Abstract
Purpose
Morbid obesity (BMI > 40) is often accompanied by metabolic disorders. In adipose tissue, serine/threonine kinase PKBβ/AktΙΙ plays a role in glucose uptake, mediated by glucose transporter 4 (GLUT4). The insulin pathway also affects aquaglyceroporin-7 (AQP7), which mediates lipolysis-derived glycerol efflux into the bloodstream. The aim of our study was to investigate the molecular mechanisms in adipocytes of adults with morbid obesity that may lead to insulin resistance (IR) and diabetes mellitus type 2 (DM2) in morbid obesity.
Methods
Primary in vitro adipocyte cultures were developed from surgical biopsies from visceral (Visc), abdominal (Sub), and gluteal subcutaneous (Glut) fat depots, from 20 lean adults and 36 adults with morbid obesity (OB), divided into two groups: 20 without (MOW) and 16 with DM2 (MODM). mRNA and protein expression (PE) of AktΙΙ, AQP7, and GLUT4 were studied with RT-PCR and Western immunoblotting (WI), respectively.
Results
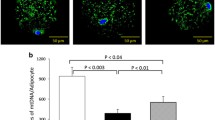
The PE of (1) AktII and basal phosphorylated AktII (pAktII) showed no difference within the groups, (2) the 37 kDa and 34 kDa isoforms of AQP7 were decreased in Visc/Sub from OB/MOW/MODM, (3) GLUT4 was decreased in Visc/Sub from OB/MOW/MODM, and (4) the 34 kDa isoform of AQP7 was decreased in Sub of MODM compared with MOW.
Conclusions
Decreased 37 kDa (presented in this study as a novel isoform) and 34 kDa isoforms of AQP7 in MOW and MODM may cause reduced lipolysis, enhancement of adipocyte hypertrophy, and impairment of insulin, signaling possibly reflected by low GLUT4 expression. This may potentially cause systemic IR, since decreased adipose GLUT4 expression may affect whole-body insulin sensitivity, increasing the risk for DM2. Furthermore, decreased subcutaneous AQP7 34 kDa could represent an early marker of IR.




Similar content being viewed by others
References
James WP (2008) The epidemiology of obesity: the size of the problem. J Intern Med 263:336–352
Goossens GH (2008) The role of adipose tissue dysfunction in the pathogenesis of obesity-related insulin resistance. Physiol Behav 94:206–218
Sun K, Kusminski CM, Scherer PE (2011) Adipose tissue remodeling and obesity. J Clin Invest 121:2094–2101
Kohn AD, Summers SA, Birnbaum MJ, Roth RA (1996) Expression of a constitutively active Akt Ser/Thr kinase in 3T3-L1 adipocytes stimulates glucose uptake and glucose transporter 4 translocation. J Biol Chem 271:31372–31378
Su X, Lodhi IJ, Saltiel AR, Stahl PD (2006) Insulin-stimulated interaction between insulin receptor substrate 1 and p85alpha and activation of protein kinase B/Akt require Rab5. J Biol Chem 281:27982–27990
Deepa SS, Dong LQ (2009) APPL1: role in adiponectin signaling and beyond. Am J Physiol Endocrinol Metab 296:E22–E36
Collins JM, Neville MJ, Pinnick KE, Hodson L, Ruyter B, van Dijk TH et al (2011) De novo lipogenesis in the differentiating human adipocyte can provide all fatty acids necessary for maturation. J Lipid Res 52:1683–1692
Kahn BB, Rossetti L, Lodish HF, Charron MJ (1991) Decreased in vivo glucose uptake but normal expression of GLUT1 and GLUT4 in skeletal muscle of diabetic rats. J Clin Invest 87:2197–2206
Abel ED, Peroni O, Kim JK, Kim YB, Boss O, Hadro E et al (2001) Adipose-selective targeting of the GLUT4 gene impairs insulin action in muscle and liver. Nature. 409:729–733
Bernat-Karpinska M, Czech A, Piatkiewicz P, Wierzbicki P, Gorski A (2010) Cellular glucose transport disturbances as a marker of the pre-diabetic state - pathogenetic and clinical significance of the assessment of GLUT4 expression. Endokrynol Pol 61:269–274
Rodriguez A, Catalan V, Gomez-Ambrosi J, Fruhbeck G (2011) Aquaglyceroporins serve as metabolic gateways in adiposity and insulin resistance control. Cell Cycle 10:1548–1556
Gambert S, Helies-Toussaint C, Grynberg A (2007) Extracellular glycerol regulates the cardiac energy balance in a working rat heart model. Am J Physiol Heart Circ Physiol 292:H1600–H1606
Matsumura K, Chang BH, Fujimiya M, Chen W, Kulkarni RN, Eguchi Y et al (2007) Aquaporin 7 is a beta-cell protein and regulator of intraislet glycerol content and glycerol kinase activity, beta-cell mass, and insulin production and secretion. Mol Cell Biol 27:6026–6037
