Abstract
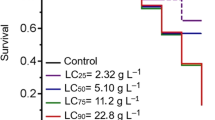
In this study, we assessed the larvicidal activity of aqueous extracts obtained from the leaves and roots of Plantago major L. and Plantago lagopus L. on fourth instar Culex pipiens L. larvae. Plant material was collected from two regions in Tunisia, Beja and Bizerte. The resulting extracts were prepared at concentrations of 10, 20, 50 and 100 ppm. The toxicity of the extracts was monitored over a 24-hour period. Our findings indicated that leaf extracts from Plantago lagopus L. collected in Bizerte exhibited higher toxicity compared to those collected in Beja up to 12 h, although these differences became insignificant at the 24-hour mark. Furthermore, extracts from the roots of Plantago lagopus L. from Bizerte demonstrated higher toxicity than those from Beja. Statistical analysis revealed no significant differences (P = 0.056) in the toxicity of leaf extracts at 100 ppm between the two species after 24 h of exposure. The root extracts exhibited lower toxicity, resulting in mortality rates ranging from 40 to 70% after 24 h. Specifically, the leaf extracts of Plantago major L. from Bizerte yielded a median lethal concentration (LC50) of 16.068 ppm ± 8.440. Additionally, the quantification of total polyphenols and flavonoids revealed distinct variations between species, plant organs, and geographical origins. Root and leaf extracts from Plantago major L. collected in Beja exhibited higher concentrations compared to those from Bizerte. Conversely, root extracts from Plantago lagopus L. collected in Bizerte displayed the highest concentrations. These preliminary findings provide a foundation for the development of a novel natural biocide for mosquito control, utilizing the distinct larvicidal properties of Plantago species extracts.







Similar content being viewed by others
Data availability
All data and analyzes are accessible in the manuscript and supplementary materials.
References
Abd El-Gawad AM, Mashaly IA, AbuZiada ME, Deweeb MR (2015) Phytotoxicity of three Plantago species on germination and seedling growth of hairy beggarticks (Bidenspilosa L.). Egypt. J Basic app Sci 2:303–309
Achakzai AKK, Achakzai P, Masood A, Kayani SA, Tareen RB (2009) Response of plant parts and age on the distribution of secondary metabolites on plants found in Quetta. Pak J Bot 41:2129–2135
Ahdiyah I, Purwan KI (2015) Pengaruh Ekstrak Daun Mangkokan (Nothopanax scutellarium) sebagai Larvasida Nyamuk Culex Sp. Jurnal Sains Dan Seni ITS 4(2):32–36
Aioub AAA, Zuo Y, Aioub AAA, Hu Z (2021) Biochemical and phytoremediation of Plantago major L. to protect tomato plants from the contamination of cypermethrin pesticide. Environ Sci Pollut 28:43992–44001. https://doi.org/10.1007/s11356-021-13853-2
Alves DS, Oliveira DF, Carvalho GA, dos Santos HM Jr, Carvalho DA, Santos MAI et al (2011) Plant extracts as an alternative to control Leucoptera coffeella (Guérin-Mèneville) (Lepidoptera: Lyonetiidae). Neotro Entomol 40:1–12
Arrese EL, Soulages JL (2010) Insect fat body: energy, metabolism, and regulation. Annu Rev Entomol 55:207–225. https://doi.org/10.1146/annurev-ento-112408-085356
Baba-Aissa A, Badaoui S (2016) Etude de l’activité biologique de quelques composés phénoliques, de deux plantes de la famille des ombellifères à l’égard de Galleria mellonella. Mémoire de Mastère 69
Bloor SJ (1995) A survey of extracts of New Zealand indigenous plants for selected biological activities. N Z J Bot 33(4):523–540. https://doi.org/10.1080/0028825X.1995.10410624
