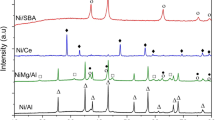
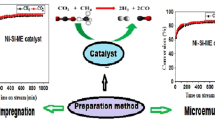
Abstract
A series of catalysts based on nickel supported on different simple oxides ZrO2, CeO2 and La2O3 were prepared by dry impregnation. These latter were prepared by microemulsion. The synthesized catalysts as well as their corresponding supports designated by Ni/ZrO2 Ni/CeO2 Ni/La2O3 were characterized by X-ray diffraction (XRD), the physisorption of nitrogen (N2) and Fourier Transform Infrared spectroscopy (FTIR). The synthesized and characterized catalysts were subsequently tested in the dry reforming of methane reaction to produce the synthesis gas. The study of the effect of the reaction temperature was carried out between 650 and 800 °C. Stability tests were carried out at 700 °C for 3 h of reaction time. The results obtained show that the catalytic performances of catalysts depends on structural properties and not on the good textural properties. Indeed, the catalyst with better textural properties is the least efficient in terms of activity and stability. The two catalysts Ni/CeO2 and Ni/ZrO2 are very active for all the temperatures studied and stable during 3 h of the test at 700 °C. On the other hand, the Ni/La2O3 catalyst exhibits low catalytic activity for all the temperatures studied and deactivate during the test time at 700 °C.







Similar content being viewed by others
References
Benson SW (1980) U.S. Patent 199: 533
Côme GM (1987) Brevet Français no. 2624115
Vedrenne I, Saint-Just J, Ben-Hadid A, Côme GM (1990) Catal Today 6:381
Dissanayake D, Rosynek MP, Kharas KCC, Lunsford JH (1991) J Catal 132:117–127
O’Connor AM, Schuurman Y, Ross JRH, Mirodatos C (2006) Catal Today 115:191–198
Nemeth M, Schay Z, Sranko D, Karolyi J, Safran G, Sajo I, Horvath A (2015) Appl Catal A: Gen 504(608):620
Abasaeed AE, Sofiu ML, Acharya K, Osman AI, Fakeeha AH, Al-Otaibi RL, Ibrahim AA, Al-Awadi AS, Bayahia H, Al-Zahrani SA, Kumar R, Al-Fatesh AS (2023) Energy Sci Eng 11:1436–1450
Ikkour K, Sellam D, Kiennemann A, Tezkratt S, Cherifi O (2009) Catal Lett 132:213–217
Gonzalez-Delacruz VM, Ternero F, Pereñíguez R, Caballero A, Holgado JP (2010) Appl Catal A: Gen 384:1–9
Sellam D, Ikkour K, Dekkar S, Messaoudi H, Belaid T, Roger AC (2019) BCREC 14:568–578
Djaidja A, Libs S, Kiennemann A, Barama A (2006) Catal Today 113:194–200
Gadalla AM, Bower B (1987) Chem Eng Sci 43:3049–3062
Hally W, Bitter JH, Seshan K, Lercher JA, Ross JRH (2001) Stud Surf Sci Catal 88:167–173
Gucci L, Erdohelyi A (2012) Catalysis for alternative energy. Springer-Verlag, New York
Ryi S-K, Lee S-W, Park J-W, Oh D-K, Park J-S, Kim SS (2014) Catal Today 236:49–56
Srinivas D, Satyanarayana CVV, Potdar HS, Ratnasamy P (2003) Appl Catal A: Gen 246:323–334
Biswas P, Kunzru D (2008) Chem Eng J 136:41–49
Barbero J, Peña MA, Campos-Martin JM, Fierro JLG, Arias PL (2003) Catal Lett 87:211–218
Dekkar S, Tezkratt S, Sellam D, Parkhomenko K, Martinez-Martin A, Roger AC (2020) Catal Lett 150:2180–2199
Barroso-Quiroga MM, Castro-Luna AE (2010) Int J Hydrogen Energy 35:6052–6056
Lu Y, Li S, Guo L, Zhang X (2010) Int J Hydrogen Energy 35:7161–7168
Li Z, Mo L, Kathiraser Y, Kawi S (2014) ACS Catal 4:1526–1536
Cai X, Dong X, Lin W (2008) J Nat Gas Chem 17:98–102
Zhu J, Peng X, Yao L, Tong D, Hu C (2012) Catal. Sci Technol 2:529–537
Wang N, Qian W, Chu W, Wei F (2016) Catal Sci Technol 6:3594–3605
Vasiliades MA, D**ovic P, Pintar A, Kovac J, Efstathiou AM (2017) Catal Sci Technol 7:5422–5434
Mo L, Leong KKM, Kawi S (2014) Catal Sci Technol 4:2107–2114
