Abstract
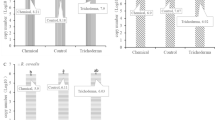
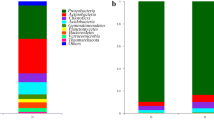
Black shank disease incited by Phytophthora nicotianae is one of the most devastating soil-borne diseases of tobacco (Nicotiana tabacum L.). Crop rotation is an effective strategy for the management of this disease. However, the efficacy of rotation with different crops in controlling tobacco black shank remains obscure. In this study, 10 different preceding crops, including rape, wheat, barley, broad bean, pea, garlic, ryegrass, cow vetch, alfalfa, and milk vetch, were selected in fields with an history of severe tobacco black shank. The effectiveness of crop rotation on tobacco black shank disease control was quantitatively determined as amount of Phytophthora nicotianae using (1) fluorescence quantitative Polymerase Chain Reaction (qPCR), (2) tobacco leaf disk assays, and (3) soil dilution plating assays. In addition, the bacterial assemblage associated with rhizosphere of different crops was analyzed by Terminal Restriction Fragment Length Polymorphism (T-RFLP). Results showed that ryegrass, garlic, and barley can effectively reduce the levels of P. nicotianae in the soil, with a consequent decrease of both morbidity and disease index. The rhizosphere bacterial community assemblages changed depending on the different preceding crops.



Similar content being viewed by others
References
Antonopoulos DF, Thomas M, Mila AL (2010) Effects of chemical control, cultivar resistance, and structure of cultivar root system on black shank incidence of tobacco. Plant Dis 94:613–620. https://doi.org/10.1094/PDIS-94-5-0613
Berendsen RL, Pieterse CMJ, Bakker PAHM (2012) The rhizosphere microbiome and plant health. Trends Plant Sci 17:478–486. https://doi.org/10.1016/j.tplants.2012.04.001
Cartwright DK, Spurr H (1998) Biological control of Phytophthora parasitica var. nicotianae on tobacco seedlings with non-pathogenic binucleate Rhizoctonia fungi. Soil Biol Biochem 30:1879–1884. https://doi.org/10.1016/S0038-0717(98)00019-4
Chuan YC, Zhang LM, Jiao YG, Luo LF, Fang YT, Liao JJ, Ji SG, Zhu SS, Yang M (2016) Control effects of tobacco and garlic rotation on tobacco black shank and a preliminary study on the inhibition mechanism. Acta Tabacaria Sinica 22(5):55–62. https://doi.org/10.16472/j.chinatobacco.2016.200
Cohen Y, Coffey MD (1986) Systemic fungicides and the control of oomycetes. Annu Rev Phytopathol 24:311–339. https://doi.org/10.1146/annurev.py.24.090186.001523
Don RH, Cox PT, Wainwright BJ, Baker K, Mattick JS (1991) ‘Touchdown’ PCR to circumvent spurious priming during gene amplification. Nucleic acids res 19:4008. https://doi.org/10.1093/nar/19.14.4008
Enwall K, Nyberg K, Bertilsson S, Cederlund H, Stenström J, Hallin S (2007) Long-term impact of fertilization on activity and composition of bacterial communities and metabolic guilds in agricultural soil. Soil Biol Biochem 39:106–115. https://doi.org/10.1016/j.soilbio.2006.06.015
Erwin DC, Ribeiro OK (1996) Phytophthora Diseases Worldwide. American Phytopathological Society Press, St. Paul, MN
Fu ZY, Zhang XY, Zhang XF, Zhou HJ, Qin YH, Ma J, Han JQ, Ye XF (2018) Effect of continuous crop** on quality of flue-cured tobacco leaves and carbon pool in tobacco growing soil. Journal of Northwest A&F University. Nat Sci Ed 46:16–22. https://doi.org/10.13207/j.cnki.jnwafu.2018.08.003
Garbeva P, van Veen JA, van Elsas JD (2004) MICROBIAL DIVERSITY IN SOIL: Selection of Microbial Populations by Plant and Soil Type and Implications for Disease Suppressiveness. Annu Rev Phytopathol 42:243–270. https://doi.org/10.1146/annurev.phyto.42.012604.135455
Geng K, Luo WF, Yang YL (2002) Population Distribution of Phytophthora parasitica var. nicotianae in Soil of Tobacco Field. Journal of Yunnan Agricultural University 17:389–391. https://doi.org/10.1006/jfls.2001.0409
Hu JF, Sang EJ, Liu JJ, Ran SF, Jiang YL (2013) Investigation on Tobacco Black Shank in Different Climatic Regions of Guizhou Province. Guizhou Agricultural Sciences 11:96–99
Janvier C, Villeneuve F, Alabouvette C, Edel-Hermann V, Mateille T, Steinberg C (2007) Soil health through soil disease suppression: which strategy from descriptors to indicators. Soil Biol Biochem 39(1):1–23. https://doi.org/10.1016/j.soilbio.2006.07.001
Kennedy AC, Smith KL (1995) Soil microbial diversity and the sustainability of agricultural soils. Plant Soil 70(1):75–86. https://doi.org/10.1007/BF02183056
Kent AD, Smith DJ, Benson BJ, Triplett EW (2003) Web-Based Phylogenetic Assignment Tool for Analysis of Terminal Restriction Fragment Length Polymorphism Profiles of Microbial Communities. Applied and Environmental Microbiology 69:6768–6776. https://doi.org/10.1128/AEM.69.11.6768-6776.2003