Boeing JS, Barizão ÉO, e Silva BC, Montanher PF, Almeida VC, Visentainer JV (2014) Evaluation of solvent effect on the extraction of phenolic compounds and antioxidant capacities from the berries: application of principal component analysis. Chem Cent J 8:48. https://doi.org/10.1186/s13065-014-0048-1
Boizot N, Charpentier JP (2006) Méthode Rapide d’évaluation Du contenu en composés phénoliques des organes d’un arbre forestier. Cahier Des Techniques d’INRA 79–82
Cachereul A (1997) Les moustiques: cycle de développement, aspects anatomophysiologiques et régulation du cycle ovarien, Thèse de Médecine Vétérinaire, Nantes 117p
Chiang LC, Chiang W, Chang MY, Ng LT, Lin CC (2002) Antiviral activity of Plantago major extracts and related compounds in vitro. Antiviral Res 55:53–62
Daghbouche S, Ammar I, Moalla Rekik D, Djazouli Z, Zebib B, Merah O (2020) Effect of phenological stages on essential oil composition of Cytisus triflorus L’Her. J King Saud Univ Sci 32(4):2383–2387. https://doi.org/10.1016/j.jksus.2020.03.020
El Husseiny IM, El Kholy SE, Othman AA (2014) Laboratory Testing of the toxicity of Jujube (Zizyphus Jujuba) Oil and Leaf extracts against Culex pipiens (Diptera: Culicidae). Afr Entomol 22(4):755–761. https://doi.org/10.4001/003.022.0411
Fei M (2016) The importance of phenology in studies of plant-herbivore-parasitoid interactions. 170 pages. PhD thesis, Wageningen University, Wageningen, NL. ISBN 978-94-6257-655-1
Ferreira C, Oliveira R (2021) Protective antifungal activity of Plantago major extract against the Phytopathogenic Fungi Phytophthora cinnamomi, Diplodia corticola and Colletotrichum species. Proceedings. 70(1):94. https://doi.org/10.3390/foods_2020-07678
Genç Y, Saraçolu I, Nagatsu A, Harput Ü (2010) Iridoid and Megastigman Glucosides from Plantago lagopus L. FABAD. J Pharm Sci 35:29–34
Ghnimi W, Dicko A, Khouja ML, El Ferchichi Ouarda H (2014) Larvicidal activity, phytochemical composition, and antioxidant properties of different parts of five populations of Ricinus communis L. Ind Crops Prod 56:43–51. https://doi.org/10.1016/j.indcrop.2014.02.036
Gonçalves S, Romano A (2016) The medicinal potential of plants from the genus Plantago (Plantaginaceae). Indus. Crops Prod 83:213–226
Guerra JGO, Cerna CE, Ochoa FYM, Landeros FJ, Aguirre ULA, Juárez AH (2020) Insecticidal activity of plant extracts against whitefly nymphs Bemisia tabaci (Hemiptera: Aleyrodidae) in laboratory. J Entomol Zool Stud 8(1):595–599
Harborne JB, Williams CA (2000) Advances in flavonoids research since 1992. Phytochem 55:481–504
Harput US, Genc Y, Saracoglu I (2012) Cytotoxic and antioxidative activities of Plantago lagopus L. and characterization of its bioactive compounds. Food Chem Toxicol 50:1554–1559
Holetz FB, Pessini GI, Sanches NR, Cortez A, Nakamura CV, Filho BP (2002) Screening of some plants used in the Brazilian folk medicine for the treatment of infectious diseases. Mem Inst Oswaldo Cruz 97:1027–1031
Hussan F, Osman BRH, MohdYusof MR, Kamaruddin NA, Othman F (2015) Plantago major treatment enhanced innate antioxidant activity in experimental acetaminophen toxicity. Asian Pac J Trop Biomed 5(9):728–732
Isah T (2019) Stress and defense responses in plant secondary metabolites production. Biol Res 52–39. https://doi.org/10.1186/s40659-019-0246-3
Isman MB (2006) Botanical insecticides, deterrents and repellents in modern agriculture and an increasingly regulated world. Annu Rev Entomol 51(1):45–66