Xu L, Miao Z, Song H, Chen W, Chou L (2014) Catal Sci Technol 4:1759–1770
Majewski AJ, Wood J, Bujalski W (2013) Int J Hydrog Energy 38:14531–14541
Trimm DL (1980) Chapter 9. Elsevier, New York
Takahashi R, Sato S, Sodesawa T, Tomiyama S (2005) Appl Catal A Gen 286:142–147
Xu S, Zhao R, Wang X (2004) Fuel Process Technol 86:123–133
Rahaman SKM, Bardhanb A, Mandal T, Chakraborty M, Karmakar K, Dhibar S, Sharma S, Chakravarty M, Ibrahim SM, Saha B (2023) N J Chem 47:10309–10321
Rahaman SKM, Chakraborty M, Mandal T, Kundu S, Dhibar S, Kumar D, Ibrahim SM, Ibrahim SM, Chakravarty M, Saha B (2023) J Mol Liquids 372:121204
Kundu S, Karmakar P, Rahaman SKM, Mitra M, Rajwar S, Dhibar S, Layek M, Sar P, Saha B (2023) N J Chem 47:4364–4373
Padi P, Shelly L, Komarala EP, Schweke D, Hayun S, Rosen BA (2020) Catal Commun 138:105951
Köck EM, Kogler M, Bielz T, Klötzer B, Penner S (2013) J Phys Chem C Nanomater Interfaces 117:17666–17673
Bachiller-Baeza B, Rodriguez-Ramos I, Guerrero-Ruiz A (1998) Langmuir 14:3556–3564
Montoya JA, Romero-Pascual E, Gimon C, Del Angel P, Monzon A (2000) Catal Today 63:71–85
Zhang ZL, Verykios XE (1995) J Chem Soc Chem Commun 71–72
Calvino-Casilda V, Martin-Aranda R, Sobczak I, Ziolek M (2006) Appl Catal A 303:121–130
Yahi N, Menad SI (2015) Green Process Synth 6:479–486
Nassos S, Elm Svensson E, Nilson M, Boutonnet M, Jairas S (2006) Appl Catal B 64
Jahanjeer A, Tokeer A, Rmanujachary KV, Lofland SE, Ganguli AK (2008) J Colloid Interface Sci 321
Damaskinos CM, Vasiliades MA, Stathopoulos VN, Efstathiou AM (2019) Catalysts 9:621
Khan SB, Faisal M, Rahman MM, Akhtar K, Asiri AM, Khan A, Alamry KA (2013) Int J Electrochem Sci 8:7284–7297
Zamiri R, Ahangar HA, Kaushal A, Zakaria A, Zamiri G, Tobaldi D, Ferreira JMF (2015) Dielectrical properties of CeO2 nanoparticles at different temperatures. PLoS ONE. https://doi.org/10.1371/journal.pone.0122989
Firdous A, Quasim I, Ahmad MM, Kotru PN (2009) J Cryst Growth 311:3855–3862
Nakamato K (1986) Infrared and Raman spectra of inorganic and coordination compound, 4th edn. Wiley, New York
Fernandes JDG, Melo DMA, Zinner LB, Salustiano CM, Silva ZR, Martinelli AE, Cerqueira M, AlvesJúnior C, Longo E, Bernardi MIB (2002) Mater Lett 53:122–125
Lustemberg PG, Ramírez PJ, Liu ZY, Gutiérrez RA, Grinter DG, Carrasco J, Senanayake SD, Rodriguez JA, Ganduglia-Pirovano MV (2016) ACS Catal 6:8184–8191
Liu ZY, Grinter DC, Lustemberg PG, Nguyen-Phan T-D, Zhou YH, Luo S, Waluyo I, Crumlin EJ, Stacchiola DJ, Zhou J (2016) Angew Chem Int Ed 55:7455–7459
Singh S, Zubenko D, Rosen BA (2016) ACS Catal 6:4199–4205
Dacquin J-P, Sellam D, Batiot-Dupeyrat C, Tougerti A, Duprez D (2014) R S Chem Sustain Chem 7:631–637
Rezaei M, Alavi SM, Sahebdelfar S, **nmei L, Qian L, Yan ZF (2007) Fuels 21:581–589
Li X, Li D, Tian H, Zeng L, Zhao ZJ, Gong J (2017) Appl Catal B 202:683–694
Mustu H, Yasyerli S, Yasyerli N, Dogu G, Dogu T, D**ovic P, Pintar A (2015) Int J Hydrogen Energy 40:3217–3228
Sophiana IC, Iskandar F, Devianto H, Nishiyama N, Bodhi YW (2022) Nanomaterials 12:1556
Funding
This study is funded by Laboratoire de Genie Chimique et de Chimie Appliquée de l'Université Mouloud Mammeri de Tizi–Ouzou.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dekkar, S. Dry Reforming of Methane Over Ni/ZrO2, Ni/CeO2 and Ni/La2O3 Catalysts: Role of Support Nature and its Synthesis by Microemulsion Method. Chemistry Africa 7, 1–11 (2024). https://doi.org/10.1007/s42250-023-00730-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42250-023-00730-3