang, S., Cai, W.W., Liang, Y.T., Li, H., Xu, M.Y., Yin, H.Q., Deng, Y., 2017. Soil bacterial quantification approaches coupling with relative abundances reflecting the changes of taxa. Scientific Reports 7, 4837.
Zheng, W.S., Lehmann, A., Ryo, M., Vályi, K.K., Rillig, M.C., 2020. Growth rate trades off with enzymatic investment in soil filamentous fungi. Scientific Reports 10, 11013.
Acknowledgments
TC acknowledges funding by the Deutsche Forschungsgemeinschaft (DFG, grant number 465123751, SPP2322 SoilSystems). SH was partly supported by DFG grant HE 6183/5-1 and SM by MA4436/1-5. We thank Alberto Canarini and Kyle Mason-Jones for important insights on fungal storage mechanisms. Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
Electronic Supplementary Material
42832_2024_243_MOESM1_ESM.pdf
Revisiting soil fungal biomarkers and conversion factors: Interspecific variability in phospholipid fatty acids, ergosterol and rDNA copy numbers
42832_2024_243_MOESM2_ESM.docx
Revisiting soil fungal biomarkers and conversion factors: Interspecific variability in phospholipid fatty acids, ergosterol and rDNA copy numbers
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Camenzind, T., Haslwimmer, H., Rillig, M.C. et al. Revisiting soil fungal biomarkers and conversion factors: Interspecific variability in phospholipid fatty acids, ergosterol and rDNA copy numbers. Soil Ecol. Lett. 6, 240243 (2024). https://doi.org/10.1007/s42832-024-0243-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42832-024-0243-5