Jahromi SG (2019) Extraction techniques of phenolic compounds from plants. Plant physiological aspects of phenolic compounds. 120 pp. ISBN: 978-1-78984-034-6. https://doi.org/10.5772/intechopen.84705
Kapp-Bitter AN, Dickhoefer U, Kreuzer M, Leiber F (2020) Mature herbs as supplements to ruminant diets: effects on in vitro ruminal fermentation and ammonia production. Anim Prod Sci 61:470–479
Khoshraftar Z, Shamel A, Safekordi AA, Ardjmand M, Zaefizadeh M (2020) Natural nanopesticides with origin of Plantago major seeds extract for Tribolium castaneum control. J Nanostructure Chem 10:255–264. https://doi.org/10.1007/s40097-020-00346-w
Kobeasy IM, Abdel-Fatah MO, Samiha M, El-Salam A, Zahret El-Ola (2011) Biochemical studies on Plantago major L. and Cyamopsis tetragonoloba L. Int J Biodivers Conserv 3(3):83–91
Köppen W (1918) Klassification Der Klimate Nach Temperatur, Niederschlag and Jahreslauf. Petermanns Geogr Mitt 64:193–203
Le Floc’h E, Loutfy B, Errol V (2010) Catalogue synonymique commenté de la flore de Tunisie. République tunisienne ministère de l’environnement et du développement durable banque nationale de gènes 277–280
Mohapatra C, Rengarajan K (1995) Manual on bioassays in the laboratory and their techniques. Cmfri Spec Publ 64:1–75
Mohsenzadeh S, Nazeri V, Mirtadzadini SM (2008) Chromosome numbers of fifteen species of Plantago L. (Plantaginaceae) from Iran. Iran J Bot 14:47–53
Nurhaifah D, Sukesi TW (2015) Efektifitas Air Perasan Kulit Jeruk Manis Sebagai Larvasida Nyamuk Aedes aegypti. Jurnal Kesehatan Masyarakat Nasional 9(3):2015
Nyunt TM, Lwin KK, Aye TT, Than MA, Chit K, Kyaw T, Hlaing OMT, Wun M, Win NN (2007) Antihypertensive effect of Plantago major Linn. Whole plant (ahkyawpaungtahtaung) on mild to moderate hypertensive patients. Myanmar Health Sci Res J 19:97–102
Orhan DD, Berrin O, Sanem H, Mecit V (2012) Assessment of antioxidant, antibacterial, antimycobacterial, and antifungal activities of some plants used as folk remedies in Turkey against dermatophytes and yeast-like fungi. Turk J Biol 36:672–686TUB. https://doi.org/10.3906/biy-1203-33
Oulebsir-Mohandkaci H, Aissa AB, Badaoui S, Bouyahiaoui H, Kaki SA, Mohammedi A (2018) Comparative study of the toxicity of phenolic compounds of coriander (Coriandrum sativum) and false fennel (Aneth Graveolens) on Galleria mellonella (Lepidoptera, Pyralidae). J Environ Integr 3:1–7
Pesantes-Sangay SJ, Calla-Poma RD, Requena-Mendizabal MF, Alvino-Vales MI, Millones- Gómez PA (2020) Chemical composition and antibacterial effect of Plantago major extract on periodontal pathogens. Pesquisa Brasileria Odontopediatria e Clínica Integrada.20:e0012. 2020. https://doi.org/10.1590/pboci.2020.100
Pratyusha S (2022) Phenolic compounds in the plant development and defense: an overview. Plant Stress Physiology - Perspectives in Agriculture. 224 pp. ISBN: 978-1-83969-867-5. https://doi.org/10.5772/intechopen.102873
Rahat AZ, Anjum S, Zehra FUB, Chishti R (2023) Antibacterial, insecticidal, antifungal and phytochemical screening of Alium Sativum, Nigela sativa and Plantago ovata. Sarhad J Agric 39(2):531–544
Regnault RC, Vincent C, Arnason JT (2012) Essential oils in insect control: low-risk products in a high-stakes world. Annu Rev Entomol 57:405–424
Reynaud J (2011) La flore du pharmacien, édition TEC & DOC, Paris. 256 pp. ISBN-13: 978-2743005290
Ribéreau-Gayon P (1968) Les composés phénoliques des végétaux. Editions Dunod, Paris, p 254