Larkin RP, Ristaino JB, Campbell CL (1995) Detection and quantification of Phytophthora capsici in soil. Phytopathology 85:1057–1063. https://doi.org/10.1094/Phyto-85-1057
Liu F, Song J, Fan Y, Mou W, ** J, Hu L (2015) A quantitative PCR method for detecting Phytophthora parasitica var. nicotianae in tobacco planting soil based on LNA sensitization. Tobacco Science & Technology 48(12):14–19. https://doi.org/10.16135/j.issn1002-0861.20151203
Lucas GB (1975) Diseases of Tobacco, 3rd edn. Fuquay-Varina, NC: Harold E. Parker and Sons. 115–141. https://doi.org/10.1016/0048-4059(76)90042-4
Mazzola M (2004) Assessment and management of soil community structure for disease suppression. Annu Rev Phytopathol 42:35–59. https://doi.org/10.1146/annurev.phyto.42.040803.140408
Meng Y, Zhang Q, Ding W, Shan W (2014) Phytophthora parasitica: a model oomycete plant pathogen. Mycology 5(2):43–51. https://doi.org/10.1080/21501203.2014.917734
Nilsson WB, Strom MS (2002) Detection and identification of bacterial pathogens of fish in kidney tissue using terminal restriction fragment length polymorphism (T-RFLP) analysis of 16S rRNA genes. Dis Aquat Org 48:175–185. https://doi.org/10.3354/dao048175
Olsson S, Alström S (2000) Characterization of bacteria in soils under barley monoculture and crop rotation. Appl Soil Ecol 32:1443–1451. https://doi.org/10.1016/S0038-0717(00)00062-6
Petersen LW, Moldrup P, Jacobsen OH, Rolston DE (1996) Relations Between Specific Surface Area and Soil Physical and Chemical Properties. Soil Science 161(1):9–21. https://doi.org/10.1097/00010694-199601000-00003
Shang ZQ (2007) Recent research advance on pathogen and disease occurrence of tobacco black shank and its integrated control. Review of China Agricultural Science and Technology 9:73–76
Tian F, Ding Y, Du B (2009) Genetic Diversity of Siderophore-producing Bacteria of Tobacco Rhizosphere. Braz J Microbiol 40:276–284. https://doi.org/10.1590/S151783822009000200013
Townsend GR, Heuberger JW (1943) Methods for estimating losses caused by diseases in fungicide experiments. Plant Dis 27:340–343
van Os GJ, van Ginkel JH (2001) Suppression of Pythium root rot in bulbous Iris in relation to biomass and activity of the soil microflora. Soil Biol Biochem 32:1447–1454. https://doi.org/10.1016/S0038-0717(01)00053-0
Volossiouk T, Robb EJ, Nazar RN (1995) Direct DNA extraction for PCR-mediated assays of soil organisms. Appl Environ Microb 61:3972–3976. https://doi.org/10.1002/bit.260480315
Wang J, Kong FY (2002) Integrated Control of Tobacco Black Shank Disease. Tobacco Science & Technology 8:45–47
Weller DM (1988) Biological control of soil-borne plant pathogens in the rhizosphere with bacteria. Annu Rev Phytopathol 73:463–469. https://doi.org/10.1146/annurev.py.26.090188.002115
Wolf FA (1935) Tobacco diseases and decays. Durham, North Carolina. 258–269
Wu WT, Dong Y, Wang XQ, Chen ZS, Lv DF, Yang CG, **e Y, Wang Y (2019) Effects of Marigold-tobacco Rotation on Soil Nematode Community Composition. Southwest China J Agric Sci 02:342–348. https://doi.org/10.16213/j.cnki.scjas.2019.2.020
Yang XB (2016) The alleviation mechanism of tobacco-garlic rotation and interplanting on soil continuous crop** obstacles. Chengdu, China, Sichuan Agriculture University, Ph. D Thesis.
Zhang K, **e LL, Wu YJ, Zhang XQ, Yang TZ (2015) Research Progress on Occurrence of Tobacco Black Shank and its Integrated Control. Journal of Agricultural Science and Technology 04:62–70. https://doi.org/10.13304/j.nykjdb.2015.1
Funding
This research was funded by the Yunnan Academy of Tobacco Agricultural Sciences (grant numbers 2020530000241012, 2019530000241007, 2020530000241013 and 2022530000241019), Yunnan Provincial Science and Technology Department (grant numbers 202001AU070012) and National Natural Science Foundation of China (grant numbers 32102161).
Author information
Authors and Affiliations
Contributions
D.F. prepared the materials and conducted the experiments; data analyzed by Z.X; X.G. wrote the original draft with C.H and C.L. review and editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Human and animal rights and informed consent
This article does not contain any studies with human participants or animals performed by any of the authors. It is original and has not been published elsewhere.
Conflict of interest
All authors declare that no competing interests exist.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gai, XT., Lu, CH., **a, ZY. et al. Crop rotation suppresses tobacco black shank disease incited by Phytophthora nicotianae and influenced the structure of rhizosphere bacterial communities. J Plant Pathol 105, 673–682 (2023). https://doi.org/10.1007/s42161-023-01411-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42161-023-01411-3