Rodriguez A, Catalan V, Gomez-Ambrosi J, Garcia-Navarro S, Rotellar F, Valenti V et al (2011) Insulin- and leptin-mediated control of aquaglyceroporins in human adipocytes and hepatocytes is mediated via the PI3K/Akt/mTOR signaling cascade. J Clin Endocrinol Metab 96:E586–E597
Marrades MP, Milagro FI, Martinez JA, Moreno-Aliaga MJ (2006) Differential expression of aquaporin 7 in adipose tissue of lean and obese high fat consumers. Biochem Biophys Res Commun 339:785–789
Oikonomou E, Kostopoulou E, Rojas-Gil AP, Georgiou G, Spiliotis BE (2018) Adipocyte aquaporin 7 (AQP7) expression in lean children and children with obesity. Possible involvement in molecular mechanisms of childhood obesity. J Pediatr Endocrinol Metab
Lebeck J, Ostergard T, Rojek A, Fuchtbauer EM, Lund S, Nielsen S et al (2012) Gender-specific effect of physical training on AQP7 protein expression in human adipose tissue. Acta Diabetol 49(Suppl 1):S215–S226
Shen FX, Gu X, Pan W, Li WP, Li W, Ye J et al (2012) Over-expression of AQP7 contributes to improve insulin resistance in adipocytes. Exp Cell Res 318:2377–2384
Laforenza U, Gastaldi G, Grazioli M, Cova E, Tritto S, Faelli A et al (2005) Expression and immunolocalization of aquaporin-7 in rat gastrointestinal tract. Biol Cell 97:605–613
McAuley KA, Williams SM, Mann JI, Walker RJ, Lewis-Barned NJ, Temple LA et al (2001) Diagnosing insulin resistance in the general population. Diabetes Care 24:460–464
Mifflin MD, St Jeor ST, Hill LA, Scott BJ, Daugherty SA, Koh YO (1990) A new predictive equation for resting energy expenditure in healthy individuals. Am J Clin Nutr 51:241–247
Karvela A, Rojas-Gil AP, Samkinidou E, Papadaki H, Pappa A, Georgiou G et al (2010) Endocannabinoid (EC) receptor, CB1, and EC enzymes’ expression in primary adipocyte cultures of lean and obese pre-pubertal children in relation to adiponectin and insulin. J Pediatr Endocrinol Metab 23:1011–1024
Kurowski TG, Lin Y, Luo Z, Tsichlis PN, Buse MG, Heydrick SJ et al (1999) Hyperglycemia inhibits insulin activation of Akt/protein kinase B but not phosphatidylinositol 3-kinase in rat skeletal muscle. Diabetes. 48:658–663
Carvalho E, Eliasson B, Wesslau C, Smith U (2000) Impaired phosphorylation and insulin-stimulated translocation to the plasma membrane of protein kinase B/Akt in adipocytes from type II diabetic subjects. Diabetologia. 43:1107–1115
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC (1985) Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 28:412–419
Manolopoulos KN, Karpe F, Frayn KN (2010) Gluteofemoral body fat as a determinant of metabolic health. Int J Obes 34:949–959
Wakayama Y, Hirako S, Ogawa T, Jimi T, Shioda S (2014) Upregulated expression of AQP 7 in the skeletal muscles of obese ob/ob mice. Acta Histochem Cytochem 47:27–33
Lebeck J, Sondergaard E, Nielsen S (2018) Increased AQP7 abundance in skeletal muscle from obese men with type 2 diabetes. Am J Physiol Endocrinol Metab 315:E367–EE73
Fruhbeck G, Lopez M, Dieguez C (2007) Role of caveolins in body weight and insulin resistance regulation. Trends Endocrinol Metab 18:177–182
Parton RG, del Pozo MA (2013) Caveolae as plasma membrane sensors, protectors and organizers. Nat Rev Mol Cell Biol 14:98–112
Le Lay S, Krief S, Farnier C, Lefrere I, Le Liepvre X, Bazin R et al (2001) Cholesterol, a cell size-dependent signal that regulates glucose metabolism and gene expression in adipocytes. J Biol Chem 276:16904–16910
Cifuentes M, Albala C, Rojas CV (2008) Differences in lipogenesis and lipolysis in obese and non-obese adult human adipocytes. Biol Res 41:197–204
Funding
Funding was provided by the University of Patras, School of Medicine, Patras, Greece.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Informed consent was obtained from all subjects. The study was approved by the Ethical Committee of the University Hospital of Patras, Greece.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mourelatou, R., Kostopoulou, E., Rojas-Gil, A.P. et al. Decreased adipocyte glucose transporter 4 (GLUT4) and aquaglyceroporin-7 (AQP7) in adults with morbid obesity: possible early markers of metabolic dysfunction. Hormones 18, 297–306 (2019). https://doi.org/10.1007/s42000-019-00130-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42000-019-00130-8