Romeh AA (2014) Phytoremediation of cyanophos insecticide by Plantago major L. in water. J Environ Health Sci Eng 12(38). https://doi.org/10.1186/2052-336X-12-38. http://www.ijehse.com/content/12/1/38
Rosłon W, Gontar Ł, Kosakowska O, Osińska E (2015) Yield and quality of plantain (Plantago major L.) herb in the second year of cultivation. Horticul Landsc Architect 36:21–32
Samuelsen AB (2000) The traditional uses, chemical constituents and biological activities of Plantago major L. a review. J Ethnopharmacol 71:1–21
Sanna F, Piluzza G, Campesi G, Molinu MG, Re GA, Sulas L (2022) Antioxidant contents in a Mediterranean Population of Plantago lanceolata L. Exploited for Quarry reclamation interventions. Plants 11:791
Schofield P, Mbugua DM, Pell AN (2001) Analysis of condensed tannins: a review. Anim Feed Sci Technicol 91:21–40
Sharma P, Mohan L, Dua KK, Srivastava CN (2011) Status of carbohydrate, protein and lipid profile in the mosquito larvae treated with certain phytoextracts. Asian Pac J Trop Med 4(4):301–304. https://doi.org/10.1016/S1995-7645(11)60090-4
Shepeleva VV, Nezhinskaya GI (2008) Immunoprotective activity of medicinal plants preparations infusion in immunodepression caused by cytostatics. Rastite ResurE 44:129–135
Shi L, Zhao W, Yang Z, Subbiah V, Suleria HAR (2022) Extraction and characterization of phenolic compounds and their potential antioxidant activities. Environ Sci Pollut Res Int 29(54):81112–81129. https://doi.org/10.1007/s11356-022-23337-6
Sotek Z, Białecka B, Pilarczyk B, Drozd R, Pilarczyk R, Tomza-Marciniak A et al (2019) Antioxidant activity and selenium and polyphenols content from selected medicinal plants natives from various areas abundant in selenium (Poland, Lithuania, and Western Ukraine). Processes 7:878
Tinkov AA, Nemereshina ON, Popova EV, Polyakova VS, Gritsenko VA, Nikonorov AA (2014) Plantago maxima leaves extract inhibits adipogenic action of a high-fat diet in female Wistar rats. Eur J Nutr 53:831–842
Turan M, Mammadov R (2021) Phenolic compounds Screening and potential of Larvicidal Activity of Water Extract of Cyclamen ciliciumBoiss. Heldr Nat Prod Biotechnol 1(1):1–8
Tutel B, Kandemir I, Kus S, Kence A (2005) Classification of Turkish Plantago L. species using numerical taxonomy. Turk J Bot 29:51–61
Velázquez-Fiz MP, Díaz-Lanza AM, Fernández-Matellano L (2000) Iridoids from Plantago Lagopus. Pharm Biol 38(4):268–270
Vigo E, Cepeda A, Gualillo O, Perez-Fernandez R (2005) In vitro anti-inflammatory activity of Pinus sylvestris and Plantago lanceolata extracts: effect on inducible NOS COX-1 COX-2 and their products in J774A.1 murine macrophages. J Pharm Phamacol 57:383–391
Zhishen J, Mengcheng T, Jianming W (1999) The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem 64:555–559
Acknowledgements
We thank Dr. Bejaoui Mustafa (Faculty of Sciences of Bizerte) for hel** us to identify the larvae stages of Culex pipiens L.
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors agree to publication.
Competing interests
The authors have declared that no competing interests exist.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bouali, A., D’hallewin, G. & Ouarda, H.E.F. Larvicidal activity on fourth instar Culex pipiens L. and phytochemical characteristics of aqueous extracts from leaves and roots of two species from the genus Plantago. Int J Trop Insect Sci 44, 759–770 (2024). https://doi.org/10.1007/s42690-024-01208-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-024-01208-6